| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Acetylcholinesterase |
|---|
| Ligand | BDBM10972 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_29079 (CHEMBL636453) |
|---|
| IC50 | 9±n/a nM |
|---|
| Citation |  Martin, LL; Davis, L; Klein, JT; Nemoto, P; Olsen, GE; Bores, GM; Camacho, F; Petko, WW; Rush, DK; Selk, D; Smith, CP; Vargas, HM; Winslow, JT; Effland, RC; Fink, DM Synthesis and preliminary structure-activity relationships of 1-[(3-fluoro-4-pyridinyl)amino]-3-methyl-1H-indol-5-yl methyl carbamate (P10358), a novel acetylcholinesterase inhibitor Bioorg Med Chem Lett7:157-162 (1997) Article Martin, LL; Davis, L; Klein, JT; Nemoto, P; Olsen, GE; Bores, GM; Camacho, F; Petko, WW; Rush, DK; Selk, D; Smith, CP; Vargas, HM; Winslow, JT; Effland, RC; Fink, DM Synthesis and preliminary structure-activity relationships of 1-[(3-fluoro-4-pyridinyl)amino]-3-methyl-1H-indol-5-yl methyl carbamate (P10358), a novel acetylcholinesterase inhibitor Bioorg Med Chem Lett7:157-162 (1997) Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Acetylcholinesterase |
|---|
| Name: | Acetylcholinesterase |
|---|
| Synonyms: | ACES_RAT | Acetylcholinesterase (AChE) | Acetylcholinesterase and butyrylcholinesterase (AChE and BChE) | Acetylcholinesterase precursor | Acetylcholinesterase, AChE | Ache |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 68193.62 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | P37136 |
|---|
| Residue: | 614 |
|---|
| Sequence: | MRPPWYPLHTPSLASPLLFLLLSLLGGGARAEGREDPQLLVRVRGGQLRGIRLKAPGGPV
SAFLGIPFAEPPVGSRRFMPPEPKRPWSGILDATTFQNVCYQYVDTLYPGFEGTEMWNPN
RELSEDCLYLNVWTPYPRPTSPTPVLIWIYGGGFYSGASSLDVYDGRFLAQVEGTVLVSM
NYRVGTFGFLALPGSREAPGNVGLLDQRLALQWVQENIAAFGGDPMSVTLFGESAGAASV
GMHILSLPSRSLFHRAVLQSGTPNGPWATVSAGEARRRATLLARLVGCPPGGAGGNDTEL
ISCLRTRPAQDLVDHEWHVLPQESIFRFSFVPVVDGDFLSDTPDALINTGDFQDLQVLVG
VVKDEGSYFLVYGVPGFSKDNESLISRAQFLAGVRIGVPQASDLAAEAVVLHYTDWLHPE
DPAHLRDAMSAVVGDHNVVCPVAQLAGRLAAQGARVYAYIFEHRASTLTWPLWMGVPHGY
EIEFIFGLPLDPSLNYTVEERIFAQRLMQYWTNFARTGDPNDPRDSKSPRWPPYTTAAQQ
YVSLNLKPLEVRRGLRAQTCAFWNRFLPKLLSATDTLDEAERQWKAEFHRWSSYMVHWKN
QFDHYSKQERCSDL
|
|
|
|---|
| BDBM10972 |
|---|
| n/a |
|---|
| Name | BDBM10972 |
|---|
| Synonyms: | (3aS,8aR)-1,3a,8-trimethyl-1H,2H,3H,3aH,8H,8aH-pyrrolo[2,3-b]indol-5-yl N-heptylcarbamate | CHEMBL433041 | Eptastigmine | Heptylphysostigmine |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H33N3O2 |
|---|
| Mol. Mass. | 359.5056 |
|---|
| SMILES | [H][C@]12N(C)CC[C@@]1(C)c1cc(OC(=O)NCCCCCCC)ccc1N2C |
|---|
| Structure | 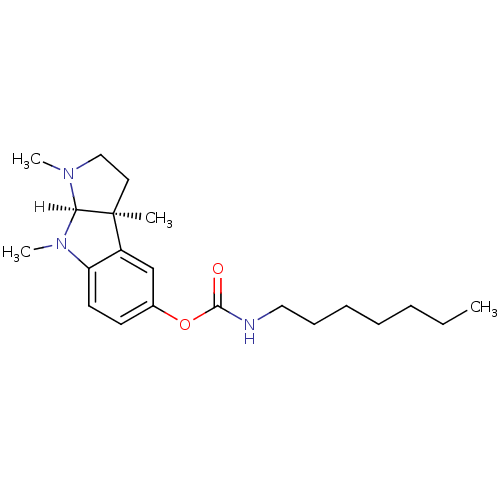
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Martin, LL; Davis, L; Klein, JT; Nemoto, P; Olsen, GE; Bores, GM; Camacho, F; Petko, WW; Rush, DK; Selk, D; Smith, CP; Vargas, HM; Winslow, JT; Effland, RC; Fink, DM Synthesis and preliminary structure-activity relationships of 1-[(3-fluoro-4-pyridinyl)amino]-3-methyl-1H-indol-5-yl methyl carbamate (P10358), a novel acetylcholinesterase inhibitor Bioorg Med Chem Lett7:157-162 (1997) Article
Martin, LL; Davis, L; Klein, JT; Nemoto, P; Olsen, GE; Bores, GM; Camacho, F; Petko, WW; Rush, DK; Selk, D; Smith, CP; Vargas, HM; Winslow, JT; Effland, RC; Fink, DM Synthesis and preliminary structure-activity relationships of 1-[(3-fluoro-4-pyridinyl)amino]-3-methyl-1H-indol-5-yl methyl carbamate (P10358), a novel acetylcholinesterase inhibitor Bioorg Med Chem Lett7:157-162 (1997) Article