| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cholecystokinin receptor type A |
|---|
| Ligand | BDBM50008170 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_47644 (CHEMBL659915) |
|---|
| Ki | 129±n/a nM |
|---|
| Citation |  Cappelli, A; Anzini, M; Vomero, S; Menziani, MC; De Benedetti, PG; Sbacchi, M; Clarke, GD; Mennuni, L Synthesis, biological evaluation, and quantitative receptor docking simulations of 2-[(acylamino)ethyl]-1,4-benzodiazepines as novel tifluadom-like ligands with high affinity and selectivity for kappa-opioid receptors. J Med Chem39:860-72 (1996) [PubMed] Article Cappelli, A; Anzini, M; Vomero, S; Menziani, MC; De Benedetti, PG; Sbacchi, M; Clarke, GD; Mennuni, L Synthesis, biological evaluation, and quantitative receptor docking simulations of 2-[(acylamino)ethyl]-1,4-benzodiazepines as novel tifluadom-like ligands with high affinity and selectivity for kappa-opioid receptors. J Med Chem39:860-72 (1996) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cholecystokinin receptor type A |
|---|
| Name: | Cholecystokinin receptor type A |
|---|
| Synonyms: | CCKAR_RAT | Cckar | Cholecystokinin peripheral | Cholecystokinin receptor | Cholecystokinin receptor type A |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 49676.37 |
|---|
| Organism: | RAT |
|---|
| Description: | Cholecystokinin central 0 RAT::P30551 |
|---|
| Residue: | 444 |
|---|
| Sequence: | MSHSPARQHLVESSRMDVVDSLLMNGSNITPPCELGLENETLFCLDQPQPSKEWQSALQI
LLYSIIFLLSVLGNTLVITVLIRNKRMRTVTNIFLLSLAVSDLMLCLFCMPFNLIPNLLK
DFIFGSAVCKTTTYFMGTSVSVSTFNLVAISLERYGAICRPLQSRVWQTKSHALKVIAAT
WCLSFTIMTPYPIYSNLVPFTKNNNQTANMCRFLLPSDAMQQSWQTFLLLILFLLPGIVM
VVAYGLISLELYQGIKFDASQKKSAKEKKPSTGSSTRYEDSDGCYLQKSRPPRKLELQQL
SSGSGGSRLNRIRSSSSAANLIAKKRVIRMLIVIVVLFFLCWMPIFSANAWRAYDTVSAE
KHLSGTPISFILLLSYTSSCVNPIIYCFMNKRFRLGFMATFPCCPNPGPPGVRGEVGEEE
DGRTIRALLSRYSYSHMSTSAPPP
|
|
|
|---|
| BDBM50008170 |
|---|
| n/a |
|---|
| Name | BDBM50008170 |
|---|
| Synonyms: | 4-(3,4-Dichloro-benzoylamino)-4-dipentylcarbamoyl-butyric acid | CHEMBL24938 | Lorglumide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H32Cl2N2O4 |
|---|
| Mol. Mass. | 459.406 |
|---|
| SMILES | CCCCCN(CCCCC)C(=O)C(CCC(O)=O)NC(=O)c1ccc(Cl)c(Cl)c1 |
|---|
| Structure | 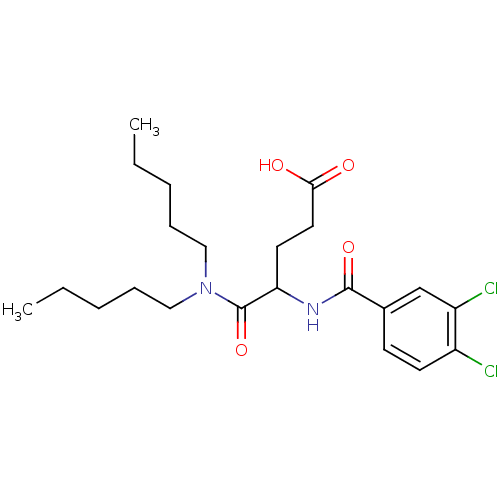
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Cappelli, A; Anzini, M; Vomero, S; Menziani, MC; De Benedetti, PG; Sbacchi, M; Clarke, GD; Mennuni, L Synthesis, biological evaluation, and quantitative receptor docking simulations of 2-[(acylamino)ethyl]-1,4-benzodiazepines as novel tifluadom-like ligands with high affinity and selectivity for kappa-opioid receptors. J Med Chem39:860-72 (1996) [PubMed] Article
Cappelli, A; Anzini, M; Vomero, S; Menziani, MC; De Benedetti, PG; Sbacchi, M; Clarke, GD; Mennuni, L Synthesis, biological evaluation, and quantitative receptor docking simulations of 2-[(acylamino)ethyl]-1,4-benzodiazepines as novel tifluadom-like ligands with high affinity and selectivity for kappa-opioid receptors. J Med Chem39:860-72 (1996) [PubMed] Article