| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Nitric oxide synthase, endothelial |
|---|
| Ligand | BDBM50116673 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_64984 |
|---|
| IC50 | 320±n/a nM |
|---|
| Citation |  Kawanaka, Y; Kobayashi, K; Kusuda, S; Tatsumi, T; Murota, M; Nishiyama, T; Hisaichi, K; Fujii, A; Hirai, K; Naka, M; Komeno, M; Nakai, H; Toda, M Design and synthesis of orally bioavailable inhibitors of inducible nitric oxide synthase. Part 1: synthesis and biological evaluation of dihydropyridin-2-imines. Bioorg Med Chem Lett12:2291-4 (2002) [PubMed] Kawanaka, Y; Kobayashi, K; Kusuda, S; Tatsumi, T; Murota, M; Nishiyama, T; Hisaichi, K; Fujii, A; Hirai, K; Naka, M; Komeno, M; Nakai, H; Toda, M Design and synthesis of orally bioavailable inhibitors of inducible nitric oxide synthase. Part 1: synthesis and biological evaluation of dihydropyridin-2-imines. Bioorg Med Chem Lett12:2291-4 (2002) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Nitric oxide synthase, endothelial |
|---|
| Name: | Nitric oxide synthase, endothelial |
|---|
| Synonyms: | Constitutive NOS | EC-NOS | Endothelial NOS | Endothelial nitric oxide synthase | NOS type III | NOS3 | NOS3_HUMAN | NOSIII | Nitric oxide synthase (inducible and endothelial) | Nitric oxide synthase, endothelial (eNOS) | Nitric-oxide synthase (endothelial and brain) | cNOS | eNOS |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 133297.84 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P29474 |
|---|
| Residue: | 1203 |
|---|
| Sequence: | MGNLKSVAQEPGPPCGLGLGLGLGLCGKQGPATPAPEPSRAPASLLPPAPEHSPPSSPLT
QPPEGPKFPRVKNWEVGSITYDTLSAQAQQDGPCTPRRCLGSLVFPRKLQGRPSPGPPAP
EQLLSQARDFINQYYSSIKRSGSQAHEQRLQEVEAEVAATGTYQLRESELVFGAKQAWRN
APRCVGRIQWGKLQVFDARDCRSAQEMFTYICNHIKYATNRGNLRSAITVFPQRCPGRGD
FRIWNSQLVRYAGYRQQDGSVRGDPANVEITELCIQHGWTPGNGRFDVLPLLLQAPDDPP
ELFLLPPELVLEVPLEHPTLEWFAALGLRWYALPAVSNMLLEIGGLEFPAAPFSGWYMST
EIGTRNLCDPHRYNILEDVAVCMDLDTRTTSSLWKDKAAVEINVAVLHSYQLAKVTIVDH
HAATASFMKHLENEQKARGGCPADWAWIVPPISGSLTPVFHQEMVNYFLSPAFRYQPDPW
KGSAAKGTGITRKKTFKEVANAVKISASLMGTVMAKRVKATILYGSETGRAQSYAQQLGR
LFRKAFDPRVLCMDEYDVVSLEHETLVLVVTSTFGNGDPPENGESFAAALMEMSGPYNSS
PRPEQHKSYKIRFNSISCSDPLVSSWRRKRKESSNTDSAGALGTLRFCVFGLGSRAYPHF
CAFARAVDTRLEELGGERLLQLGQGDELCGQEEAFRGWAQAAFQAACETFCVGEDAKAAA
RDIFSPKRSWKRQRYRLSAQAEGLQLLPGLIHVHRRKMFQATIRSVENLQSSKSTRATIL
VRLDTGGQEGLQYQPGDHIGVCPPNRPGLVEALLSRVEDPPAPTEPVAVEQLEKGSPGGP
PPGWVRDPRLPPCTLRQALTFFLDITSPPSPQLLRLLSTLAEEPREQQELEALSQDPRRY
EEWKWFRCPTLLEVLEQFPSVALPAPLLLTQLPLLQPRYYSVSSAPSTHPGEIHLTVAVL
AYRTQDGLGPLHYGVCSTWLSQLKPGDPVPCFIRGAPSFRLPPDPSLPCILVGPGTGIAP
FRGFWQERLHDIESKGLQPTPMTLVFGCRCSQLDHLYRDEVQNAQQRGVFGRVLTAFSRE
PDNPKTYVQDILRTELAAEVHRVLCLERGHMFVCGDVTMATNVLQTVQRILATEGDMELD
EAGDVIGVLRDQQRYHEDIFGLTLRTQEVTSRIRTQSFSLQERQLRGAVPWAFDPPGSDT
NSP
|
|
|
|---|
| BDBM50116673 |
|---|
| n/a |
|---|
| Name | BDBM50116673 |
|---|
| Synonyms: | 3,4-Dimethyl-5,6-dihydro-1H-pyridin-(2Z)-ylideneamine | CHEMBL75484 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C7H12N2 |
|---|
| Mol. Mass. | 124.1836 |
|---|
| SMILES | CC1=C(C)C(N)=NCC1 |c:1,5| |
|---|
| Structure | 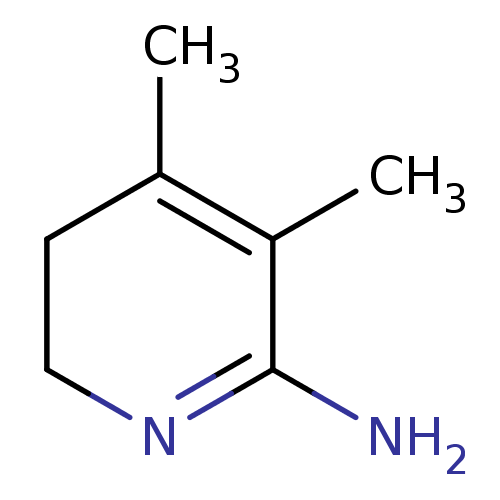
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Kawanaka, Y; Kobayashi, K; Kusuda, S; Tatsumi, T; Murota, M; Nishiyama, T; Hisaichi, K; Fujii, A; Hirai, K; Naka, M; Komeno, M; Nakai, H; Toda, M Design and synthesis of orally bioavailable inhibitors of inducible nitric oxide synthase. Part 1: synthesis and biological evaluation of dihydropyridin-2-imines. Bioorg Med Chem Lett12:2291-4 (2002) [PubMed]
Kawanaka, Y; Kobayashi, K; Kusuda, S; Tatsumi, T; Murota, M; Nishiyama, T; Hisaichi, K; Fujii, A; Hirai, K; Naka, M; Komeno, M; Nakai, H; Toda, M Design and synthesis of orally bioavailable inhibitors of inducible nitric oxide synthase. Part 1: synthesis and biological evaluation of dihydropyridin-2-imines. Bioorg Med Chem Lett12:2291-4 (2002) [PubMed]