| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prostaglandin G/H synthase 1/2 |
|---|
| Ligand | BDBM50076640 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_51502 (CHEMBL665837) |
|---|
| IC50 | 60±n/a nM |
|---|
| Citation |  Connolly, PJ; Wetter, SK; Beers, KN; Hamel, SC; Chen, RH; Wachter, MP; Ansell, J; Singer, MM; Steber, M; Ritchie, DM; Argentieri, DC N-hydroxyurea and hydroxamic acid inhibitors of cyclooxygenase and 5-lipoxygenase. Bioorg Med Chem Lett9:979-84 (1999) [PubMed] Connolly, PJ; Wetter, SK; Beers, KN; Hamel, SC; Chen, RH; Wachter, MP; Ansell, J; Singer, MM; Steber, M; Ritchie, DM; Argentieri, DC N-hydroxyurea and hydroxamic acid inhibitors of cyclooxygenase and 5-lipoxygenase. Bioorg Med Chem Lett9:979-84 (1999) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prostaglandin G/H synthase 1/2 |
|---|
| Name: | Prostaglandin G/H synthase 1/2 |
|---|
| Synonyms: | Cyclooxygenase |
|---|
| Type: | n/a |
|---|
| Mol. Mass.: | n/a |
|---|
| Description: | ASSAY_ID of ChEMBL is 1666355 |
|---|
| Components: | This complex has 2 components. |
|---|
| Component 1 |
| Name: | Prostaglandin G/H synthase 2 |
|---|
| Synonyms: | Cox-2 | Cox2 | Cyclooxygenase | PGH2_RAT | Ptgs2 |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 69173.51 |
|---|
| Organism: | RAT |
|---|
| Description: | COX-2 0 RAT::P35355 |
|---|
| Residue: | 604 |
|---|
| Sequence: | MLFRAVLLCAALALSHAANPCCSNPCQNRGECMSIGFDQYKCDCTRTGFYGENCTTPEFL
TRIKLLLKPTPNTVHYILTHFKGVWNIVNNIPFLRNSIMRYVLTSRSHLIDSPPTYNVHY
GYKSWEAFSNLSYYTRALPPVADDCPTPMGVKGNKELPDSKEVLEKVLLRREFIPDPQGT
NMMFAFFAQHFTHQFFKTDQKRGPGFTRGLGHGVDLNHVYGETLDRQHKLRLFQDGKLKY
QVIGGEVYPPTVKDTQVDMIYPPHVPEHLRFAVGQEVFGLVPGLMMYATIWLREHNRVCD
ILKQEHPEWDDERLFQTSRLILIGETIKIVIEDYVQHLSGYHFKLKFDPELLFNQQFQYQ
NRIASEFNTLYHWHPLLPDTFNIEDQEYTFKQFLYNNSILLEHGLAHFVESFTRQIAGRV
AGGRNVPIAVQAVAKASIDQSREMKYQSLNEYRKRFSLKPYTSFEELTGEKEMAAELKAL
YHDIDAMELYPALLVEKPRPDAIFGETMVELGAPFSLKGLMGNPICSPQYWKPSTFGGEV
GFRIINTASIQSLICNNVKGCPFASFNVQDPQPTKTATINASASHSRLDDINPTVLIKRR
STEL
|
|
|
|---|
| Component 2 |
| Name: | Prostaglandin G/H synthase 1 |
|---|
| Synonyms: | Cox-1 | Cox1 | Cyclooxygenase | Cyclooxygenase-1 | PGH1_RAT | Prostaglandin G/H synthase (cyclooxygenase) | Ptgs1 |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 69033.56 |
|---|
| Organism: | RAT |
|---|
| Description: | COX-1 0 RAT::Q63921 |
|---|
| Residue: | 602 |
|---|
| Sequence: | MSRRSLSLQFPLLLLLLLLPPPPVLLTDAGVPSPVNPCCYYPCQNQGVCVRFGLDHYQCD
CTRTGYSGPNCTIPEIWTWLRSSLRPSPSFTHFLLTHGYWIWEFVNATFIREVLMRLVIT
VRSNLIPSPPTYNTAHDYISWESFSNVSYYTRILPSVPKDCPTPMGTKGKKQLPDIHLLA
QRLLLRREFIPGPQGTNVLFAFFAQHFTHQFFKTSGKMGPGFTKALGHGVDLGHIYGDSL
ERQYHLRLFKDGKLKYQVLDGEVYPPSVEQASVLMRYPPGVPPEKQMAVGQEVFGLLPGL
MLFSTIWLREHNRVCDLLKEEHPTWDDEQLFQTTRLILIGETIKIIIEEYVQHLSGYFLQ
LKFDPELLFRAQFQYRNRIALEFNHLYHWHPLMPDSFQVGSQEYSYEQFLFNTSMLVDYG
VEALVDAFSRQRAGRIGGGRNFDYHVLHVAEDVIKESREMRLQSFNEYRKRFGLKPYTSF
QEFTGEKEMAAELEELYGDIDALEFYPGLMLEKCQPNSLFGESMIEMGAPFSLKGLLGNP
ICSPEYWKPSTFGGDVGFNIVNTASLKKLVCLNTKTCPYVSFRVPDYPGDDGSVFVRPST
EL
|
|
|
|---|
| BDBM50076640 |
|---|
| n/a |
|---|
| Name | BDBM50076640 |
|---|
| Synonyms: | CHEMBL173133 | N-Hydroxy-N-{3-[1-(4-methoxy-phenyl)-5-p-tolyl-1H-pyrazol-3-yl]-prop-2-ynyl}-propionamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H23N3O3 |
|---|
| Mol. Mass. | 389.447 |
|---|
| SMILES | CCC(=O)N(O)CC#Cc1cc(-c2ccc(C)cc2)n(n1)-c1ccc(OC)cc1 |
|---|
| Structure | 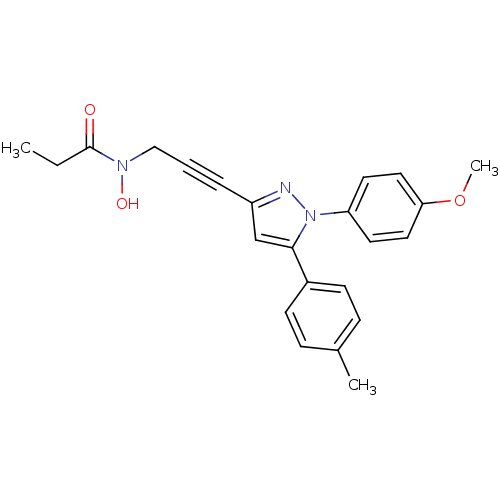
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Connolly, PJ; Wetter, SK; Beers, KN; Hamel, SC; Chen, RH; Wachter, MP; Ansell, J; Singer, MM; Steber, M; Ritchie, DM; Argentieri, DC N-hydroxyurea and hydroxamic acid inhibitors of cyclooxygenase and 5-lipoxygenase. Bioorg Med Chem Lett9:979-84 (1999) [PubMed]
Connolly, PJ; Wetter, SK; Beers, KN; Hamel, SC; Chen, RH; Wachter, MP; Ansell, J; Singer, MM; Steber, M; Ritchie, DM; Argentieri, DC N-hydroxyurea and hydroxamic acid inhibitors of cyclooxygenase and 5-lipoxygenase. Bioorg Med Chem Lett9:979-84 (1999) [PubMed]