| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Alpha-1A adrenergic receptor |
|---|
| Ligand | BDBM50219049 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_445302 (CHEMBL894443) |
|---|
| Ki | 150±n/a nM |
|---|
| Citation |  Chen, CA; Jiang, Y; Lu, K; Daniewska, I; Mazza, CG; Negron, L; Forray, C; Parola, T; Li, B; Hegde, LG; Wolinsky, TD; Craig, DA; Kong, R; Wetzel, JM; Andersen, K; Marzabadi, MR Synthesis and SAR investigations for novel melanin-concentrating hormone 1 receptor (MCH1) antagonists part 2: A hybrid strategy combining key fragments of HTS hits. J Med Chem50:3883-90 (2007) [PubMed] Article Chen, CA; Jiang, Y; Lu, K; Daniewska, I; Mazza, CG; Negron, L; Forray, C; Parola, T; Li, B; Hegde, LG; Wolinsky, TD; Craig, DA; Kong, R; Wetzel, JM; Andersen, K; Marzabadi, MR Synthesis and SAR investigations for novel melanin-concentrating hormone 1 receptor (MCH1) antagonists part 2: A hybrid strategy combining key fragments of HTS hits. J Med Chem50:3883-90 (2007) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Alpha-1A adrenergic receptor |
|---|
| Name: | Alpha-1A adrenergic receptor |
|---|
| Synonyms: | ADA1A_HUMAN | ADRA1A | ADRA1C | Adrenergic alpha1A | Alpha 1A-adrenoceptor | Alpha 1A-adrenoreceptor | Alpha adrenergic receptor 1a | Alpha-1C adrenergic receptor | Alpha-adrenergic receptor 1c | Cerebral cortex alpha adrenergic receptor | adrenergic Alpha1 | adrenergic Alpha1C |
|---|
| Type: | Cell-surface receptors |
|---|
| Mol. Mass.: | 51511.67 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P35348 |
|---|
| Residue: | 466 |
|---|
| Sequence: | MVFLSGNASDSSNCTQPPAPVNISKAILLGVILGGLILFGVLGNILVILSVACHRHLHSV
THYYIVNLAVADLLLTSTVLPFSAIFEVLGYWAFGRVFCNIWAAVDVLCCTASIMGLCII
SIDRYIGVSYPLRYPTIVTQRRGLMALLCVWALSLVISIGPLFGWRQPAPEDETICQINE
EPGYVLFSALGSFYLPLAIILVMYCRVYVVAKRESRGLKSGLKTDKSDSEQVTLRIHRKN
APAGGSGMASAKTKTHFSVRLLKFSREKKAAKTLGIVVGCFVLCWLPFFLVMPIGSFFPD
FKPSETVFKIVFWLGYLNSCINPIIYPCSSQEFKKAFQNVLRIQCLCRKQSSKHALGYTL
HPPSQAVEGQHKDMVRIPVGSRETFYRISKTDGVCEWKFFSSMPRGSARITVSKDQSSCT
TARVRSKSFLQVCCCVGPSTPSLDKNHQVPTIKVHTISLSENGEEV
|
|
|
|---|
| BDBM50219049 |
|---|
| n/a |
|---|
| Name | BDBM50219049 |
|---|
| Synonyms: | 1-(4-Chloro-phenyl)-4-[4-(4-chloro-phenyl)-4-hydroxy-piperidin-1-yl]-butan-1-one | 1-(4-chlorophenyl)-4-(4-(4-chlorophenyl)-4-hydroxypiperidin-1-yl)butan-1-one | CHEMBL363548 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H23Cl2NO2 |
|---|
| Mol. Mass. | 392.319 |
|---|
| SMILES | OC1(CCN(CCCC(=O)c2ccc(Cl)cc2)CC1)c1ccc(Cl)cc1 |
|---|
| Structure | 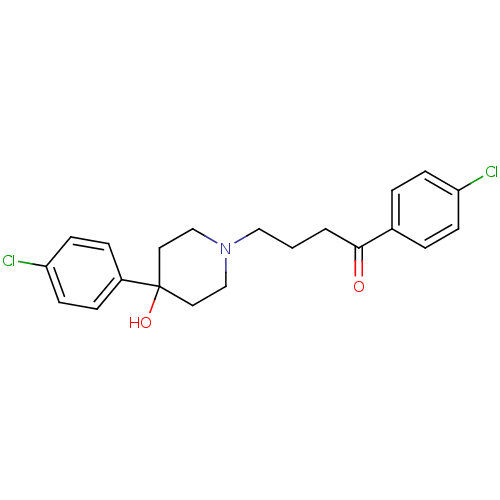
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Chen, CA; Jiang, Y; Lu, K; Daniewska, I; Mazza, CG; Negron, L; Forray, C; Parola, T; Li, B; Hegde, LG; Wolinsky, TD; Craig, DA; Kong, R; Wetzel, JM; Andersen, K; Marzabadi, MR Synthesis and SAR investigations for novel melanin-concentrating hormone 1 receptor (MCH1) antagonists part 2: A hybrid strategy combining key fragments of HTS hits. J Med Chem50:3883-90 (2007) [PubMed] Article
Chen, CA; Jiang, Y; Lu, K; Daniewska, I; Mazza, CG; Negron, L; Forray, C; Parola, T; Li, B; Hegde, LG; Wolinsky, TD; Craig, DA; Kong, R; Wetzel, JM; Andersen, K; Marzabadi, MR Synthesis and SAR investigations for novel melanin-concentrating hormone 1 receptor (MCH1) antagonists part 2: A hybrid strategy combining key fragments of HTS hits. J Med Chem50:3883-90 (2007) [PubMed] Article