| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Coagulation factor VII |
|---|
| Ligand | BDBM50106240 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_48457 (CHEMBL662862) |
|---|
| Ki | 1700±n/a nM |
|---|
| Citation |  Mackman, RL; Katz, BA; Breitenbucher, JG; Hui, HC; Verner, E; Luong, C; Liu, L; Sprengeler, PA Exploiting subsite S1 of trypsin-like serine proteases for selectivity: potent and selective inhibitors of urokinase-type plasminogen activator. J Med Chem44:3856-71 (2001) [PubMed] Mackman, RL; Katz, BA; Breitenbucher, JG; Hui, HC; Verner, E; Luong, C; Liu, L; Sprengeler, PA Exploiting subsite S1 of trypsin-like serine proteases for selectivity: potent and selective inhibitors of urokinase-type plasminogen activator. J Med Chem44:3856-71 (2001) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Coagulation factor VII |
|---|
| Name: | Coagulation factor VII |
|---|
| Synonyms: | Eptacog alfa | F7 | FA7_HUMAN | Factor VIIa | Factor VIIa (fVIIa) | Proconvertin | SPCA | Thrombin and coagulation factor VII | serum prothrombin conversion accelerator |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 51599.89 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 466 |
|---|
| Sequence: | MVSQALRLLCLLLGLQGCLAAGGVAKASGGETRDMPWKPGPHRVFVTQEEAHGVLHRRRR
ANAFLEELRPGSLERECKEEQCSFEEAREIFKDAERTKLFWISYSDGDQCASSPCQNGGS
CKDQLQSYICFCLPAFEGRNCETHKDDQLICVNENGGCEQYCSDHTGTKRSCRCHEGYSL
LADGVSCTPTVEYPCGKIPILEKRNASKPQGRIVGGKVCPKGECPWQVLLLVNGAQLCGG
TLINTIWVVSAAHCFDKIKNWRNLIAVLGEHDLSEHDGDEQSRRVAQVIIPSTYVPGTTN
HDIALLRLHQPVVLTDHVVPLCLPERTFSERTLAFVRFSLVSGWGQLLDRGATALELMVL
NVPRLMTQDCLQQSRKVGDSPNITEYMFCAGYSDGSKDSCKGDSGGPHATHYRGTWYLTG
IVSWGQGCATVGHFGVYTRVSQYIEWLQKLMRSEPRPGVLLRAPFP
|
|
|
|---|
| BDBM50106240 |
|---|
| n/a |
|---|
| Name | BDBM50106240 |
|---|
| Synonyms: | 6-Chloro-2-(2-hydroxy-biphenyl-3-yl)-1H-indole-5-carboxamidine | CHEMBL339902 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H16ClN3O |
|---|
| Mol. Mass. | 361.824 |
|---|
| SMILES | NC(=N)c1cc2cc([nH]c2cc1Cl)-c1cccc(-c2ccccc2)c1O |
|---|
| Structure | 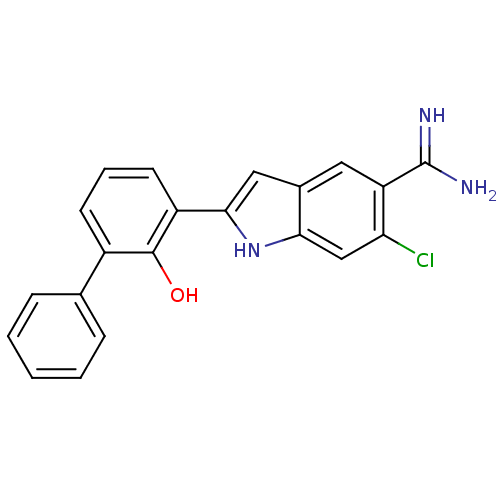
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Mackman, RL; Katz, BA; Breitenbucher, JG; Hui, HC; Verner, E; Luong, C; Liu, L; Sprengeler, PA Exploiting subsite S1 of trypsin-like serine proteases for selectivity: potent and selective inhibitors of urokinase-type plasminogen activator. J Med Chem44:3856-71 (2001) [PubMed]
Mackman, RL; Katz, BA; Breitenbucher, JG; Hui, HC; Verner, E; Luong, C; Liu, L; Sprengeler, PA Exploiting subsite S1 of trypsin-like serine proteases for selectivity: potent and selective inhibitors of urokinase-type plasminogen activator. J Med Chem44:3856-71 (2001) [PubMed]