 Found 861 hits with Last Name = 'davis' and Initial = 'l'
Found 861 hits with Last Name = 'davis' and Initial = 'l' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Cathepsin K
(Homo sapiens (Human)) | BDBM50098576
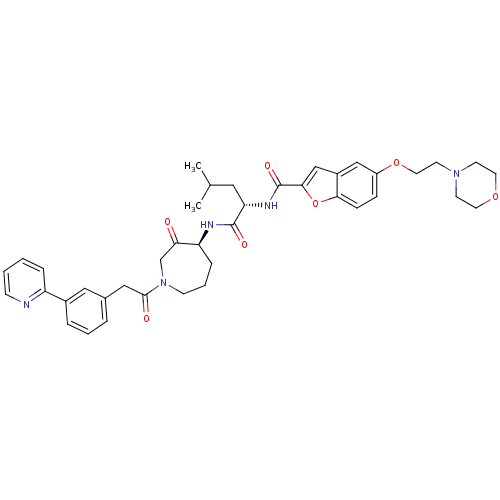
(5-(2-MORPHOLIN-4-YLETHOXY)BENZOFURAN-2-CARBOXYLIC ...)Show SMILES CC(C)C[C@H](NC(=O)c1cc2cc(OCCN3CCOCC3)ccc2o1)C(=O)N[C@H]1CCCN(CC1=O)C(=O)Cc1cccc(c1)-c1ccccn1 Show InChI InChI=1S/C40H47N5O7/c1-27(2)21-34(43-40(49)37-25-30-24-31(11-12-36(30)52-37)51-20-17-44-15-18-50-19-16-44)39(48)42-33-10-6-14-45(26-35(33)46)38(47)23-28-7-5-8-29(22-28)32-9-3-4-13-41-32/h3-5,7-9,11-13,22,24-25,27,33-34H,6,10,14-21,23,26H2,1-2H3,(H,42,48)(H,43,49)/t33-,34-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
PubMed
| 0.00480 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of Human cathepsin K |
J Med Chem 44: 1380-95 (2001)
BindingDB Entry DOI: 10.7270/Q2QR4WDC |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cathepsin K
(Homo sapiens (Human)) | BDBM19769

((2S)-2-(1-benzofuran-2-ylformamido)-4-methyl-N-[(4...)Show SMILES CC(C)C[C@H](NC(=O)c1cc2ccccc2o1)C(=O)N[C@H]1CCCN(CC1=O)S(=O)(=O)c1ccccn1 |r| Show InChI InChI=1S/C26H30N4O6S/c1-17(2)14-20(29-26(33)23-15-18-8-3-4-10-22(18)36-23)25(32)28-19-9-7-13-30(16-21(19)31)37(34,35)24-11-5-6-12-27-24/h3-6,8,10-12,15,17,19-20H,7,9,13-14,16H2,1-2H3,(H,28,32)(H,29,33)/t19-,20-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
PubMed
| 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of Human cathepsin K |
J Med Chem 44: 1380-95 (2001)
BindingDB Entry DOI: 10.7270/Q2QR4WDC |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Procathepsin L
(Homo sapiens (Human)) | BDBM50098576

(5-(2-MORPHOLIN-4-YLETHOXY)BENZOFURAN-2-CARBOXYLIC ...)Show SMILES CC(C)C[C@H](NC(=O)c1cc2cc(OCCN3CCOCC3)ccc2o1)C(=O)N[C@H]1CCCN(CC1=O)C(=O)Cc1cccc(c1)-c1ccccn1 Show InChI InChI=1S/C40H47N5O7/c1-27(2)21-34(43-40(49)37-25-30-24-31(11-12-36(30)52-37)51-20-17-44-15-18-50-19-16-44)39(48)42-33-10-6-14-45(26-35(33)46)38(47)23-28-7-5-8-29(22-28)32-9-3-4-13-41-32/h3-5,7-9,11-13,22,24-25,27,33-34H,6,10,14-21,23,26H2,1-2H3,(H,42,48)(H,43,49)/t33-,34-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| 0.490 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against Human cathepsin L |
J Med Chem 44: 1380-95 (2001)
BindingDB Entry DOI: 10.7270/Q2QR4WDC |
More data for this
Ligand-Target Pair | |
Tryptase beta-2
(Homo sapiens (Human)) | BDBM50156461
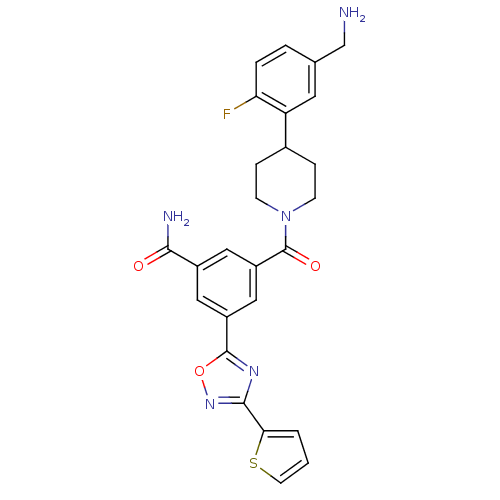
(3-[4-(5-Aminomethyl-2-fluoro-phenyl)-piperidine-1-...)Show SMILES NCc1ccc(F)c(c1)C1CCN(CC1)C(=O)c1cc(cc(c1)-c1nc(no1)-c1cccs1)C(N)=O Show InChI InChI=1S/C26H24FN5O3S/c27-21-4-3-15(14-28)10-20(21)16-5-7-32(8-6-16)26(34)19-12-17(23(29)33)11-18(13-19)25-30-24(31-35-25)22-2-1-9-36-22/h1-4,9-13,16H,5-8,14,28H2,(H2,29,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Aventis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibitory activity against human mast cell tryptase beta |
Bioorg Med Chem Lett 14: 6053-6 (2004)
Article DOI: 10.1016/j.bmcl.2004.09.065
BindingDB Entry DOI: 10.7270/Q2KH0MST |
More data for this
Ligand-Target Pair | |
Tryptase beta-2
(Homo sapiens (Human)) | BDBM50156460
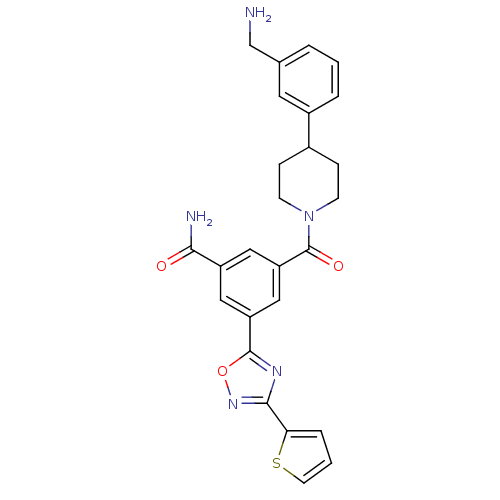
(3-[4-(3-Aminomethyl-phenyl)-piperidine-1-carbonyl]...)Show SMILES NCc1cccc(c1)C1CCN(CC1)C(=O)c1cc(cc(c1)-c1nc(no1)-c1cccs1)C(N)=O Show InChI InChI=1S/C26H25N5O3S/c27-15-16-3-1-4-18(11-16)17-6-8-31(9-7-17)26(33)21-13-19(23(28)32)12-20(14-21)25-29-24(30-34-25)22-5-2-10-35-22/h1-5,10-14,17H,6-9,15,27H2,(H2,28,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Aventis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibitory activity against human mast cell tryptase beta |
Bioorg Med Chem Lett 14: 6053-6 (2004)
Article DOI: 10.1016/j.bmcl.2004.09.065
BindingDB Entry DOI: 10.7270/Q2KH0MST |
More data for this
Ligand-Target Pair | |
Cathepsin K
(Homo sapiens (Human)) | BDBM50098580
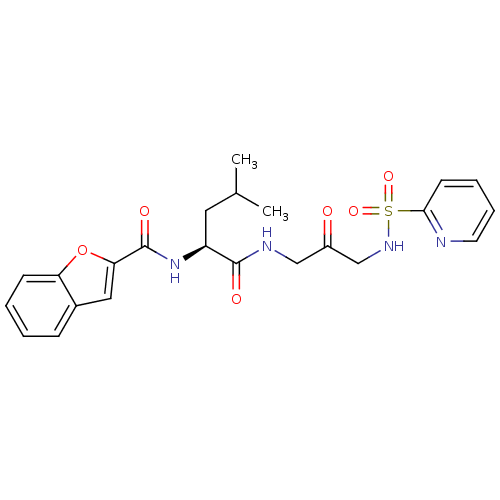
(Benzofuran-2-carboxylic acid {3-methyl-1-[2-oxo-3-...)Show SMILES CC(C)C[C@H](NC(=O)c1cc2ccccc2o1)C(=O)NCC(=O)CNS(=O)(=O)c1ccccn1 Show InChI InChI=1S/C23H26N4O6S/c1-15(2)11-18(27-23(30)20-12-16-7-3-4-8-19(16)33-20)22(29)25-13-17(28)14-26-34(31,32)21-9-5-6-10-24-21/h3-10,12,15,18,26H,11,13-14H2,1-2H3,(H,25,29)(H,27,30)/t18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of Human cathepsin K |
J Med Chem 44: 1380-95 (2001)
BindingDB Entry DOI: 10.7270/Q2QR4WDC |
More data for this
Ligand-Target Pair | |
D(1A) dopamine receptor
(Homo sapiens (Human)) | BDBM50429069
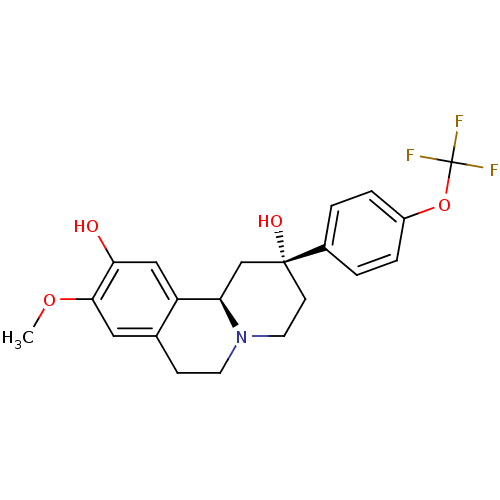
(CHEMBL2335736)Show SMILES COc1cc2CCN3CC[C@@](O)(C[C@@H]3c2cc1O)c1ccc(OC(F)(F)F)cc1 |r| Show InChI InChI=1S/C21H22F3NO4/c1-28-19-10-13-6-8-25-9-7-20(27,12-17(25)16(13)11-18(19)26)14-2-4-15(5-3-14)29-21(22,23)24/h2-5,10-11,17,26-27H,6-9,12H2,1H3/t17-,20-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi US
Curated by ChEMBL
| Assay Description
Displacement of [3H]SCH23390 from dopamine D1 receptor (unknown origin) expressed in CHO cell membranes after 60 mins |
Bioorg Med Chem Lett 23: 1498-501 (2013)
Article DOI: 10.1016/j.bmcl.2012.12.046
BindingDB Entry DOI: 10.7270/Q290255G |
More data for this
Ligand-Target Pair | |
Cathepsin K
(Homo sapiens (Human)) | BDBM50098584
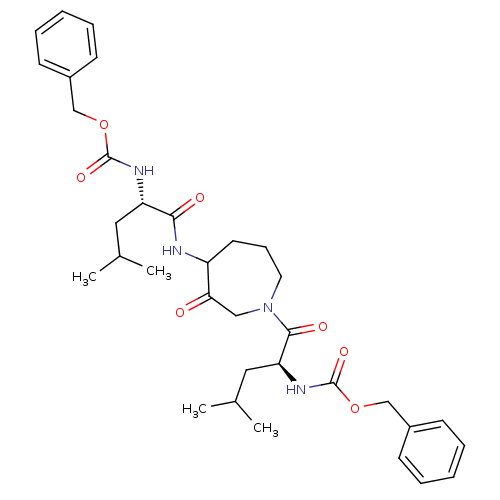
(CHEMBL31947 | {1-[4-(2-Benzyloxycarbonylamino-4-me...)Show SMILES CC(C)C[C@H](NC(=O)OCc1ccccc1)C(=O)NC1CCCN(CC1=O)C(=O)[C@H](CC(C)C)NC(=O)OCc1ccccc1 Show InChI InChI=1S/C34H46N4O7/c1-23(2)18-28(36-33(42)44-21-25-12-7-5-8-13-25)31(40)35-27-16-11-17-38(20-30(27)39)32(41)29(19-24(3)4)37-34(43)45-22-26-14-9-6-10-15-26/h5-10,12-15,23-24,27-29H,11,16-22H2,1-4H3,(H,35,40)(H,36,42)(H,37,43)/t27?,28-,29-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of Human cathepsin K |
J Med Chem 44: 1380-95 (2001)
BindingDB Entry DOI: 10.7270/Q2QR4WDC |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Androgen receptor
(Homo sapiens (Human)) | BDBM18161

