| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Sphingosine 1-phosphate receptor 3 |
|---|
| Ligand | BDBM50249259 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_499714 (CHEMBL980649) |
|---|
| IC50 | 50±n/a nM |
|---|
| Citation |  Evindar, G; Satz, AL; Bernier, SG; Kavarana, MJ; Doyle, E; Lorusso, J; Taghizadeh, N; Halley, K; Hutchings, A; Kelley, MS; Wright, AD; Saha, AK; Hannig, G; Morgan, BA; Westlin, WF Synthesis and evaluation of arylalkoxy- and biarylalkoxy-phenylamide and phenylimidazoles as potent and selective sphingosine-1-phosphate receptor subtype-1 agonists. Bioorg Med Chem Lett19:2315-9 (2009) [PubMed] Article Evindar, G; Satz, AL; Bernier, SG; Kavarana, MJ; Doyle, E; Lorusso, J; Taghizadeh, N; Halley, K; Hutchings, A; Kelley, MS; Wright, AD; Saha, AK; Hannig, G; Morgan, BA; Westlin, WF Synthesis and evaluation of arylalkoxy- and biarylalkoxy-phenylamide and phenylimidazoles as potent and selective sphingosine-1-phosphate receptor subtype-1 agonists. Bioorg Med Chem Lett19:2315-9 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Sphingosine 1-phosphate receptor 3 |
|---|
| Name: | Sphingosine 1-phosphate receptor 3 |
|---|
| Synonyms: | C9orf108 | C9orf47 | EDG3 | Endothelial differentiation G-protein coupled receptor 3 | S1P receptor 3 | S1P receptor Edg-3 | S1P3 | S1PR3 | S1PR3_HUMAN | Sphingosine 1-phosphate receptor | Sphingosine 1-phosphate receptor 3 (S1P3) | Sphingosine 1-phosphate receptor Edg-3 |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 42278.13 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q99500 |
|---|
| Residue: | 378 |
|---|
| Sequence: | MATALPPRLQPVRGNETLREHYQYVGKLAGRLKEASEGSTLTTVLFLVICSFIVLENLMV
LIAIWKNNKFHNRMYFFIGNLALCDLLAGIAYKVNILMSGKKTFSLSPTVWFLREGSMFV
ALGASTCSLLAIAIERHLTMIKMRPYDANKRHRVFLLIGMCWLIAFTLGALPILGWNCLH
NLPDCSTILPLYSKKYIAFCISIFTAILVTIVILYARIYFLVKSSSRKVANHNNSERSMA
LLRTVVIVVSVFIACWSPLFILFLIDVACRVQACPILFKAQWFIVLAVLNSAMNPVIYTL
ASKEMRRAFFRLVCNCLVRGRGARASPIQPALDPSRSKSSSSNNSSHSPKVKEDLPHTAP
SSCIMDKNAALQNGIFCN
|
|
|
|---|
| BDBM50249259 |
|---|
| n/a |
|---|
| Name | BDBM50249259 |
|---|
| Synonyms: | (S)-2-amino-2-methyl-3-(4-(2-(2'-methylbiphenyl-4-yl)ethoxy)phenylamino)-3-oxopropyl dihydrogen phosphate | CHEMBL516380 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H29N2O6P |
|---|
| Mol. Mass. | 484.4813 |
|---|
| SMILES | Cc1ccccc1-c1ccc(CCOc2ccc(NC(=O)[C@@](C)(N)COP(O)(O)=O)cc2)cc1 |r| |
|---|
| Structure | 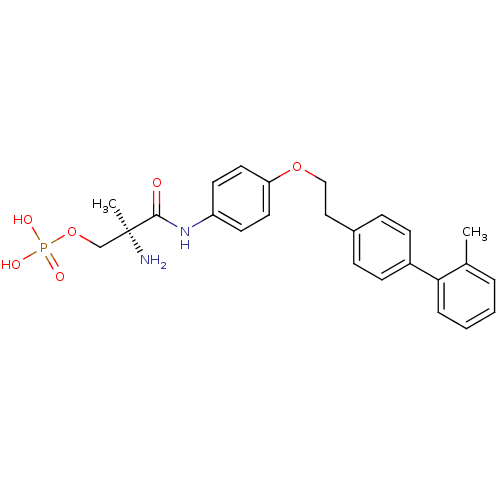
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Evindar, G; Satz, AL; Bernier, SG; Kavarana, MJ; Doyle, E; Lorusso, J; Taghizadeh, N; Halley, K; Hutchings, A; Kelley, MS; Wright, AD; Saha, AK; Hannig, G; Morgan, BA; Westlin, WF Synthesis and evaluation of arylalkoxy- and biarylalkoxy-phenylamide and phenylimidazoles as potent and selective sphingosine-1-phosphate receptor subtype-1 agonists. Bioorg Med Chem Lett19:2315-9 (2009) [PubMed] Article
Evindar, G; Satz, AL; Bernier, SG; Kavarana, MJ; Doyle, E; Lorusso, J; Taghizadeh, N; Halley, K; Hutchings, A; Kelley, MS; Wright, AD; Saha, AK; Hannig, G; Morgan, BA; Westlin, WF Synthesis and evaluation of arylalkoxy- and biarylalkoxy-phenylamide and phenylimidazoles as potent and selective sphingosine-1-phosphate receptor subtype-1 agonists. Bioorg Med Chem Lett19:2315-9 (2009) [PubMed] Article