| Reaction Details |
|---|
 | Report a problem with these data |
| Target | D(4) dopamine receptor |
|---|
| Ligand | BDBM55121 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_422463 (CHEMBL911046) |
|---|
| EC50 | 2.2±n/a nM |
|---|
| Citation |  Patel, MV; Kolasa, T; Mortell, K; Matulenko, MA; Hakeem, AA; Rohde, JJ; Nelson, SL; Cowart, MD; Nakane, M; Miller, LN; Uchic, ME; Terranova, MA; El-Kouhen, OF; Donnelly-Roberts, DL; Namovic, MT; Hollingsworth, PR; Chang, R; Martino, BR; Wetter, JM; Marsh, KC; Martin, R; Darbyshire, JF; Gintant, G; Hsieh, GC; Moreland, RB; Sullivan, JP; Brioni, JD; Stewart, AO Discovery of 3-methyl-N-(1-oxy-3',4',5',6'-tetrahydro-2'H-[2,4'-bipyridine]-1'-ylmethyl)benzamide (ABT-670), an orally bioavailable dopamine D4 agonist for the treatment of erectile dysfunction. J Med Chem49:7450-65 (2006) [PubMed] Article Patel, MV; Kolasa, T; Mortell, K; Matulenko, MA; Hakeem, AA; Rohde, JJ; Nelson, SL; Cowart, MD; Nakane, M; Miller, LN; Uchic, ME; Terranova, MA; El-Kouhen, OF; Donnelly-Roberts, DL; Namovic, MT; Hollingsworth, PR; Chang, R; Martino, BR; Wetter, JM; Marsh, KC; Martin, R; Darbyshire, JF; Gintant, G; Hsieh, GC; Moreland, RB; Sullivan, JP; Brioni, JD; Stewart, AO Discovery of 3-methyl-N-(1-oxy-3',4',5',6'-tetrahydro-2'H-[2,4'-bipyridine]-1'-ylmethyl)benzamide (ABT-670), an orally bioavailable dopamine D4 agonist for the treatment of erectile dysfunction. J Med Chem49:7450-65 (2006) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| D(4) dopamine receptor |
|---|
| Name: | D(4) dopamine receptor |
|---|
| Synonyms: | D(2C) dopamine receptor | DOPAMINE D4 | DOPAMINE D4.2 | DOPAMINE D4.4 | DRD4 | DRD4_HUMAN | Dopamine D4 receptor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 48373.19 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P21917 |
|---|
| Residue: | 419 |
|---|
| Sequence: | MGNRSTADADGLLAGRGPAAGASAGASAGLAGQGAAALVGGVLLIGAVLAGNSLVCVSVA
TERALQTPTNSFIVSLAAADLLLALLVLPLFVYSEVQGGAWLLSPRLCDALMAMDVMLCT
ASIFNLCAISVDRFVAVAVPLRYNRQGGSRRQLLLIGATWLLSAAVAAPVLCGLNDVRGR
DPAVCRLEDRDYVVYSSVCSFFLPCPLMLLLYWATFRGLQRWEVARRAKLHGRAPRRPSG
PGPPSPTPPAPRLPQDPCGPDCAPPAPGLPRGPCGPDCAPAAPSLPQDPCGPDCAPPAPG
LPPDPCGSNCAPPDAVRAAALPPQTPPQTRRRRRAKITGRERKAMRVLPVVVGAFLLCWT
PFFVVHITQALCPACSVPPRLVSAVTWLGYVNSALNPVIYTVFNAEFRNVFRKALRACC
|
|
|
|---|
| BDBM55121 |
|---|
| n/a |
|---|
| Name | BDBM55121 |
|---|
| Synonyms: | 3-HYDROXYTYRAMINE HYDROCHLORIDE | 4-(2-aminoethyl)benzene-1,2-diol;hydrochloride | 4-(2-aminoethyl)pyrocatechol;hydrochloride | 4-(2-azanylethyl)benzene-1,2-diol;hydrochloride | Dopamine | MLS000069419 | SMR000059081 | cid_65340 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C8H11NO2 |
|---|
| Mol. Mass. | 153.1784 |
|---|
| SMILES | NCCc1ccc(O)c(O)c1 |
|---|
| Structure | 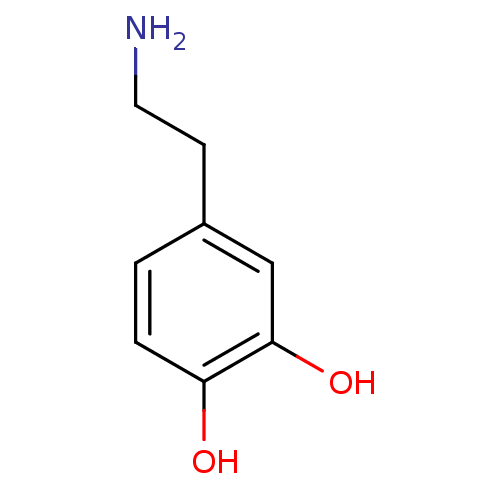
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Patel, MV; Kolasa, T; Mortell, K; Matulenko, MA; Hakeem, AA; Rohde, JJ; Nelson, SL; Cowart, MD; Nakane, M; Miller, LN; Uchic, ME; Terranova, MA; El-Kouhen, OF; Donnelly-Roberts, DL; Namovic, MT; Hollingsworth, PR; Chang, R; Martino, BR; Wetter, JM; Marsh, KC; Martin, R; Darbyshire, JF; Gintant, G; Hsieh, GC; Moreland, RB; Sullivan, JP; Brioni, JD; Stewart, AO Discovery of 3-methyl-N-(1-oxy-3',4',5',6'-tetrahydro-2'H-[2,4'-bipyridine]-1'-ylmethyl)benzamide (ABT-670), an orally bioavailable dopamine D4 agonist for the treatment of erectile dysfunction. J Med Chem49:7450-65 (2006) [PubMed] Article
Patel, MV; Kolasa, T; Mortell, K; Matulenko, MA; Hakeem, AA; Rohde, JJ; Nelson, SL; Cowart, MD; Nakane, M; Miller, LN; Uchic, ME; Terranova, MA; El-Kouhen, OF; Donnelly-Roberts, DL; Namovic, MT; Hollingsworth, PR; Chang, R; Martino, BR; Wetter, JM; Marsh, KC; Martin, R; Darbyshire, JF; Gintant, G; Hsieh, GC; Moreland, RB; Sullivan, JP; Brioni, JD; Stewart, AO Discovery of 3-methyl-N-(1-oxy-3',4',5',6'-tetrahydro-2'H-[2,4'-bipyridine]-1'-ylmethyl)benzamide (ABT-670), an orally bioavailable dopamine D4 agonist for the treatment of erectile dysfunction. J Med Chem49:7450-65 (2006) [PubMed] Article