| Reaction Details |
|---|
 | Report a problem with these data |
| Target | MAP kinase-activated protein kinase 2 |
|---|
| Ligand | BDBM50189603 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_368600 (CHEMBL866184) |
|---|
| IC50 | >50000±n/a nM |
|---|
| Citation |  Borzilleri, RM; Bhide, RS; Barrish, JC; D'Arienzo, CJ; Derbin, GM; Fargnoli, J; Hunt, JT; Jeyaseelan, R; Kamath, A; Kukral, DW; Marathe, P; Mortillo, S; Qian, L; Tokarski, JS; Wautlet, BS; Zheng, X; Lombardo, LJ Discovery and evaluation of N-cyclopropyl- 2,4-difluoro-5-((2-(pyridin-2-ylamino)thiazol-5- ylmethyl)amino)benzamide (BMS-605541), a selective and orally efficacious inhibitor of vascular endothelial growth factor receptor-2. J Med Chem49:3766-9 (2006) [PubMed] Article Borzilleri, RM; Bhide, RS; Barrish, JC; D'Arienzo, CJ; Derbin, GM; Fargnoli, J; Hunt, JT; Jeyaseelan, R; Kamath, A; Kukral, DW; Marathe, P; Mortillo, S; Qian, L; Tokarski, JS; Wautlet, BS; Zheng, X; Lombardo, LJ Discovery and evaluation of N-cyclopropyl- 2,4-difluoro-5-((2-(pyridin-2-ylamino)thiazol-5- ylmethyl)amino)benzamide (BMS-605541), a selective and orally efficacious inhibitor of vascular endothelial growth factor receptor-2. J Med Chem49:3766-9 (2006) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| MAP kinase-activated protein kinase 2 |
|---|
| Name: | MAP kinase-activated protein kinase 2 |
|---|
| Synonyms: | MAP kinase-activated protein kinase 2 (MAPKAPK2) | MAP kinase-activated protein kinase 2 (MK2) | MAP kinase-activated protein kinase 2 (p38/MK2) | MAPK-Activated Protein Kinase 2 (MK2) | MAPK-activated protein kinase 2 | MAPK2_HUMAN | MAPKAP kinase 2 | MAPKAPK-2 | MAPKAPK2 | MK2 |
|---|
| Type: | Serine/threonine-protein kinase |
|---|
| Mol. Mass.: | 45579.87 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P49137 |
|---|
| Residue: | 400 |
|---|
| Sequence: | MLSNSQGQSPPVPFPAPAPPPQPPTPALPHPPAQPPPPPPQQFPQFHVKSGLQIKKNAII
DDYKVTSQVLGLGINGKVLQIFNKRTQEKFALKMLQDCPKARREVELHWRASQCPHIVRI
VDVYENLYAGRKCLLIVMECLDGGELFSRIQDRGDQAFTEREASEIMKSIGEAIQYLHSI
NIAHRDVKPENLLYTSKRPNAILKLTDFGFAKETTSHNSLTTPCYTPYYVAPEVLGPEKY
DKSCDMWSLGVIMYILLCGYPPFYSNHGLAISPGMKTRIRMGQYEFPNPEWSEVSEEVKM
LIRNLLKTEPTQRMTITEFMNHPWIMQSTKVPQTPLHTSRVLKEDKERWEDVKEEMTSAL
ATMRVDYEQIKIKKIEDASNPLLLKRRKKARALEAAALAH
|
|
|
|---|
| BDBM50189603 |
|---|
| n/a |
|---|
| Name | BDBM50189603 |
|---|
| Synonyms: | CHEMBL377734 | N-cyclopropyl-2,4-difluoro-5-((2-(pyridin-2-ylamino)thiazol-5-yl)methylamino)benzamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H17F2N5OS |
|---|
| Mol. Mass. | 401.433 |
|---|
| SMILES | Fc1cc(F)c(cc1NCc1cnc(Nc2ccccn2)s1)C(=O)NC1CC1 |
|---|
| Structure | 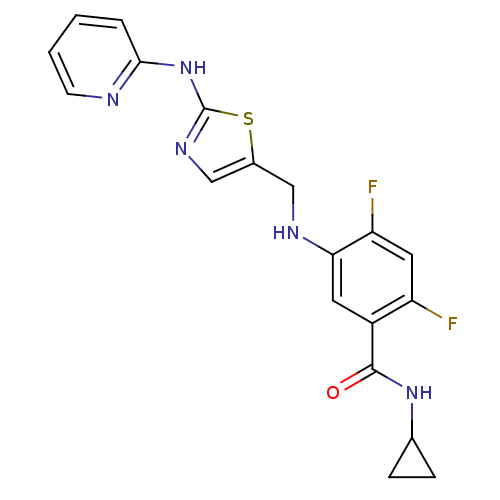
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Borzilleri, RM; Bhide, RS; Barrish, JC; D'Arienzo, CJ; Derbin, GM; Fargnoli, J; Hunt, JT; Jeyaseelan, R; Kamath, A; Kukral, DW; Marathe, P; Mortillo, S; Qian, L; Tokarski, JS; Wautlet, BS; Zheng, X; Lombardo, LJ Discovery and evaluation of N-cyclopropyl- 2,4-difluoro-5-((2-(pyridin-2-ylamino)thiazol-5- ylmethyl)amino)benzamide (BMS-605541), a selective and orally efficacious inhibitor of vascular endothelial growth factor receptor-2. J Med Chem49:3766-9 (2006) [PubMed] Article
Borzilleri, RM; Bhide, RS; Barrish, JC; D'Arienzo, CJ; Derbin, GM; Fargnoli, J; Hunt, JT; Jeyaseelan, R; Kamath, A; Kukral, DW; Marathe, P; Mortillo, S; Qian, L; Tokarski, JS; Wautlet, BS; Zheng, X; Lombardo, LJ Discovery and evaluation of N-cyclopropyl- 2,4-difluoro-5-((2-(pyridin-2-ylamino)thiazol-5- ylmethyl)amino)benzamide (BMS-605541), a selective and orally efficacious inhibitor of vascular endothelial growth factor receptor-2. J Med Chem49:3766-9 (2006) [PubMed] Article