((1S,2S,7S,10R,11S,14S,15S)-14-hydroxy-2,15-dimethy...)Show SMILES [H][C@@]12CC[C@H](O)[C@@]1(C)CC[C@@]1([H])[C@@]2([H])CC[C@@]2([H])CC(=O)CC[C@]12C |r| Show InChI InChI=1S/C19H30O2/c1-18-9-7-13(20)11-12(18)3-4-14-15-5-6-17(21)19(15,2)10-8-16(14)18/h12,14-17,21H,3-11H2,1-2H3/t12-,14-,15-,16-,17-,18-,19-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ligand Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Agonist activity to the human androgen receptor (hAR) in CV-1 cells |
Bioorg Med Chem Lett 9: 1335-40 (1999)
BindingDB Entry DOI: 10.7270/Q2JM2B4G |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Androgen receptor
(Homo sapiens (Human)) | BDBM50077104
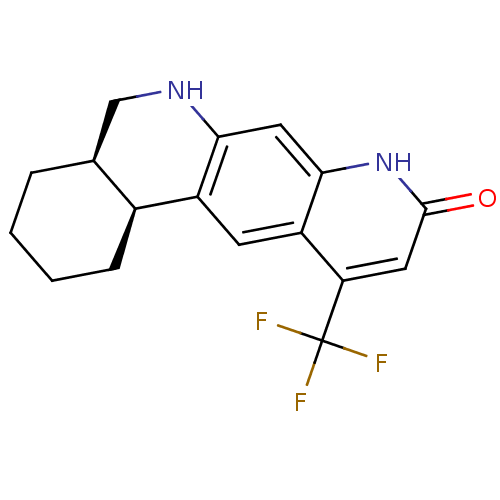
(CHEMBL431493 | Cis-(4aR,12bS)-11-Trifluoromethyl-2...)Show SMILES FC(F)(F)c1cc(=O)[nH]c2cc3NC[C@@H]4CCCC[C@@H]4c3cc12 Show InChI InChI=1S/C17H17F3N2O/c18-17(19,20)13-6-16(23)22-15-7-14-11(5-12(13)15)10-4-2-1-3-9(10)8-21-14/h5-7,9-10,21H,1-4,8H2,(H,22,23)/t9-,10-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ligand Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Binding affinity to the human androgen receptor (hAR), using [3H]DHT as radioligand in a competitive binding assay |
Bioorg Med Chem Lett 9: 1335-40 (1999)
BindingDB Entry DOI: 10.7270/Q2JM2B4G |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM50077095

