

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | GTPase KRas | ||
| Ligand | BDBM50590537 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_2197024 (CHEMBL5109540) | ||
| IC50 | 77±n/a nM | ||
| Citation |  Imaizumi, T; Akaiwa, M; Abe, T; Nigawara, T; Koike, T; Satake, Y; Watanabe, K; Kaneko, O; Amano, Y; Mori, K; Yamanaka, Y; Nagashima, T; Shimazaki, M; Kuramoto, K Discovery and biological evaluation of 1-{2,7-diazaspiro[3.5]nonan-2-yl}prop-2-en-1-one derivatives as covalent inhibitors of KRAS G12C with favorable metabolic stability and anti-tumor activity. Bioorg Med Chem71:0 (2022) [PubMed] Article Imaizumi, T; Akaiwa, M; Abe, T; Nigawara, T; Koike, T; Satake, Y; Watanabe, K; Kaneko, O; Amano, Y; Mori, K; Yamanaka, Y; Nagashima, T; Shimazaki, M; Kuramoto, K Discovery and biological evaluation of 1-{2,7-diazaspiro[3.5]nonan-2-yl}prop-2-en-1-one derivatives as covalent inhibitors of KRAS G12C with favorable metabolic stability and anti-tumor activity. Bioorg Med Chem71:0 (2022) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| GTPase KRas | |||
| Name: | GTPase KRas | ||
| Synonyms: | GTPase KRas, N-terminally processed | K-Ras 2 | KRAS | KRAS2 | Ki-Ras | RASK2 | RASK_HUMAN | c-K-ras | c-Ki-ras | ||
| Type: | PROTEIN | ||
| Mol. Mass.: | 21656.10 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | ChEMBL_1476955 | ||
| Residue: | 189 | ||
| Sequence: |
| ||
| BDBM50590537 | |||
| n/a | |||
| Name | BDBM50590537 | ||
| Synonyms: | CHEMBL5205152 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C33H37ClFN7O2 | ||
| Mol. Mass. | 618.144 | ||
| SMILES | CCN1CCC(CC1)Oc1nc(N2CCC3(CN(C3)C(=O)C=C)CC2)c2cc(Cl)c(c(F)c2n1)-c1c(C)ccc2[nH]ncc12 |(8.57,-1.08,;8.57,-2.62,;7.23,-3.39,;5.9,-2.62,;4.56,-3.39,;4.56,-4.93,;5.9,-5.7,;7.23,-4.93,;3.23,-5.7,;1.9,-4.93,;1.89,-3.41,;.56,-2.64,;.56,-1.1,;-.77,-.33,;-.77,1.21,;.56,1.98,;-.55,3.09,;.54,4.17,;1.65,3.07,;.54,5.71,;-.79,6.48,;1.88,6.48,;1.88,8.02,;1.9,1.21,;1.9,-.33,;-.76,-3.41,;-2.09,-2.64,;-3.43,-3.4,;-4.76,-2.63,;-3.43,-4.94,;-2.09,-5.72,;-2.09,-7.26,;-.76,-4.94,;.58,-5.71,;-4.76,-5.71,;-4.76,-7.25,;-3.43,-8.02,;-6.08,-8.02,;-7.42,-7.26,;-7.42,-5.72,;-8.57,-4.68,;-7.94,-3.28,;-6.41,-3.44,;-6.09,-4.95,)| | ||
| Structure | 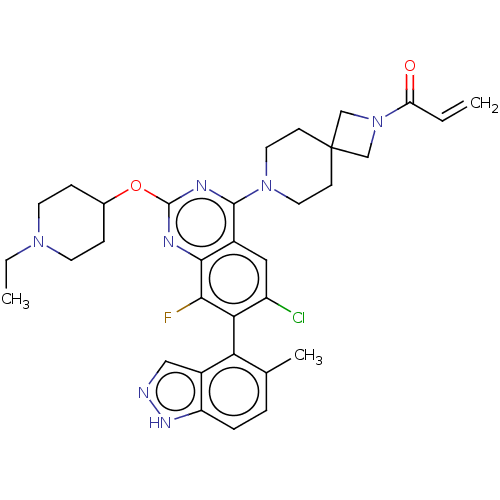
| ||