| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prostaglandin G/H synthase 1 |
|---|
| Ligand | BDBM50094988 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_51516 |
|---|
| IC50 | >100000±n/a nM |
|---|
| Citation |  Nicoll-Griffith, DA; Yergey, JA; Trimble, LA; Silva, JM; Li, C; Chauret, N; Gauthier, JY; Grimm, E; Léger, S; Roy, P; Thérien, M; Wang, Z; Prasit, P; Zamboni, R; Young, RN; Brideau, C; Chan, CC; Mancini, J; Riendeau, D Synthesis, characterization, and activity of metabolites derived from the cyclooxygenase-2 inhibitor rofecoxib (MK-0966, Vioxx). Bioorg Med Chem Lett10:2683-6 (2000) [PubMed] Nicoll-Griffith, DA; Yergey, JA; Trimble, LA; Silva, JM; Li, C; Chauret, N; Gauthier, JY; Grimm, E; Léger, S; Roy, P; Thérien, M; Wang, Z; Prasit, P; Zamboni, R; Young, RN; Brideau, C; Chan, CC; Mancini, J; Riendeau, D Synthesis, characterization, and activity of metabolites derived from the cyclooxygenase-2 inhibitor rofecoxib (MK-0966, Vioxx). Bioorg Med Chem Lett10:2683-6 (2000) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prostaglandin G/H synthase 1 |
|---|
| Name: | Prostaglandin G/H synthase 1 |
|---|
| Synonyms: | COX-1 | COX1 | Cyclooxygenase | Cyclooxygenase-1 | Cyclooxygenase-1 (COX-1) | PGH1_HUMAN | PTGS1 | Prostaglandin G/H synthase (cyclooxygenase) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 68692.62 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P23219 |
|---|
| Residue: | 599 |
|---|
| Sequence: | MSRSLLLWFLLFLLLLPPLPVLLADPGAPTPVNPCCYYPCQHQGICVRFGLDRYQCDCTR
TGYSGPNCTIPGLWTWLRNSLRPSPSFTHFLLTHGRWFWEFVNATFIREMLMRLVLTVRS
NLIPSPPTYNSAHDYISWESFSNVSYYTRILPSVPKDCPTPMGTKGKKQLPDAQLLARRF
LLRRKFIPDPQGTNLMFAFFAQHFTHQFFKTSGKMGPGFTKALGHGVDLGHIYGDNLERQ
YQLRLFKDGKLKYQVLDGEMYPPSVEEAPVLMHYPRGIPPQSQMAVGQEVFGLLPGLMLY
ATLWLREHNRVCDLLKAEHPTWGDEQLFQTTRLILIGETIKIVIEEYVQQLSGYFLQLKF
DPELLFGVQFQYRNRIAMEFNHLYHWHPLMPDSFKVGSQEYSYEQFLFNTSMLVDYGVEA
LVDAFSRQIAGRIGGGRNMDHHILHVAVDVIRESREMRLQPFNEYRKRFGMKPYTSFQEL
VGEKEMAAELEELYGDIDALEFYPGLLLEKCHPNSIFGESMIEIGAPFSLKGLLGNPICS
PEYWKPSTFGGEVGFNIVKTATLKKLVCLNTKTCPYVSFRVPDASQDDGPAVERPSTEL
|
|
|
|---|
| BDBM50094988 |
|---|
| n/a |
|---|
| Name | BDBM50094988 |
|---|
| Synonyms: | (3R,4S)-4-(4-Methanesulfonyl-phenyl)-3-phenyl-dihydro-furan-2-one | CHEMBL89392 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C17H16O4S |
|---|
| Mol. Mass. | 316.372 |
|---|
| SMILES | CS(=O)(=O)c1ccc(cc1)[C@H]1COC(=O)[C@H]1c1ccccc1 |
|---|
| Structure | 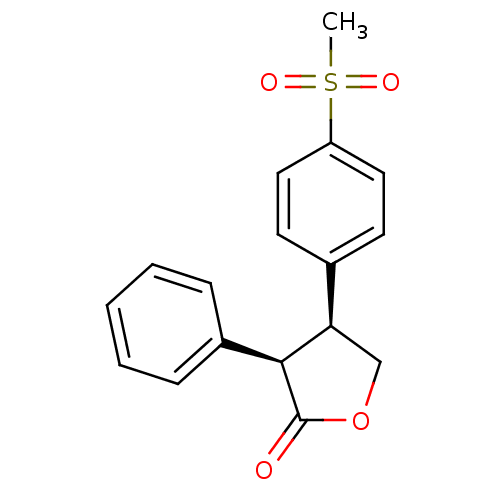
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Nicoll-Griffith, DA; Yergey, JA; Trimble, LA; Silva, JM; Li, C; Chauret, N; Gauthier, JY; Grimm, E; Léger, S; Roy, P; Thérien, M; Wang, Z; Prasit, P; Zamboni, R; Young, RN; Brideau, C; Chan, CC; Mancini, J; Riendeau, D Synthesis, characterization, and activity of metabolites derived from the cyclooxygenase-2 inhibitor rofecoxib (MK-0966, Vioxx). Bioorg Med Chem Lett10:2683-6 (2000) [PubMed]
Nicoll-Griffith, DA; Yergey, JA; Trimble, LA; Silva, JM; Li, C; Chauret, N; Gauthier, JY; Grimm, E; Léger, S; Roy, P; Thérien, M; Wang, Z; Prasit, P; Zamboni, R; Young, RN; Brideau, C; Chan, CC; Mancini, J; Riendeau, D Synthesis, characterization, and activity of metabolites derived from the cyclooxygenase-2 inhibitor rofecoxib (MK-0966, Vioxx). Bioorg Med Chem Lett10:2683-6 (2000) [PubMed]