(CHEMBL281945 | Trans-7-Methyl-4-trifluoromethyl-6-...)Show SMILES C[C@H]1CNc2cc3[nH]c(=O)cc(c3cc2C1CCC(F)(F)F)C(F)(F)F Show InChI InChI=1S/C17H16F6N2O/c1-8-7-24-13-6-14-11(12(17(21,22)23)5-15(26)25-14)4-10(13)9(8)2-3-16(18,19)20/h4-6,8-9,24H,2-3,7H2,1H3,(H,25,26)/t8-,9?/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ligand Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Binding affinity to the human androgen receptor (hAR), using [3H]DHT as radioligand in a competitive binding assay |
Bioorg Med Chem Lett 9: 1335-40 (1999)
BindingDB Entry DOI: 10.7270/Q2JM2B4G |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP3 subtype
(BOVINE) | BDBM50101828
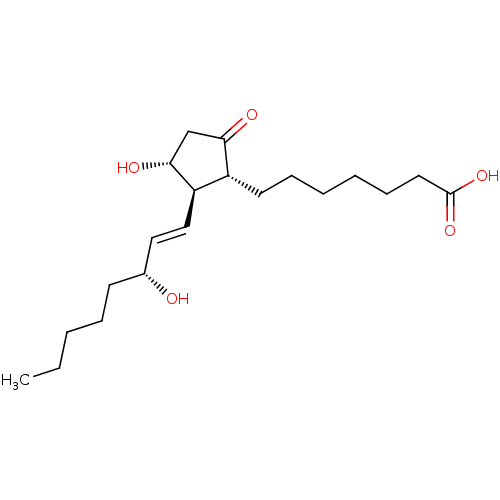
(7-[(1R,2R,3R)-3-Hydroxy-2-((E)-(R)-3-hydroxy-oct-1...)Show SMILES CCCCC[C@@H](O)\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1CCCCCCC(O)=O Show InChI InChI=1S/C20H34O5/c1-2-3-6-9-15(21)12-13-17-16(18(22)14-19(17)23)10-7-4-5-8-11-20(24)25/h12-13,15-17,19,21,23H,2-11,14H2,1H3,(H,24,25)/b13-12+/t15-,16-,17-,19-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Alcon Laboratories Inc.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1094-102 (1998)
BindingDB Entry DOI: 10.7270/Q2KK99BC |
More data for this
Ligand-Target Pair | |
Procathepsin L
(Homo sapiens (Human)) | BDBM19769

((2S)-2-(1-benzofuran-2-ylformamido)-4-methyl-N-[(4...)Show SMILES CC(C)C[C@H](NC(=O)c1cc2ccccc2o1)C(=O)N[C@H]1CCCN(CC1=O)S(=O)(=O)c1ccccn1 |r| Show InChI InChI=1S/C26H30N4O6S/c1-17(2)14-20(29-26(33)23-15-18-8-3-4-10-22(18)36-23)25(32)28-19-9-7-13-30(16-21(19)31)37(34,35)24-11-5-6-12-27-24/h3-6,8,10-12,15,17,19-20H,7,9,13-14,16H2,1-2H3,(H,28,32)(H,29,33)/t19-,20-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| 2.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against Human cathepsin L |
J Med Chem 44: 1380-95 (2001)
BindingDB Entry DOI: 10.7270/Q2QR4WDC |
More data for this
Ligand-Target Pair | |
Cathepsin K
(Homo sapiens (Human)) | BDBM50098579
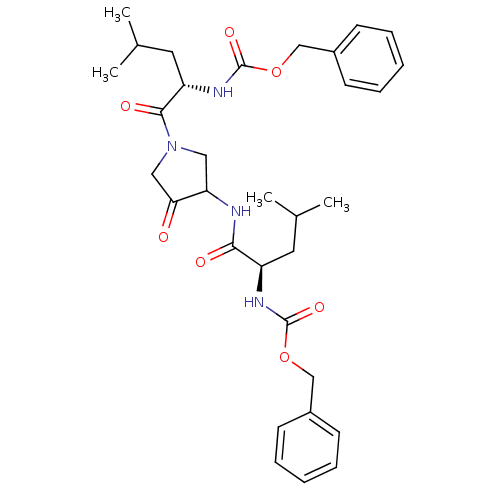
(CHEMBL29483 | {1-[3-(2-Benzyloxycarbonylamino-4-me...)Show SMILES CC(C)C[C@@H](NC(=O)OCc1ccccc1)C(=O)NC1CN(CC1=O)C(=O)[C@H](CC(C)C)NC(=O)OCc1ccccc1 Show InChI InChI=1S/C32H42N4O7/c1-21(2)15-25(34-31(40)42-19-23-11-7-5-8-12-23)29(38)33-27-17-36(18-28(27)37)30(39)26(16-22(3)4)35-32(41)43-20-24-13-9-6-10-14-24/h5-14,21-22,25-27H,15-20H2,1-4H3,(H,33,38)(H,34,40)(H,35,41)/t25-,26+,27?/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 2.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of Human cathepsin K |
J Med Chem 44: 1380-95 (2001)
BindingDB Entry DOI: 10.7270/Q2QR4WDC |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Cathepsin K
(Homo sapiens (Human)) | BDBM50098575
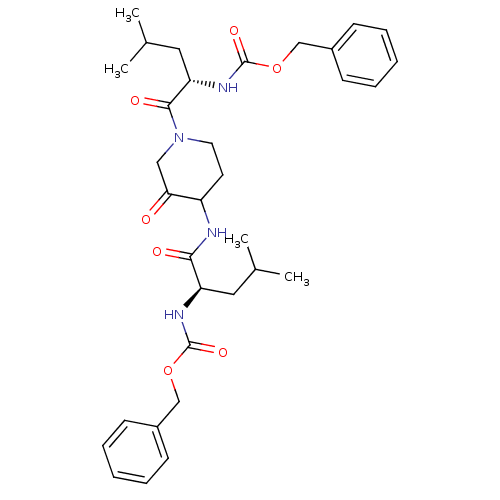
(CHEMBL281086 | {1-[4-(2-Benzyloxycarbonylamino-4-m...)Show SMILES CC(C)C[C@@H](NC(=O)OCc1ccccc1)C(=O)NC1CCN(CC1=O)C(=O)[C@H](CC(C)C)NC(=O)OCc1ccccc1 Show InChI InChI=1S/C33H44N4O7/c1-22(2)17-27(35-32(41)43-20-24-11-7-5-8-12-24)30(39)34-26-15-16-37(19-29(26)38)31(40)28(18-23(3)4)36-33(42)44-21-25-13-9-6-10-14-25/h5-14,22-23,26-28H,15-21H2,1-4H3,(H,34,39)(H,35,41)(H,36,42)/t26?,27-,28+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 2.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of Human cathepsin K |
J Med Chem 44: 1380-95 (2001)
BindingDB Entry DOI: 10.7270/Q2QR4WDC |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Prostaglandin E2 receptor EP3 subtype
(BOVINE) | BDBM35847

((15S)-prostaglandin E2 | (5Z,11alpha,13E,15S)-11,1...)Show SMILES CCCCC[C@H](O)\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1C\C=C/CCCC(O)=O Show InChI InChI=1S/C20H32O5/c1-2-3-6-9-15(21)12-13-17-16(18(22)14-19(17)23)10-7-4-5-8-11-20(24)25/h4,7,12-13,15-17,19,21,23H,2-3,5-6,8-11,14H2,1H3,(H,24,25)/b7-4-,13-12+/t15-,16+,17+,19+/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Alcon Laboratories Inc.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1094-102 (1998)
BindingDB Entry DOI: 10.7270/Q2KK99BC |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Androgen receptor
(Homo sapiens (Human)) | BDBM18161

((1S,2S,7S,10R,11S,14S,15S)-14-hydroxy-2,15-dimethy...)Show SMILES [H][C@@]12CC[C@H](O)[C@@]1(C)CC[C@@]1([H])[C@@]2([H])CC[C@@]2([H])CC(=O)CC[C@]12C |r| Show InChI InChI=1S/C19H30O2/c1-18-9-7-13(20)11-12(18)3-4-14-15-5-6-17(21)19(15,2)10-8-16(14)18/h12,14-17,21H,3-11H2,1-2H3/t12-,14-,15-,16-,17-,18-,19-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ligand Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Transcriptional repression in HepG2 cells expressing human glucocorticoid receptor |
J Med Chem 42: 210-2 (1999)
Article DOI: 10.1021/jm9806648
BindingDB Entry DOI: 10.7270/Q2F76D8W |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM50429069

