| Reaction Details |
|---|
 | Report a problem with these data |
| Target | C-C chemokine receptor type 5 |
|---|
| Ligand | BDBM50096557 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_39503 |
|---|
| IC50 | 30±n/a nM |
|---|
| Citation |  Willoughby, CA; Berk, SC; Rosauer, KG; Degrado, S; Chapman, KT; Gould, SL; Springer, MS; Malkowitz, L; Schleif, WA; Hazuda, D; Miller, M; Kessler, J; Danzeisen, R; Holmes, K; Lineberger, J; Carella, A; Carver, G; Emini, EA Combinatorial synthesis of CCR5 antagonists. Bioorg Med Chem Lett11:3137-41 (2001) [PubMed] Willoughby, CA; Berk, SC; Rosauer, KG; Degrado, S; Chapman, KT; Gould, SL; Springer, MS; Malkowitz, L; Schleif, WA; Hazuda, D; Miller, M; Kessler, J; Danzeisen, R; Holmes, K; Lineberger, J; Carella, A; Carver, G; Emini, EA Combinatorial synthesis of CCR5 antagonists. Bioorg Med Chem Lett11:3137-41 (2001) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| C-C chemokine receptor type 5 |
|---|
| Name: | C-C chemokine receptor type 5 |
|---|
| Synonyms: | C-C CKR-5 | C-C chemokine receptor type 5 | CC-CKR-5 | CCR-5 | CCR5 | CCR5/mu opioid receptor complex | CCR5_HUMAN | CD_antigen=CD195 | CHEMR13 | CMKBR5 | HIV-1 fusion coreceptor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 40540.21 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P51681 |
|---|
| Residue: | 352 |
|---|
| Sequence: | MDYQVSSPIYDINYYTSEPCQKINVKQIAARLLPPLYSLVFIFGFVGNMLVILILINCKR
LKSMTDIYLLNLAISDLFFLLTVPFWAHYAAAQWDFGNTMCQLLTGLYFIGFFSGIFFII
LLTIDRYLAVVHAVFALKARTVTFGVVTSVITWVVAVFASLPGIIFTRSQKEGLHYTCSS
HFPYSQYQFWKNFQTLKIVILGLVLPLLVMVICYSGILKTLLRCRNEKKRHRAVRLIFTI
MIVYFLFWAPYNIVLLLNTFQEFFGLNNCSSSNRLDQAMQVTETLGMTHCCINPIIYAFV
GEKFRNYLLVFFQKHIAKRFCKCCSIFQQEAPERASSVYTRSTGEQEISVGL
|
|
|
|---|
| BDBM50096557 |
|---|
| n/a |
|---|
| Name | BDBM50096557 |
|---|
| Synonyms: | CHEMBL312701 | N-[(S)-2-(3-Chloro-phenyl)-4-(4-phenyl-piperidin-1-yl)-butyl]-N-methyl-benzenesulfonamide | N-[2-(3-Chloro-phenyl)-4-(4-phenyl-piperidin-1-yl)-butyl]-N-methyl-benzenesulfonamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C28H33ClN2O2S |
|---|
| Mol. Mass. | 497.092 |
|---|
| SMILES | CN(C[C@@H](CCN1CCC(CC1)c1ccccc1)c1cccc(Cl)c1)S(=O)(=O)c1ccccc1 |
|---|
| Structure | 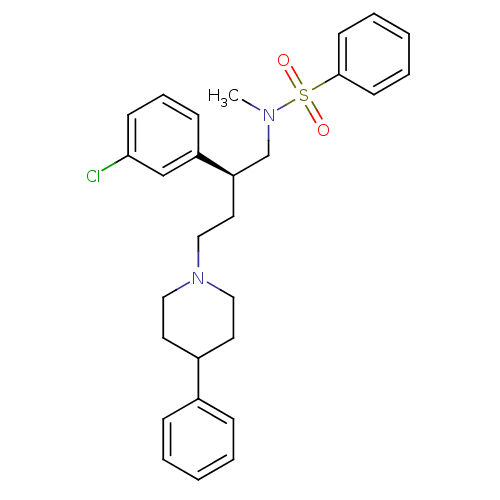
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Willoughby, CA; Berk, SC; Rosauer, KG; Degrado, S; Chapman, KT; Gould, SL; Springer, MS; Malkowitz, L; Schleif, WA; Hazuda, D; Miller, M; Kessler, J; Danzeisen, R; Holmes, K; Lineberger, J; Carella, A; Carver, G; Emini, EA Combinatorial synthesis of CCR5 antagonists. Bioorg Med Chem Lett11:3137-41 (2001) [PubMed]
Willoughby, CA; Berk, SC; Rosauer, KG; Degrado, S; Chapman, KT; Gould, SL; Springer, MS; Malkowitz, L; Schleif, WA; Hazuda, D; Miller, M; Kessler, J; Danzeisen, R; Holmes, K; Lineberger, J; Carella, A; Carver, G; Emini, EA Combinatorial synthesis of CCR5 antagonists. Bioorg Med Chem Lett11:3137-41 (2001) [PubMed]