| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Reverse transcriptase/RNaseH |
|---|
| Ligand | BDBM50171983 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_306910 (CHEMBL829189) |
|---|
| pH | 7.8±n/a |
|---|
| IC50 | 23±n/a nM |
|---|
| Comments | extracted |
|---|
| Citation |  O'Meara, JA; Yoakim, C; Bonneau, PR; Bös, M; Cordingley, MG; Déziel, R; Doyon, L; Duan, J; Garneau, M; Guse, I; Landry, S; Malenfant, E; Naud, J; Ogilvie, WW; Thavonekham, B; Simoneau, B Novel 8-substituted dipyridodiazepinone inhibitors with a broad-spectrum of activity against HIV-1 strains resistant to non-nucleoside reverse transcriptase inhibitors. J Med Chem48:5580-8 (2005) [PubMed] Article O'Meara, JA; Yoakim, C; Bonneau, PR; Bös, M; Cordingley, MG; Déziel, R; Doyon, L; Duan, J; Garneau, M; Guse, I; Landry, S; Malenfant, E; Naud, J; Ogilvie, WW; Thavonekham, B; Simoneau, B Novel 8-substituted dipyridodiazepinone inhibitors with a broad-spectrum of activity against HIV-1 strains resistant to non-nucleoside reverse transcriptase inhibitors. J Med Chem48:5580-8 (2005) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Reverse transcriptase/RNaseH |
|---|
| Name: | Reverse transcriptase/RNaseH |
|---|
| Synonyms: | HIV-1 Reverse Transcriptase RNase H | Human immunodeficiency virus type 1 reverse transcriptase | Reverse transcriptase/RNaseH |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 65229.15 |
|---|
| Organism: | Human immunodeficiency virus 1 |
|---|
| Description: | ChEMBL_1473730 |
|---|
| Residue: | 566 |
|---|
| Sequence: | PISPIETVPVKLKPGMDGPKVKQWPLTEEKIKALVEICTEMEKEGKISKIGPENPYNTPV
FAIKKKDSTKWRKLVDFRELNKRTQDFWEVQLGIPHPAGLKKRKSVTVLDVGDAYFSVPL
DEDFRKYTAFTIPSINNETPGIRYQYNVLPQGWKGSPAIFQSSMTKILEPFRKQNPDIVI
YQYMDDLYVGSDLEIGQHRTKIEELRQHLLRWGLTTPDKKHQKEPPFLWMGYELHPDKWT
VQPIVLPEKDSWTVNDIQKLVGKLNWASQIYPGIRVRQLCKLLRGTKALTEVIPLTEEAE
LELAENREILKEPVHGVYYDPSKDLIAEIQKQGQGQWTYQIYQEPFKNLRTGKYARMRGA
HTNDVKQLTEAVQKITTESIVIWGKTPKFKLPIQKETWETWWTEYWQATWIPEWEFVNTP
PLVKLWYQLEKEPIVGAETFYVDGAANRETKLGKAGYVTNRGRQKVVTLTDTTNQKTELQ
AIYLALQDSGLEVNIVTDSQYALGIIQAQPDQSESELVNQIIEQLIKKEKVYLAWVPAHK
GIGGNEQVDKLVSAGIRKVLFLDGID
|
|
|
|---|
| BDBM50171983 |
|---|
| n/a |
|---|
| Name | BDBM50171983 |
|---|
| Synonyms: | CHEMBL370699 | {4-[2-(5-Ethyl-10-methyl-11-oxo-10,11-dihydro-5H-4,5,6,10-tetraaza-dibenzo[a,d]cyclohepten-2-yl)-ethoxy]-3-methyl-phenyl}-acetic acid |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H26N4O4 |
|---|
| Mol. Mass. | 446.4983 |
|---|
| SMILES | CCN1c2ncccc2N(C)C(=O)c2cc(CCOc3ccc(CC(O)=O)cc3C)cnc12 |
|---|
| Structure | 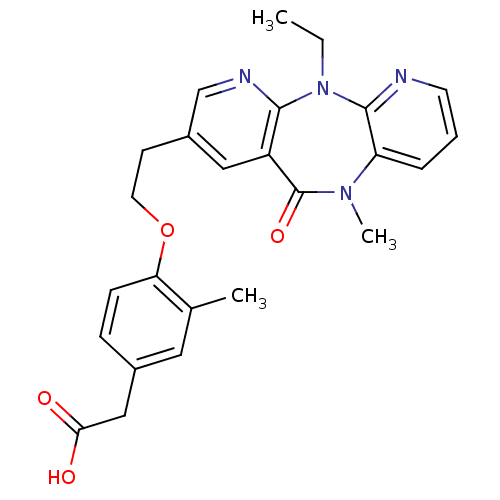
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 O'Meara, JA; Yoakim, C; Bonneau, PR; Bös, M; Cordingley, MG; Déziel, R; Doyon, L; Duan, J; Garneau, M; Guse, I; Landry, S; Malenfant, E; Naud, J; Ogilvie, WW; Thavonekham, B; Simoneau, B Novel 8-substituted dipyridodiazepinone inhibitors with a broad-spectrum of activity against HIV-1 strains resistant to non-nucleoside reverse transcriptase inhibitors. J Med Chem48:5580-8 (2005) [PubMed] Article
O'Meara, JA; Yoakim, C; Bonneau, PR; Bös, M; Cordingley, MG; Déziel, R; Doyon, L; Duan, J; Garneau, M; Guse, I; Landry, S; Malenfant, E; Naud, J; Ogilvie, WW; Thavonekham, B; Simoneau, B Novel 8-substituted dipyridodiazepinone inhibitors with a broad-spectrum of activity against HIV-1 strains resistant to non-nucleoside reverse transcriptase inhibitors. J Med Chem48:5580-8 (2005) [PubMed] Article