(CHEMBL2335736)Show SMILES COc1cc2CCN3CC[C@@](O)(C[C@@H]3c2cc1O)c1ccc(OC(F)(F)F)cc1 |r| Show InChI InChI=1S/C21H22F3NO4/c1-28-19-10-13-6-8-25-9-7-20(27,12-17(25)16(13)11-18(19)26)14-2-4-15(5-3-14)29-21(22,23)24/h2-5,10-11,17,26-27H,6-9,12H2,1H3/t17-,20-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi US
Curated by ChEMBL
| Assay Description
Displacement of [3H]N-methylspiperone from dopamine D2 receptor (unknown origin) expressed in CHO cell membranes after 60 mins |
Bioorg Med Chem Lett 23: 1498-501 (2013)
Article DOI: 10.1016/j.bmcl.2012.12.046
BindingDB Entry DOI: 10.7270/Q290255G |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM50073060
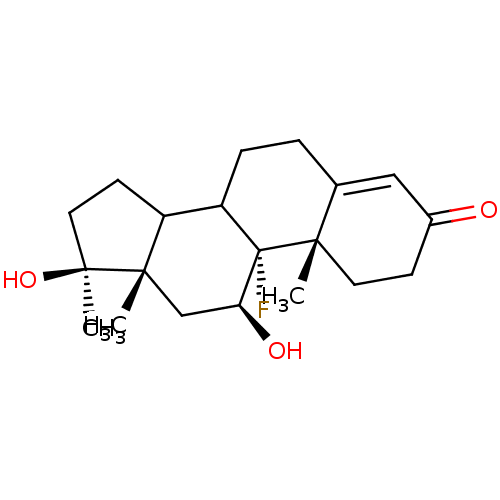
((9R,10S,11S,13S,17S)-9-Fluoro-11,17-dihydroxy-10,1...)Show SMILES C[C@]1(O)CCC2C3CCC4=CC(=O)CC[C@]4(C)[C@@]3(F)[C@@H](O)C[C@]12C |t:9| Show InChI InChI=1S/C20H29FO3/c1-17-8-6-13(22)10-12(17)4-5-15-14-7-9-19(3,24)18(14,2)11-16(23)20(15,17)21/h10,14-16,23-24H,4-9,11H2,1-3H3/t14?,15?,16-,17-,18-,19-,20-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ligand Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Binding affinity for human Androgen receptor expressed in COS-1 cells |
J Med Chem 42: 210-2 (1999)
Article DOI: 10.1021/jm9806648
BindingDB Entry DOI: 10.7270/Q2F76D8W |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM19769

((2S)-2-(1-benzofuran-2-ylformamido)-4-methyl-N-[(4...)Show SMILES CC(C)C[C@H](NC(=O)c1cc2ccccc2o1)C(=O)N[C@H]1CCCN(CC1=O)S(=O)(=O)c1ccccn1 |r| Show InChI InChI=1S/C26H30N4O6S/c1-17(2)14-20(29-26(33)23-15-18-8-3-4-10-22(18)36-23)25(32)28-19-9-7-13-30(16-21(19)31)37(34,35)24-11-5-6-12-27-24/h3-6,8,10-12,15,17,19-20H,7,9,13-14,16H2,1-2H3,(H,28,32)(H,29,33)/t19-,20-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against Human cathepsin S |
J Med Chem 44: 1380-95 (2001)
BindingDB Entry DOI: 10.7270/Q2QR4WDC |
More data for this
Ligand-Target Pair | |
D(1A) dopamine receptor
(Homo sapiens (Human)) | BDBM50429068
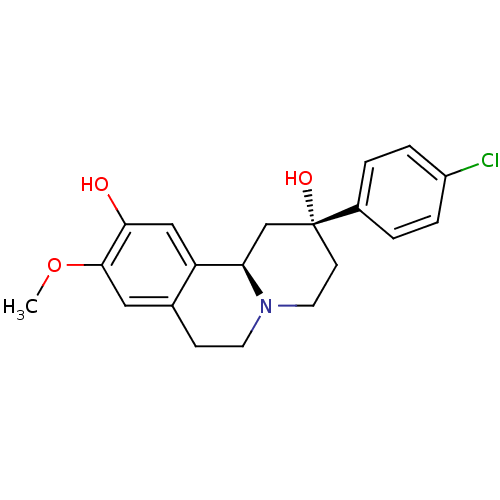
(CHEMBL2335740)Show SMILES COc1cc2CCN3CC[C@@](O)(C[C@@H]3c2cc1O)c1ccc(Cl)cc1 |r| Show InChI InChI=1S/C20H22ClNO3/c1-25-19-10-13-6-8-22-9-7-20(24,14-2-4-15(21)5-3-14)12-17(22)16(13)11-18(19)23/h2-5,10-11,17,23-24H,6-9,12H2,1H3/t17-,20-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi US
Curated by ChEMBL
| Assay Description
Displacement of [3H]SCH23390 from dopamine D1 receptor (unknown origin) expressed in CHO cell membranes after 60 mins |
Bioorg Med Chem Lett 23: 1498-501 (2013)
Article DOI: 10.1016/j.bmcl.2012.12.046
BindingDB Entry DOI: 10.7270/Q290255G |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP3 subtype
(BOVINE) | BDBM35847

((15S)-prostaglandin E2 | (5Z,11alpha,13E,15S)-11,1...)Show SMILES CCCCC[C@H](O)\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1C\C=C/CCCC(O)=O Show InChI InChI=1S/C20H32O5/c1-2-3-6-9-15(21)12-13-17-16(18(22)14-19(17)23)10-7-4-5-8-11-20(24)25/h4,7,12-13,15-17,19,21,23H,2-3,5-6,8-11,14H2,1H3,(H,24,25)/b7-4-,13-12+/t15-,16+,17+,19+/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Alcon Laboratories Inc.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1094-102 (1998)
BindingDB Entry DOI: 10.7270/Q2KK99BC |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Prostaglandin E2 receptor EP3 subtype
(BOVINE) | BDBM50101828

(7-[(1R,2R,3R)-3-Hydroxy-2-((E)-(R)-3-hydroxy-oct-1...)Show SMILES CCCCC[C@@H](O)\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1CCCCCCC(O)=O Show InChI InChI=1S/C20H34O5/c1-2-3-6-9-15(21)12-13-17-16(18(22)14-19(17)23)10-7-4-5-8-11-20(24)25/h12-13,15-17,19,21,23H,2-11,14H2,1H3,(H,24,25)/b13-12+/t15-,16-,17-,19-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Alcon Laboratories Inc.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1094-102 (1998)
BindingDB Entry DOI: 10.7270/Q2KK99BC |
More data for this
Ligand-Target Pair | |
Tryptase beta-2
(Homo sapiens (Human)) | BDBM50156457
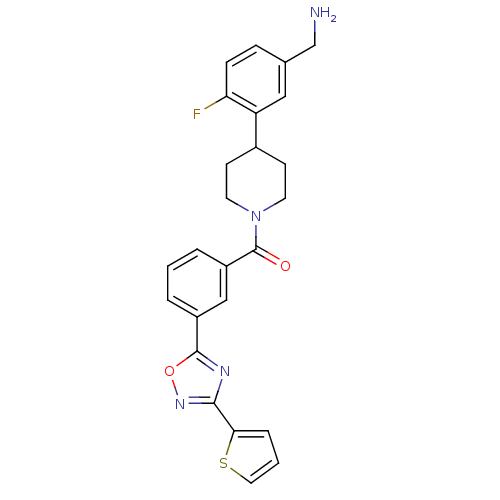
(CHEMBL186730 | [4-(5-Aminomethyl-2-fluoro-phenyl)-...)Show SMILES NCc1ccc(F)c(c1)C1CCN(CC1)C(=O)c1cccc(c1)-c1nc(no1)-c1cccs1 Show InChI InChI=1S/C25H23FN4O2S/c26-21-7-6-16(15-27)13-20(21)17-8-10-30(11-9-17)25(31)19-4-1-3-18(14-19)24-28-23(29-32-24)22-5-2-12-33-22/h1-7,12-14,17H,8-11,15,27H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Aventis Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibitory activity against human mast cell tryptase beta |
Bioorg Med Chem Lett 14: 6053-6 (2004)
Article DOI: 10.1016/j.bmcl.2004.09.065
BindingDB Entry DOI: 10.7270/Q2KH0MST |
More data for this
Ligand-Target Pair | |
Integrin alpha-IIb/beta-3
(Homo sapiens (Human)) | BDBM50036088
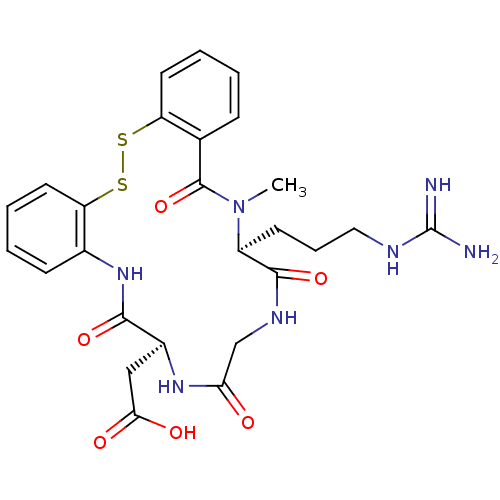
(CHEMBL18734 | [(6S,13S)-13-(3-Guanidino-propyl)-14...)Show SMILES CN1[C@@H](CCCNC(N)=N)C(=O)NCC(=O)N[C@@H](CC(O)=O)C(=O)Nc2ccccc2SSc2ccccc2C1=O Show InChI InChI=1S/C26H31N7O6S2/c1-33-18(9-6-12-29-26(27)28)24(38)30-14-21(34)31-17(13-22(35)36)23(37)32-16-8-3-5-11-20(16)41-40-19-10-4-2-7-15(19)25(33)39/h2-5,7-8,10-11,17-18H,6,9,12-14H2,1H3,(H,30,38)(H,31,34)(H,32,37)(H,35,36)(H4,27,28,29)/t17-,18-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 4.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Dissociation constant for platelet glycoprotein alphaIIb-beta3 integrin from human platelets |
Bioorg Med Chem Lett 3: 1179-1184 (1993)
Article DOI: 10.1016/S0960-894X(00)80310-8
BindingDB Entry DOI: 10.7270/Q2W959PC |
More data for this
Ligand-Target Pair | |
Cathepsin K
(Homo sapiens (Human)) | BDBM50098581

