| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50213744 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_447520 (CHEMBL895419) |
|---|
| IC50 | >25000±n/a nM |
|---|
| Citation |  Wang, Y; Busch-Petersen, J; Wang, F; Ma, L; Fu, W; Kerns, JK; Jin, J; Palovich, MR; Shen, JK; Burman, M; Foley, JJ; Schmidt, DB; Hunsberger, GE; Sarau, HM; Widdowson, KL 3-Arylamino-2H-1,2,4-benzothiadiazin-5-ol 1,1-dioxides as novel and selective CXCR2 antagonists. Bioorg Med Chem Lett17:3864-7 (2007) [PubMed] Article Wang, Y; Busch-Petersen, J; Wang, F; Ma, L; Fu, W; Kerns, JK; Jin, J; Palovich, MR; Shen, JK; Burman, M; Foley, JJ; Schmidt, DB; Hunsberger, GE; Sarau, HM; Widdowson, KL 3-Arylamino-2H-1,2,4-benzothiadiazin-5-ol 1,1-dioxides as novel and selective CXCR2 antagonists. Bioorg Med Chem Lett17:3864-7 (2007) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50213744 |
|---|
| n/a |
|---|
| Name | BDBM50213744 |
|---|
| Synonyms: | 3-(2-chloro-phenylamino)-7-nitro-1,1-dioxo-1,4-dihydro-1lambda6-benzo[1,2,4]thiadiazin-5-ol | CHEMBL231924 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C13H9ClN4O5S |
|---|
| Mol. Mass. | 368.752 |
|---|
| SMILES | Oc1cc(cc2c1N=C(Nc1ccccc1Cl)NS2(=O)=O)[N+]([O-])=O |t:8| |
|---|
| Structure | 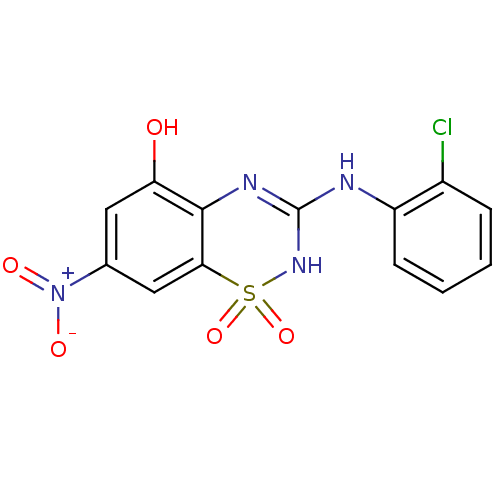
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Wang, Y; Busch-Petersen, J; Wang, F; Ma, L; Fu, W; Kerns, JK; Jin, J; Palovich, MR; Shen, JK; Burman, M; Foley, JJ; Schmidt, DB; Hunsberger, GE; Sarau, HM; Widdowson, KL 3-Arylamino-2H-1,2,4-benzothiadiazin-5-ol 1,1-dioxides as novel and selective CXCR2 antagonists. Bioorg Med Chem Lett17:3864-7 (2007) [PubMed] Article
Wang, Y; Busch-Petersen, J; Wang, F; Ma, L; Fu, W; Kerns, JK; Jin, J; Palovich, MR; Shen, JK; Burman, M; Foley, JJ; Schmidt, DB; Hunsberger, GE; Sarau, HM; Widdowson, KL 3-Arylamino-2H-1,2,4-benzothiadiazin-5-ol 1,1-dioxides as novel and selective CXCR2 antagonists. Bioorg Med Chem Lett17:3864-7 (2007) [PubMed] Article