| Reaction Details |
|---|
 | Report a problem with these data |
| Target | P2Y purinoceptor 12 |
|---|
| Ligand | BDBM50297387 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_582724 (CHEMBL1054986) |
|---|
| EC50 | 1.3±n/a nM |
|---|
| Citation |  Hutchinson, JH; Li, Y; Arruda, JM; Baccei, C; Bain, G; Chapman, C; Correa, L; Darlington, J; King, CD; Lee, C; Lorrain, D; Prodanovich, P; Rong, H; Santini, A; Stock, N; Prasit, P; Evans, JF 5-lipoxygenase-activating protein inhibitors: development of 3-[3-tert-butylsulfanyl-1-[4-(6-methoxy-pyridin-3-yl)-benzyl]-5-(pyridin-2-ylmethoxy)-1H-indol-2-yl]-2,2-dimethyl-propionic acid (AM103). J Med Chem52:5803-15 (2009) [PubMed] Article Hutchinson, JH; Li, Y; Arruda, JM; Baccei, C; Bain, G; Chapman, C; Correa, L; Darlington, J; King, CD; Lee, C; Lorrain, D; Prodanovich, P; Rong, H; Santini, A; Stock, N; Prasit, P; Evans, JF 5-lipoxygenase-activating protein inhibitors: development of 3-[3-tert-butylsulfanyl-1-[4-(6-methoxy-pyridin-3-yl)-benzyl]-5-(pyridin-2-ylmethoxy)-1H-indol-2-yl]-2,2-dimethyl-propionic acid (AM103). J Med Chem52:5803-15 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| P2Y purinoceptor 12 |
|---|
| Name: | P2Y purinoceptor 12 |
|---|
| Synonyms: | ADP-glucose receptor | ADPG-R | HORK3 | P2RY12 | P2T(AC) | P2Y purinoceptor 12 | P2Y(AC) | P2Y(ADP) | P2Y(cyc) | P2Y12 | P2Y12 platelet ADP receptor | P2Y12_HUMAN | Purinergic receptor P2Y12 | SP1999 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 39458.48 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q9H244 |
|---|
| Residue: | 342 |
|---|
| Sequence: | MQAVDNLTSAPGNTSLCTRDYKITQVLFPLLYTVLFFVGLITNGLAMRIFFQIRSKSNFI
IFLKNTVISDLLMILTFPFKILSDAKLGTGPLRTFVCQVTSVIFYFTMYISISFLGLITI
DRYQKTTRPFKTSNPKNLLGAKILSVVIWAFMFLLSLPNMILTNRQPRDKNVKKCSFLKS
EFGLVWHEIVNYICQVIFWINFLIVIVCYTLITKELYRSYVRTRGVGKVPRKKVNVKVFI
IIAVFFICFVPFHFARIPYTLSQTRDVFDCTAENTLFYVKESTLWLTSLNACLDPFIYFF
LCKSFRNSLISMLKCPNSATSLSQDNRKKEQDGGDPNEETPM
|
|
|
|---|
| BDBM50297387 |
|---|
| n/a |
|---|
| Name | BDBM50297387 |
|---|
| Synonyms: | (5S,6R,7E,9E,11Z,14Z)-6-(cystein-S-yl)-5-hydroxyicosa-7,9,11,14-tetraenoic acidS-{(1R,2E,4E,6Z,9Z)-1-[(1S)-4-carboxy-1-hydroxybutyl]pentadeca-2,4,6,9-tetraen-1-yl}-L-cysteine | (5S-(5R*,6S*(S*),7E,9E,11Z,14Z))-6-((2-Amino-2-carboxyethyl)thio)-5-hydroxy-7,9,11,14-eicosatetraenoic acid | 5S-hydroxy,6R-(S-cysteinyl),7E,9E,11Z,14Z-eicosatetraenoic acid | CHEMBL509456 | LTE4 | leukotriene E4 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H37NO5S |
|---|
| Mol. Mass. | 439.609 |
|---|
| SMILES | CCCCC\C=C/C\C=C/C=C/C=C/[C@@H](SC[C@H](N)C(O)=O)[C@@H](O)CCCC(O)=O |r| |
|---|
| Structure | 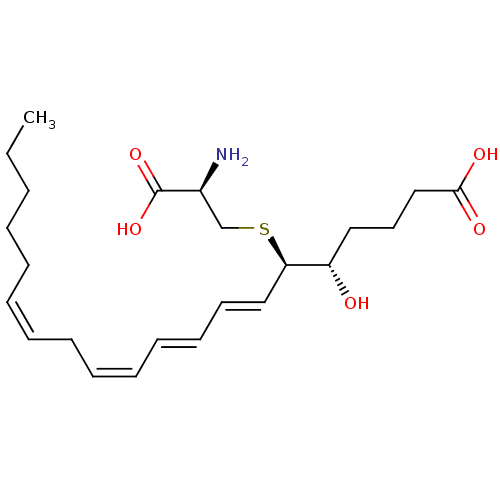
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Hutchinson, JH; Li, Y; Arruda, JM; Baccei, C; Bain, G; Chapman, C; Correa, L; Darlington, J; King, CD; Lee, C; Lorrain, D; Prodanovich, P; Rong, H; Santini, A; Stock, N; Prasit, P; Evans, JF 5-lipoxygenase-activating protein inhibitors: development of 3-[3-tert-butylsulfanyl-1-[4-(6-methoxy-pyridin-3-yl)-benzyl]-5-(pyridin-2-ylmethoxy)-1H-indol-2-yl]-2,2-dimethyl-propionic acid (AM103). J Med Chem52:5803-15 (2009) [PubMed] Article
Hutchinson, JH; Li, Y; Arruda, JM; Baccei, C; Bain, G; Chapman, C; Correa, L; Darlington, J; King, CD; Lee, C; Lorrain, D; Prodanovich, P; Rong, H; Santini, A; Stock, N; Prasit, P; Evans, JF 5-lipoxygenase-activating protein inhibitors: development of 3-[3-tert-butylsulfanyl-1-[4-(6-methoxy-pyridin-3-yl)-benzyl]-5-(pyridin-2-ylmethoxy)-1H-indol-2-yl]-2,2-dimethyl-propionic acid (AM103). J Med Chem52:5803-15 (2009) [PubMed] Article