(5-(2-Morpholin-4-yl-ethoxy)-benzofuran-2-carboxyli...)Show SMILES CC(C)C[C@H](NC(=O)c1cc2cc(OCCN3CCOCC3)ccc2o1)C(=O)N[C@@H]1CCCN(CC1=O)C(=O)Cc1cccc(c1)-c1ccccn1 Show InChI InChI=1S/C40H47N5O7/c1-27(2)21-34(43-40(49)37-25-30-24-31(11-12-36(30)52-37)51-20-17-44-15-18-50-19-16-44)39(48)42-33-10-6-14-45(26-35(33)46)38(47)23-28-7-5-8-29(22-28)32-9-3-4-13-41-32/h3-5,7-9,11-13,22,24-25,27,33-34H,6,10,14-21,23,26H2,1-2H3,(H,42,48)(H,43,49)/t33-,34+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 4.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of Human cathepsin K |
J Med Chem 44: 1380-95 (2001)
BindingDB Entry DOI: 10.7270/Q2QR4WDC |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Cathepsin K
(Rattus norvegicus) | BDBM50098576

(5-(2-MORPHOLIN-4-YLETHOXY)BENZOFURAN-2-CARBOXYLIC ...)Show SMILES CC(C)C[C@H](NC(=O)c1cc2cc(OCCN3CCOCC3)ccc2o1)C(=O)N[C@H]1CCCN(CC1=O)C(=O)Cc1cccc(c1)-c1ccccn1 Show InChI InChI=1S/C40H47N5O7/c1-27(2)21-34(43-40(49)37-25-30-24-31(11-12-36(30)52-37)51-20-17-44-15-18-50-19-16-44)39(48)42-33-10-6-14-45(26-35(33)46)38(47)23-28-7-5-8-29(22-28)32-9-3-4-13-41-32/h3-5,7-9,11-13,22,24-25,27,33-34H,6,10,14-21,23,26H2,1-2H3,(H,42,48)(H,43,49)/t33-,34-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
PubMed
| 4.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against Rat cathepsin K |
J Med Chem 44: 1380-95 (2001)
BindingDB Entry DOI: 10.7270/Q2QR4WDC |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
D(1A) dopamine receptor
(Homo sapiens (Human)) | BDBM50429067
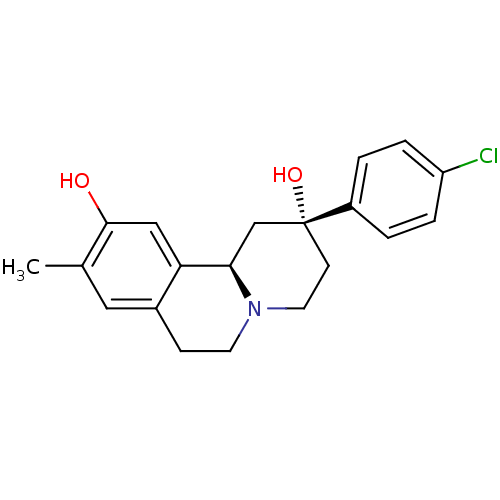
(CHEMBL2335737)Show SMILES Cc1cc2CCN3CC[C@@](O)(C[C@@H]3c2cc1O)c1ccc(Cl)cc1 |r| Show InChI InChI=1S/C20H22ClNO2/c1-13-10-14-6-8-22-9-7-20(24,15-2-4-16(21)5-3-15)12-18(22)17(14)11-19(13)23/h2-5,10-11,18,23-24H,6-9,12H2,1H3/t18-,20-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 5.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi US
Curated by ChEMBL
| Assay Description
Displacement of [3H]SCH23390 from dopamine D1 receptor (unknown origin) expressed in CHO cell membranes after 60 mins |
Bioorg Med Chem Lett 23: 1498-501 (2013)
Article DOI: 10.1016/j.bmcl.2012.12.046
BindingDB Entry DOI: 10.7270/Q290255G |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM18190
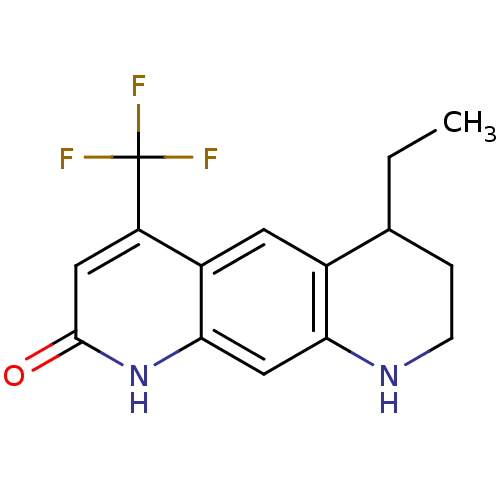
(6-ethyl-4-(trifluoromethyl)-1H,2H,6H,7H,8H,9H-pyri...)Show InChI InChI=1S/C15H15F3N2O/c1-2-8-3-4-19-12-7-13-10(5-9(8)12)11(15(16,17)18)6-14(21)20-13/h5-8,19H,2-4H2,1H3,(H,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ligand Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Binding affinity to the human androgen receptor (hAR), using [3H]DHT as radioligand in a competitive binding assay |
Bioorg Med Chem Lett 9: 1335-40 (1999)
BindingDB Entry DOI: 10.7270/Q2JM2B4G |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP3 subtype
(BOVINE) | BDBM82094
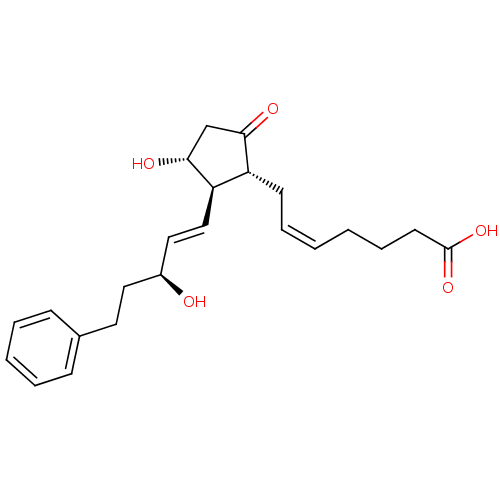
(17-PHENYL TRINOR PROSTAGLANDIN E2 | CAS_38315-43-4...)Show SMILES O[C@@H](CCc1ccccc1)\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1C\C=C/CCCC(O)=O |r| Show InChI InChI=1S/C23H30O5/c24-18(13-12-17-8-4-3-5-9-17)14-15-20-19(21(25)16-22(20)26)10-6-1-2-7-11-23(27)28/h1,3-6,8-9,14-15,18-20,22,24,26H,2,7,10-13,16H2,(H,27,28)/b6-1-,15-14+/t18-,19+,20+,22+/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Alcon Laboratories Inc.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1094-102 (1998)
BindingDB Entry DOI: 10.7270/Q2KK99BC |
More data for this
Ligand-Target Pair | |
Cathepsin K
(Homo sapiens (Human)) | BDBM19782
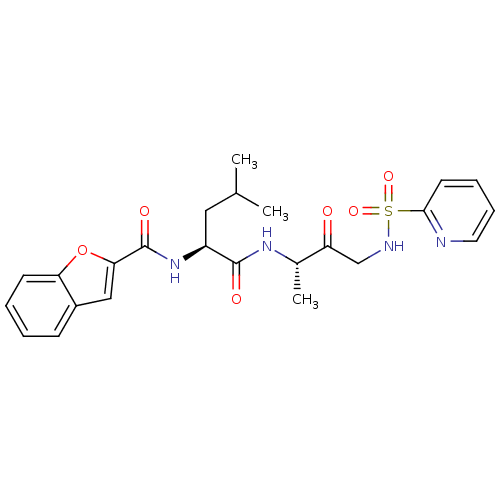
