| Reaction Details |
|---|
 | Report a problem with these data |
| Target | C-C chemokine receptor type 3 |
|---|
| Ligand | BDBM50297172 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_581435 (CHEMBL1055679) |
|---|
| IC50 | 0.980000±n/a nM |
|---|
| Citation |  Sato, I; Morihira, K; Inami, H; Kubota, H; Morokata, T; Suzuki, K; Ohno, K; Iura, Y; Nitta, A; Imaoka, T; Takahashi, T; Takeuchi, M; Ohta, M; Tsukamoto, S Synthesis, biological evaluation, and metabolic stability of acrylamide derivatives as novel CCR3 antagonists. Bioorg Med Chem17:5989-6002 (2009) [PubMed] Article Sato, I; Morihira, K; Inami, H; Kubota, H; Morokata, T; Suzuki, K; Ohno, K; Iura, Y; Nitta, A; Imaoka, T; Takahashi, T; Takeuchi, M; Ohta, M; Tsukamoto, S Synthesis, biological evaluation, and metabolic stability of acrylamide derivatives as novel CCR3 antagonists. Bioorg Med Chem17:5989-6002 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| C-C chemokine receptor type 3 |
|---|
| Name: | C-C chemokine receptor type 3 |
|---|
| Synonyms: | C-C CKR-3 | C-C chemokine receptor type 3 (CCR3) | CC-CKR-3 | CCR-3 | CCR3 | CCR3_HUMAN | CD_antigen=CD193 | CKR3 | CMKBR3 | Eosinophil eotaxin receptor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 41053.88 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P51677 |
|---|
| Residue: | 355 |
|---|
| Sequence: | MTTSLDTVETFGTTSYYDDVGLLCEKADTRALMAQFVPPLYSLVFTVGLLGNVVVVMILI
KYRRLRIMTNIYLLNLAISDLLFLVTLPFWIHYVRGHNWVFGHGMCKLLSGFYHTGLYSE
IFFIILLTIDRYLAIVHAVFALRARTVTFGVITSIVTWGLAVLAALPEFIFYETEELFEE
TLCSALYPEDTVYSWRHFHTLRMTIFCLVLPLLVMAICYTGIIKTLLRCPSKKKYKAIRL
IFVIMAVFFIFWTPYNVAILLSSYQSILFGNDCERSKHLDLVMLVTEVIAYSHCCMNPVI
YAFVGERFRKYLRHFFHRHLLMHLGRYIPFLPSEKLERTSSVSPSTAEPELSIVF
|
|
|
|---|
| BDBM50297172 |
|---|
| n/a |
|---|
| Name | BDBM50297172 |
|---|
| Synonyms: | CHEMBL560275 | N-{(3R)-1-[(6-Fluoro-2-naphthyl)methyl]pyrrolidin-3-yl}-2-[1-(2-hydroxy-4-methylbenzoyl)piperidin-4-ylidene]-acetamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C30H32FN3O3 |
|---|
| Mol. Mass. | 501.5918 |
|---|
| SMILES | [#6]-c1ccc(-[#6](=O)-[#7]-2-[#6]-[#6]\[#6](-[#6]-[#6]-2)=[#6]/[#6](=O)-[#7]-[#6@@H]-2-[#6]-[#6]-[#7](-[#6]-c3ccc4cc(F)ccc4c3)-[#6]-2)c(-[#8])c1 |r| |
|---|
| Structure | 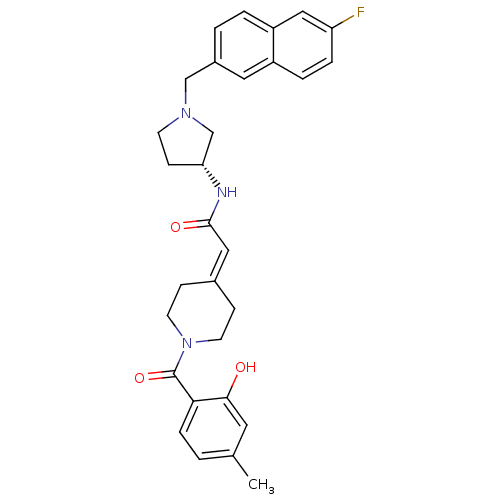
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Sato, I; Morihira, K; Inami, H; Kubota, H; Morokata, T; Suzuki, K; Ohno, K; Iura, Y; Nitta, A; Imaoka, T; Takahashi, T; Takeuchi, M; Ohta, M; Tsukamoto, S Synthesis, biological evaluation, and metabolic stability of acrylamide derivatives as novel CCR3 antagonists. Bioorg Med Chem17:5989-6002 (2009) [PubMed] Article
Sato, I; Morihira, K; Inami, H; Kubota, H; Morokata, T; Suzuki, K; Ohno, K; Iura, Y; Nitta, A; Imaoka, T; Takahashi, T; Takeuchi, M; Ohta, M; Tsukamoto, S Synthesis, biological evaluation, and metabolic stability of acrylamide derivatives as novel CCR3 antagonists. Bioorg Med Chem17:5989-6002 (2009) [PubMed] Article