| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Urokinase-type plasminogen activator |
|---|
| Ligand | BDBM50175075 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_326971 (CHEMBL859772) |
|---|
| Ki | >14000±n/a nM |
|---|
| Citation |  Shi, Y; Zhang, J; Stein, PD; Shi, M; O'Connor, SP; Bisaha, SN; Li, C; Atwal, KS; Bisacchi, GS; Sitkoff, D; Pudzianowski, AT; Liu, EC; Hartl, KS; Seiler, SM; Youssef, S; Steinbacher, TE; Schumacher, WA; Rendina, AR; Bozarth, JM; Peterson, TL; Zhang, G; Zahler, R Ketene aminal-based lactam derivatives as a novel class of orally active FXa inhibitors. Bioorg Med Chem Lett15:5453-8 (2005) [PubMed] Article Shi, Y; Zhang, J; Stein, PD; Shi, M; O'Connor, SP; Bisaha, SN; Li, C; Atwal, KS; Bisacchi, GS; Sitkoff, D; Pudzianowski, AT; Liu, EC; Hartl, KS; Seiler, SM; Youssef, S; Steinbacher, TE; Schumacher, WA; Rendina, AR; Bozarth, JM; Peterson, TL; Zhang, G; Zahler, R Ketene aminal-based lactam derivatives as a novel class of orally active FXa inhibitors. Bioorg Med Chem Lett15:5453-8 (2005) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Urokinase-type plasminogen activator |
|---|
| Name: | Urokinase-type plasminogen activator |
|---|
| Synonyms: | 3.4.21.73 | PLAU | U-plasminogen activator | UROK_HUMAN | Urokinase | Urokinase-type plasminogen activator (uPA) | Urokinase-type plasminogen activator chain B | Urokinase-type plasminogen activator long chain A | Urokinase-type plasminogen activator short chain A | Urokinase-type plasminogen activator/surface receptor | uPA |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 48528.62 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P00749 |
|---|
| Residue: | 431 |
|---|
| Sequence: | MRALLARLLLCVLVVSDSKGSNELHQVPSNCDCLNGGTCVSNKYFSNIHWCNCPKKFGGQ
HCEIDKSKTCYEGNGHFYRGKASTDTMGRPCLPWNSATVLQQTYHAHRSDALQLGLGKHN
YCRNPDNRRRPWCYVQVGLKLLVQECMVHDCADGKKPSSPPEELKFQCGQKTLRPRFKII
GGEFTTIENQPWFAAIYRRHRGGSVTYVCGGSLISPCWVISATHCFIDYPKKEDYIVYLG
RSRLNSNTQGEMKFEVENLILHKDYSADTLAHHNDIALLKIRSKEGRCAQPSRTIQTICL
PSMYNDPQFGTSCEITGFGKENSTDYLYPEQLKMTVVKLISHRECQQPHYYGSEVTTKML
CAADPQWKTDSCQGDSGGPLVCSLQGRMTLTGIVSWGRGCALKDKPGVYTRVSHFLPWIR
SHTKEENGLAL
|
|
|
|---|
| BDBM50175075 |
|---|
| n/a |
|---|
| Name | BDBM50175075 |
|---|
| Synonyms: | (S,Z)-4-hydroxybutyl 2-cyano-3-(2-methylbenzofuran-5-ylamino)-3-(2-oxo-1-(2-oxo-2-(pyrrolidin-1-yl)ethyl)azepan-3-ylamino)acrylate | CHEMBL198629 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C29H37N5O6 |
|---|
| Mol. Mass. | 551.634 |
|---|
| SMILES | Cc1cc2cc(ccc2o1)N=C(N[C@H]1CCCCN(CC(=O)N2CCCC2)C1=O)C(C#N)C(=O)OCCCCO |w:10.11| |
|---|
| Structure | 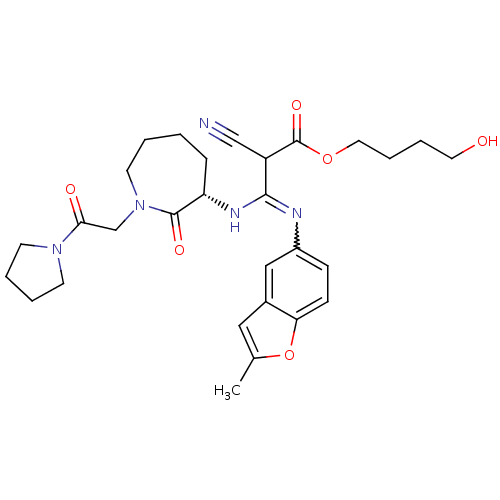
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Shi, Y; Zhang, J; Stein, PD; Shi, M; O'Connor, SP; Bisaha, SN; Li, C; Atwal, KS; Bisacchi, GS; Sitkoff, D; Pudzianowski, AT; Liu, EC; Hartl, KS; Seiler, SM; Youssef, S; Steinbacher, TE; Schumacher, WA; Rendina, AR; Bozarth, JM; Peterson, TL; Zhang, G; Zahler, R Ketene aminal-based lactam derivatives as a novel class of orally active FXa inhibitors. Bioorg Med Chem Lett15:5453-8 (2005) [PubMed] Article
Shi, Y; Zhang, J; Stein, PD; Shi, M; O'Connor, SP; Bisaha, SN; Li, C; Atwal, KS; Bisacchi, GS; Sitkoff, D; Pudzianowski, AT; Liu, EC; Hartl, KS; Seiler, SM; Youssef, S; Steinbacher, TE; Schumacher, WA; Rendina, AR; Bozarth, JM; Peterson, TL; Zhang, G; Zahler, R Ketene aminal-based lactam derivatives as a novel class of orally active FXa inhibitors. Bioorg Med Chem Lett15:5453-8 (2005) [PubMed] Article