((2S)-2-(1-benzofuran-2-ylformamido)-4-methyl-N-[(2...)Show SMILES CC(C)C[C@H](NC(=O)c1cc2ccccc2o1)C(=O)N[C@@H](C)C(=O)CNS(=O)(=O)c1ccccn1 |r| Show InChI InChI=1S/C24H28N4O6S/c1-15(2)12-18(28-24(31)21-13-17-8-4-5-9-20(17)34-21)23(30)27-16(3)19(29)14-26-35(32,33)22-10-6-7-11-25-22/h4-11,13,15-16,18,26H,12,14H2,1-3H3,(H,27,30)(H,28,31)/t16-,18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 7.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of Human cathepsin K |
J Med Chem 44: 1380-95 (2001)
BindingDB Entry DOI: 10.7270/Q2QR4WDC |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM50077112
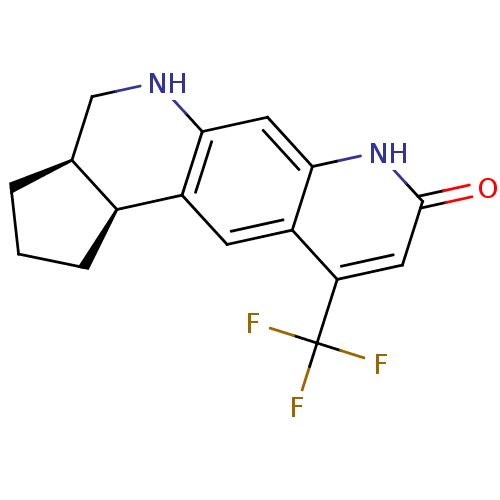
(CHEMBL27812 | Cis-(3aR,11bS)-10-Trifluoromethyl-1,...)Show SMILES FC(F)(F)c1cc(=O)[nH]c2cc3NC[C@@H]4CCC[C@@H]4c3cc12 Show InChI InChI=1S/C16H15F3N2O/c17-16(18,19)12-5-15(22)21-14-6-13-10(4-11(12)14)9-3-1-2-8(9)7-20-13/h4-6,8-9,20H,1-3,7H2,(H,21,22)/t8-,9-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ligand Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Agonist activity to the human androgen receptor (hAR) in CV-1 cells |
Bioorg Med Chem Lett 9: 1335-40 (1999)
BindingDB Entry DOI: 10.7270/Q2JM2B4G |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM50077115
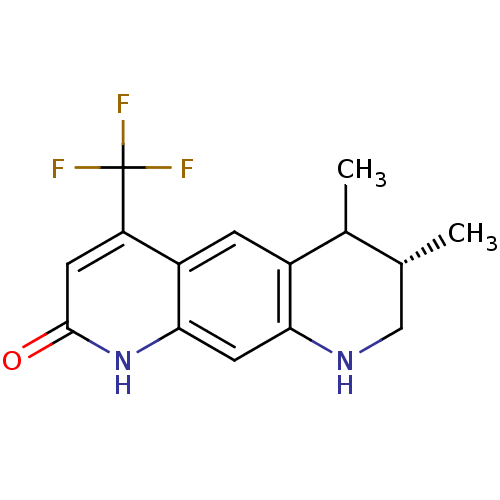
(CHEMBL282204 | Cis-6,7-Dimethyl-4-trifluoromethyl-...)Show SMILES C[C@@H]1CNc2cc3[nH]c(=O)cc(c3cc2C1C)C(F)(F)F Show InChI InChI=1S/C15H15F3N2O/c1-7-6-19-12-5-13-10(3-9(12)8(7)2)11(15(16,17)18)4-14(21)20-13/h3-5,7-8,19H,6H2,1-2H3,(H,20,21)/t7-,8?/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ligand Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Binding affinity to the human androgen receptor (hAR), using [3H]DHT as radioligand in a competitive binding assay |
Bioorg Med Chem Lett 9: 1335-40 (1999)
BindingDB Entry DOI: 10.7270/Q2JM2B4G |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM50077105
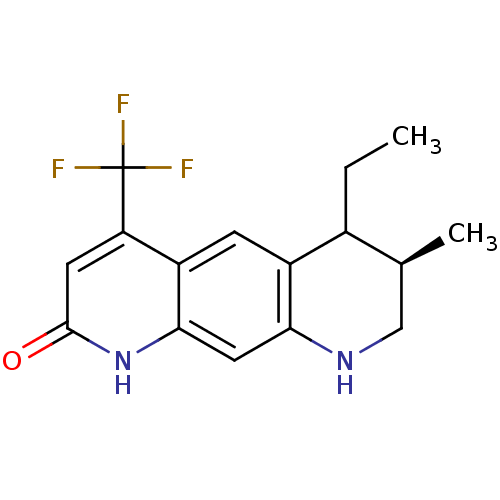
(CHEMBL418198 | Trans-6-Ethyl-7-methyl-4-trifluorom...)Show SMILES CCC1[C@@H](C)CNc2cc3[nH]c(=O)cc(c3cc12)C(F)(F)F Show InChI InChI=1S/C16H17F3N2O/c1-3-9-8(2)7-20-13-6-14-11(4-10(9)13)12(16(17,18)19)5-15(22)21-14/h4-6,8-9,20H,3,7H2,1-2H3,(H,21,22)/t8-,9?/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ligand Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Binding affinity to the human androgen receptor (hAR), using [3H]DHT as radioligand in a competitive binding assay |
Bioorg Med Chem Lett 9: 1335-40 (1999)
BindingDB Entry DOI: 10.7270/Q2JM2B4G |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP3 subtype
(BOVINE) | BDBM85174
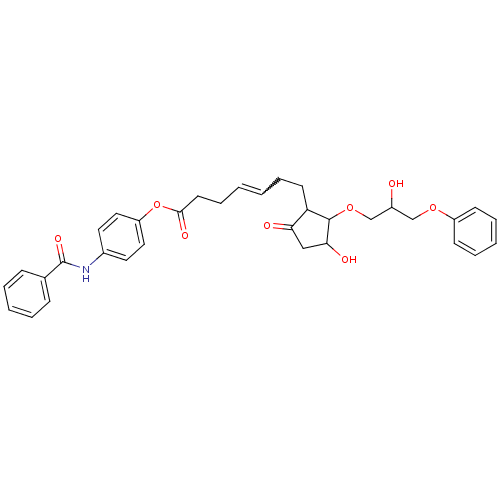
(CAS_5311224 | GR 63799X | NSC_5311224)Show SMILES OC(COC1C(O)CC(=O)C1CCC=CCCC(=O)Oc1ccc(NC(=O)c2ccccc2)cc1)COc1ccccc1 |w:13.13| Show InChI InChI=1S/C34H37NO8/c36-26(22-41-27-13-7-4-8-14-27)23-42-33-29(30(37)21-31(33)38)15-9-1-2-10-16-32(39)43-28-19-17-25(18-20-28)35-34(40)24-11-5-3-6-12-24/h1-8,11-14,17-20,26,29,31,33,36,38H,9-10,15-16,21-23H2,(H,35,40) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Alcon Laboratories Inc.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1094-102 (1998)
BindingDB Entry DOI: 10.7270/Q2KK99BC |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM50077111
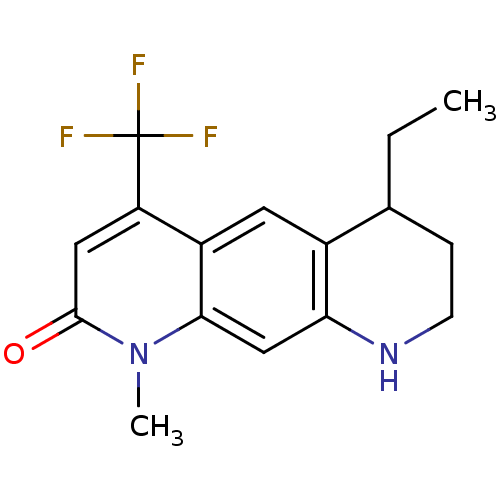
(6-Ethyl-1-methyl-4-trifluoromethyl-6,7,8,9-tetrahy...)Show InChI InChI=1S/C16H17F3N2O/c1-3-9-4-5-20-13-8-14-11(6-10(9)13)12(16(17,18)19)7-15(22)21(14)2/h6-9,20H,3-5H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ligand Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Binding affinity to the human androgen receptor (hAR), using [3H]DHT as radioligand in a competitive binding assay |
Bioorg Med Chem Lett 9: 1335-40 (1999)
BindingDB Entry DOI: 10.7270/Q2JM2B4G |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM50077097

