| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Retinoic acid receptor RXR-alpha |
|---|
| Ligand | BDBM50032675 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1912868 (CHEMBL4415451) |
|---|
| EC50 | 22±n/a nM |
|---|
| Citation |  Yamada, S; Kawasaki, M; Fujihara, M; Watanabe, M; Takamura, Y; Takioku, M; Nishioka, H; Takeuchi, Y; Makishima, M; Motoyama, T; Ito, S; Tokiwa, H; Nakano, S; Kakuta, H Competitive Binding Assay with an Umbelliferone-Based Fluorescent Rexinoid for Retinoid X Receptor Ligand Screening. J Med Chem62:8809-8818 (2019) [PubMed] Article Yamada, S; Kawasaki, M; Fujihara, M; Watanabe, M; Takamura, Y; Takioku, M; Nishioka, H; Takeuchi, Y; Makishima, M; Motoyama, T; Ito, S; Tokiwa, H; Nakano, S; Kakuta, H Competitive Binding Assay with an Umbelliferone-Based Fluorescent Rexinoid for Retinoid X Receptor Ligand Screening. J Med Chem62:8809-8818 (2019) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Retinoic acid receptor RXR-alpha |
|---|
| Name: | Retinoic acid receptor RXR-alpha |
|---|
| Synonyms: | NR2B1 | Nuclear receptor subfamily 2 group B member 1 | Nuclear receptor subfamily 4 group A member 2 | Nuclear receptor subfamily 4 group A member 2/Retinoic acid receptor RXR-alpha | RXRA | RXRA_HUMAN | Retinoic acid receptor RXR-alpha/gamma | Retinoid X receptor alpha | Retinoid receptor |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 50820.38 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_1456363 |
|---|
| Residue: | 462 |
|---|
| Sequence: | MDTKHFLPLDFSTQVNSSLTSPTGRGSMAAPSLHPSLGPGIGSPGQLHSPISTLSSPING
MGPPFSVISSPMGPHSMSVPTTPTLGFSTGSPQLSSPMNPVSSSEDIKPPLGLNGVLKVP
AHPSGNMASFTKHICAICGDRSSGKHYGVYSCEGCKGFFKRTVRKDLTYTCRDNKDCLID
KRQRNRCQYCRYQKCLAMGMKREAVQEERQRGKDRNENEVESTSSANEDMPVERILEAEL
AVEPKTETYVEANMGLNPSSPNDPVTNICQAADKQLFTLVEWAKRIPHFSELPLDDQVIL
LRAGWNELLIASFSHRSIAVKDGILLATGLHVHRNSAHSAGVGAIFDRVLTELVSKMRDM
QMDKTELGCLRAIVLFNPDSKGLSNPAEVEALREKVYASLEAYCKHKYPEQPGRFAKLLL
RLPALRSIGLKCLEHLFFFKLIGDTPIDTFLMEMLEAPHQMT
|
|
|
|---|
| BDBM50032675 |
|---|
| n/a |
|---|
| Name | BDBM50032675 |
|---|
| Synonyms: | 4-[1-(3,5,5,8,8-pentamethyl-5,6,7,8-tetrahydronaphthalen-2-yl)vinyl]benzoic acid | BEXAROTENE | CHEMBL1023 | p-(1-(5,6,7,8-Tetrahydro-3,5,5,8,8-pentamethyl-2-naphthyl)vinyl)benzoic acid |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H28O2 |
|---|
| Mol. Mass. | 348.4779 |
|---|
| SMILES | Cc1cc2c(cc1C(=C)c1ccc(cc1)C(O)=O)C(C)(C)CCC2(C)C |
|---|
| Structure | 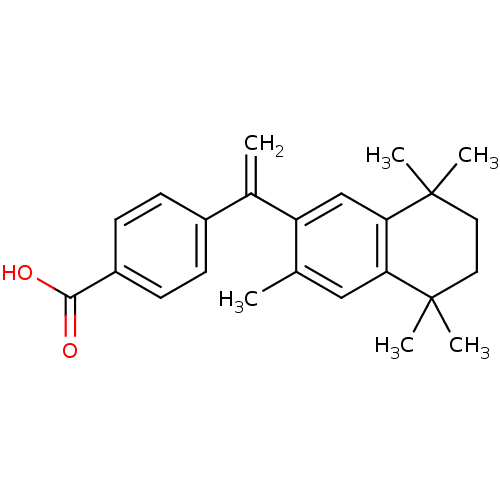
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Yamada, S; Kawasaki, M; Fujihara, M; Watanabe, M; Takamura, Y; Takioku, M; Nishioka, H; Takeuchi, Y; Makishima, M; Motoyama, T; Ito, S; Tokiwa, H; Nakano, S; Kakuta, H Competitive Binding Assay with an Umbelliferone-Based Fluorescent Rexinoid for Retinoid X Receptor Ligand Screening. J Med Chem62:8809-8818 (2019) [PubMed] Article
Yamada, S; Kawasaki, M; Fujihara, M; Watanabe, M; Takamura, Y; Takioku, M; Nishioka, H; Takeuchi, Y; Makishima, M; Motoyama, T; Ito, S; Tokiwa, H; Nakano, S; Kakuta, H Competitive Binding Assay with an Umbelliferone-Based Fluorescent Rexinoid for Retinoid X Receptor Ligand Screening. J Med Chem62:8809-8818 (2019) [PubMed] Article