(6-Methyl-4-trifluoromethyl-6,7,8,9-tetrahydro-1H-p...)Show InChI InChI=1S/C14H13F3N2O/c1-7-2-3-18-11-6-12-9(4-8(7)11)10(14(15,16)17)5-13(20)19-12/h4-7,18H,2-3H2,1H3,(H,19,20) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ligand Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Antagonistic activity against human androgen receptor (hAR) in CV-1 cells was determined as a function of maximal inhibition of dihydrotestosterone u... |
Bioorg Med Chem Lett 9: 1335-40 (1999)
BindingDB Entry DOI: 10.7270/Q2JM2B4G |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP3 subtype
(BOVINE) | BDBM85174

(CAS_5311224 | GR 63799X | NSC_5311224)Show SMILES OC(COC1C(O)CC(=O)C1CCC=CCCC(=O)Oc1ccc(NC(=O)c2ccccc2)cc1)COc1ccccc1 |w:13.13| Show InChI InChI=1S/C34H37NO8/c36-26(22-41-27-13-7-4-8-14-27)23-42-33-29(30(37)21-31(33)38)15-9-1-2-10-16-32(39)43-28-19-17-25(18-20-28)35-34(40)24-11-5-3-6-12-24/h1-8,11-14,17-20,26,29,31,33,36,38H,9-10,15-16,21-23H2,(H,35,40) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Alcon Laboratories Inc.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1094-102 (1998)
BindingDB Entry DOI: 10.7270/Q2KK99BC |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP3 subtype
(BOVINE) | BDBM85183
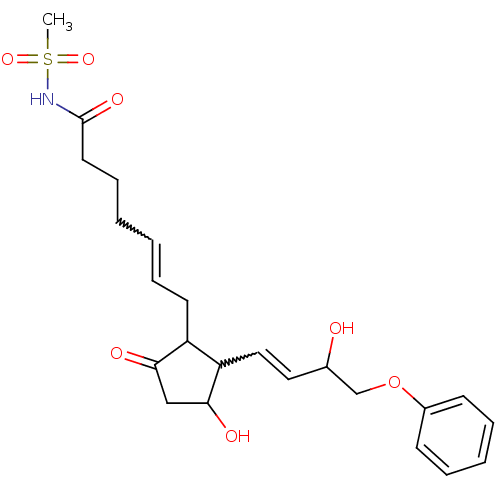
(CAS_60325-46-4 | NSC_43251 | SULPROSTONE)Show SMILES CS(=O)(=O)NC(=O)CCCC=CCC1C(C=CC(O)COc2ccccc2)C(O)CC1=O |w:10.9,15.14| Show InChI InChI=1S/C23H31NO7S/c1-32(29,30)24-23(28)12-8-3-2-7-11-19-20(22(27)15-21(19)26)14-13-17(25)16-31-18-9-5-4-6-10-18/h2,4-7,9-10,13-14,17,19-20,22,25,27H,3,8,11-12,15-16H2,1H3,(H,24,28) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| PubMed
| 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Alcon Laboratories Inc.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1094-102 (1998)
BindingDB Entry DOI: 10.7270/Q2KK99BC |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP3 subtype
(BOVINE) | BDBM85338
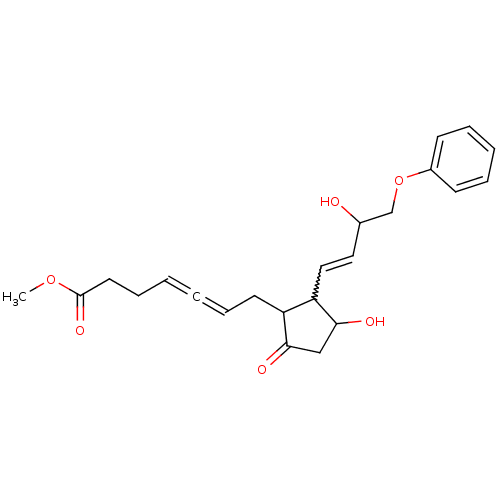
(CAS_73121-56-9 | ENPROSTIL | NSC_5282207)Show SMILES COC(=O)CCC=C=CCC1C(C=CC(O)COc2ccccc2)C(O)CC1=O |w:12.11,(-.79,-17.87,;.54,-17.1,;1.88,-17.87,;1.88,-19.41,;3.21,-17.1,;4.55,-17.87,;5.88,-17.1,;7.21,-17.87,;8.55,-18.64,;9.88,-17.87,;11.21,-18.64,;12.62,-18.02,;12.94,-16.51,;14.41,-16.03,;14.73,-14.53,;13.58,-13.5,;16.19,-14.05,;16.51,-12.55,;17.98,-12.07,;18.3,-10.56,;19.76,-10.09,;20.9,-11.12,;20.58,-12.62,;19.12,-13.1,;13.65,-19.16,;15.18,-19,;12.88,-20.49,;11.38,-20.17,;10.23,-21.2,)| Show InChI InChI=1S/C23H28O6/c1-28-23(27)12-8-3-2-7-11-19-20(22(26)15-21(19)25)14-13-17(24)16-29-18-9-5-4-6-10-18/h3-7,9-10,13-14,17,19-20,22,24,26H,8,11-12,15-16H2,1H3 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| PubMed
| 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Alcon Laboratories Inc.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1094-102 (1998)
BindingDB Entry DOI: 10.7270/Q2KK99BC |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM50077108
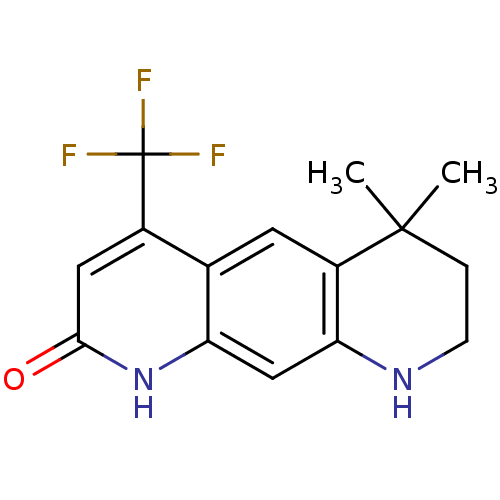
(6,6-Dimethyl-4-trifluoromethyl-6,7,8,9-tetrahydro-...)Show InChI InChI=1S/C15H15F3N2O/c1-14(2)3-4-19-12-7-11-8(5-10(12)14)9(15(16,17)18)6-13(21)20-11/h5-7,19H,3-4H2,1-2H3,(H,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ligand Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Binding affinity to the human androgen receptor (hAR), using [3H]DHT as radioligand in a competitive binding assay |
Bioorg Med Chem Lett 9: 1335-40 (1999)
BindingDB Entry DOI: 10.7270/Q2JM2B4G |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM50429067

(CHEMBL2335737)Show SMILES Cc1cc2CCN3CC[C@@](O)(C[C@@H]3c2cc1O)c1ccc(Cl)cc1 |r| Show InChI InChI=1S/C20H22ClNO2/c1-13-10-14-6-8-22-9-7-20(24,15-2-4-16(21)5-3-15)12-18(22)17(14)11-19(13)23/h2-5,10-11,18,23-24H,6-9,12H2,1H3/t18-,20-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi US
Curated by ChEMBL
| Assay Description
Displacement of [3H]N-methylspiperone from dopamine D2 receptor (unknown origin) expressed in CHO cell membranes after 60 mins |
Bioorg Med Chem Lett 23: 1498-501 (2013)
Article DOI: 10.1016/j.bmcl.2012.12.046
BindingDB Entry DOI: 10.7270/Q290255G |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM50077109
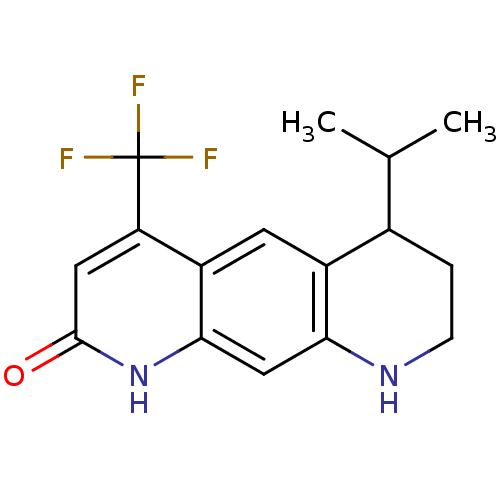
(6-Isopropyl-4-trifluoromethyl-6,7,8,9-tetrahydro-1...)Show InChI InChI=1S/C16H17F3N2O/c1-8(2)9-3-4-20-13-7-14-11(5-10(9)13)12(16(17,18)19)6-15(22)21-14/h5-9,20H,3-4H2,1-2H3,(H,21,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 13 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ligand Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Binding affinity to the human androgen receptor (hAR), using [3H]DHT as radioligand in a competitive binding assay |
Bioorg Med Chem Lett 9: 1335-40 (1999)
BindingDB Entry DOI: 10.7270/Q2JM2B4G |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50098576

(5-(2-MORPHOLIN-4-YLETHOXY)BENZOFURAN-2-CARBOXYLIC ...)Show SMILES CC(C)C[C@H](NC(=O)c1cc2cc(OCCN3CCOCC3)ccc2o1)C(=O)N[C@H]1CCCN(CC1=O)C(=O)Cc1cccc(c1)-c1ccccn1 Show InChI InChI=1S/C40H47N5O7/c1-27(2)21-34(43-40(49)37-25-30-24-31(11-12-36(30)52-37)51-20-17-44-15-18-50-19-16-44)39(48)42-33-10-6-14-45(26-35(33)46)38(47)23-28-7-5-8-29(22-28)32-9-3-4-13-41-32/h3-5,7-9,11-13,22,24-25,27,33-34H,6,10,14-21,23,26H2,1-2H3,(H,42,48)(H,43,49)/t33-,34-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| 14 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against Human cathepsin S |
J Med Chem 44: 1380-95 (2001)
BindingDB Entry DOI: 10.7270/Q2QR4WDC |
More data for this
Ligand-Target Pair | |
Cathepsin K
(Homo sapiens (Human)) | BDBM50098578
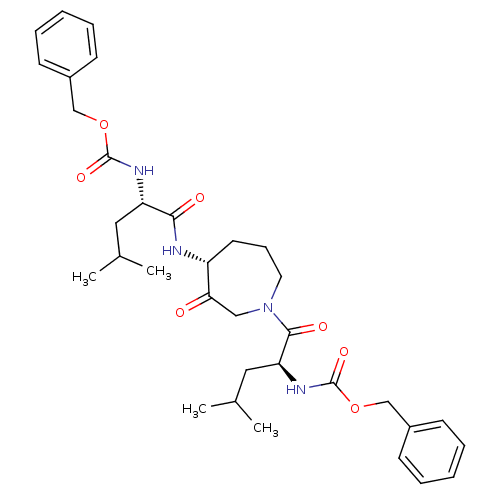
(CHEMBL286034 | {1-[4-(2-Benzyloxycarbonylamino-4-m...)Show SMILES CC(C)C[C@H](NC(=O)OCc1ccccc1)C(=O)N[C@@H]1CCCN(CC1=O)C(=O)[C@H](CC(C)C)NC(=O)OCc1ccccc1 Show InChI InChI=1S/C34H46N4O7/c1-23(2)18-28(36-33(42)44-21-25-12-7-5-8-13-25)31(40)35-27-16-11-17-38(20-30(27)39)32(41)29(19-24(3)4)37-34(43)45-22-26-14-9-6-10-15-26/h5-10,12-15,23-24,27-29H,11,16-22H2,1-4H3,(H,35,40)(H,36,42)(H,37,43)/t27-,28+,29+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 15 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of Human cathepsin K |
J Med Chem 44: 1380-95 (2001)
BindingDB Entry DOI: 10.7270/Q2QR4WDC |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM50429068

(CHEMBL2335740)Show SMILES COc1cc2CCN3CC[C@@](O)(C[C@@H]3c2cc1O)c1ccc(Cl)cc1 |r| Show InChI InChI=1S/C20H22ClNO3/c1-25-19-10-13-6-8-22-9-7-20(24,14-2-4-15(21)5-3-14)12-17(22)16(13)11-18(19)23/h2-5,10-11,17,23-24H,6-9,12H2,1H3/t17-,20-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 15 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi US
Curated by ChEMBL
| Assay Description
Displacement of [3H]N-methylspiperone from dopamine D2 receptor (unknown origin) expressed in CHO cell membranes after 60 mins |
Bioorg Med Chem Lett 23: 1498-501 (2013)
Article DOI: 10.1016/j.bmcl.2012.12.046
BindingDB Entry DOI: 10.7270/Q290255G |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM50077096

(CHEMBL25802 | Trans-11-Trifluoromethyl-2,3,4,4a,5,...)Show SMILES FC(F)(F)c1cc(=O)[nH]c2cc3NC[C@H]4CCCC[C@@H]4c3cc12 Show InChI InChI=1S/C17H17F3N2O/c18-17(19,20)13-6-16(23)22-15-7-14-11(5-12(13)15)10-4-2-1-3-9(10)8-21-14/h5-7,9-10,21H,1-4,8H2,(H,22,23)/t9-,10+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 15 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ligand Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Agonist activity to the human androgen receptor (hAR) in CV-1 cells |
Bioorg Med Chem Lett 9: 1335-40 (1999)
BindingDB Entry DOI: 10.7270/Q2JM2B4G |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM50077099

(CHEMBL436344 | Trans-6,7-Dimethyl-4-trifluoromethy...)Show SMILES C[C@H]1CNc2cc3[nH]c(=O)cc(c3cc2C1C)C(F)(F)F Show InChI InChI=1S/C15H15F3N2O/c1-7-6-19-12-5-13-10(3-9(12)8(7)2)11(15(16,17)18)4-14(21)20-13/h3-5,7-8,19H,6H2,1-2H3,(H,20,21)/t7-,8?/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 16 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ligand Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Binding affinity to the human androgen receptor (hAR), using [3H]DHT as radioligand in a competitive binding assay |
Bioorg Med Chem Lett 9: 1335-40 (1999)
BindingDB Entry DOI: 10.7270/Q2JM2B4G |
More data for this
Ligand-Target Pair | |
Procathepsin L
(Homo sapiens (Human)) | BDBM50098575

(CHEMBL281086 | {1-[4-(2-Benzyloxycarbonylamino-4-m...)Show SMILES CC(C)C[C@@H](NC(=O)OCc1ccccc1)C(=O)NC1CCN(CC1=O)C(=O)[C@H](CC(C)C)NC(=O)OCc1ccccc1 Show InChI InChI=1S/C33H44N4O7/c1-22(2)17-27(35-32(41)43-20-24-11-7-5-8-12-24)30(39)34-26-15-16-37(19-29(26)38)31(40)28(18-23(3)4)36-33(42)44-21-25-13-9-6-10-14-25/h5-14,22-23,26-28H,15-21H2,1-4H3,(H,34,39)(H,35,41)(H,36,42)/t26?,27-,28+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 16 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against Human cathepsin L |
J Med Chem 44: 1380-95 (2001)
BindingDB Entry DOI: 10.7270/Q2QR4WDC |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP3 subtype
(BOVINE) | BDBM82094

(17-PHENYL TRINOR PROSTAGLANDIN E2 | CAS_38315-43-4...)Show SMILES O[C@@H](CCc1ccccc1)\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1C\C=C/CCCC(O)=O |r| Show InChI InChI=1S/C23H30O5/c24-18(13-12-17-8-4-3-5-9-17)14-15-20-19(21(25)16-22(20)26)10-6-1-2-7-11-23(27)28/h1,3-6,8-9,14-15,18-20,22,24,26H,2,7,10-13,16H2,(H,27,28)/b6-1-,15-14+/t18-,19+,20+,22+/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 16 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Alcon Laboratories Inc.
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1094-102 (1998)
BindingDB Entry DOI: 10.7270/Q2KK99BC |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data