| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Voltage-dependent T-type calcium channel subunit alpha-1H |
|---|
| Ligand | BDBM50322966 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_643468 (CHEMBL1212332) |
|---|
| IC50 | 71±n/a nM |
|---|
| Citation |  Smith, EM; Sorota, S; Kim, HM; McKittrick, BA; Nechuta, TL; Bennett, C; Knutson, C; Burnett, DA; Kieselgof, J; Tan, Z; Rindgen, D; Bridal, T; Zhou, X; Jia, YP; Dong, Z; Mullins, D; Zhang, X; Priestley, T; Correll, CC; Tulshian, D; Czarniecki, M; Greenlee, WJ T-type calcium channel blockers: spiro-piperidine azetidines and azetidinones-optimization, design and synthesis. Bioorg Med Chem Lett20:4602-6 (2010) [PubMed] Article Smith, EM; Sorota, S; Kim, HM; McKittrick, BA; Nechuta, TL; Bennett, C; Knutson, C; Burnett, DA; Kieselgof, J; Tan, Z; Rindgen, D; Bridal, T; Zhou, X; Jia, YP; Dong, Z; Mullins, D; Zhang, X; Priestley, T; Correll, CC; Tulshian, D; Czarniecki, M; Greenlee, WJ T-type calcium channel blockers: spiro-piperidine azetidines and azetidinones-optimization, design and synthesis. Bioorg Med Chem Lett20:4602-6 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Voltage-dependent T-type calcium channel subunit alpha-1H |
|---|
| Name: | Voltage-dependent T-type calcium channel subunit alpha-1H |
|---|
| Synonyms: | CAC1H_HUMAN | CACNA1H | Low-voltage-activated calcium channel alpha1 3.2 subunit | Voltage-gated T-type calcium channel | Voltage-gated calcium channel | Voltage-gated calcium channel subunit alpha Cav3.2 | voltage-dependent T-type calcium channel subunit alpha-1H isoform a |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 259187.68 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Calcium channel (Type T,L) 0 0::O95180 |
|---|
| Residue: | 2353 |
|---|
| Sequence: | MTEGARAADEVRVPLGAPPPGPAALVGASPESPGAPGREAERGSELGVSPSESPAAERGA
ELGADEEQRVPYPALAATVFFCLGQTTRPRSWCLRLVCNPWFEHVSMLVIMLNCVTLGMF
RPCEDVECGSERCNILEAFDAFIFAFFAVEMVIKMVALGLFGQKCYLGDTWNRLDFFIVV
AGMMEYSLDGHNVSLSAIRTVRVLRPLRAINRVPSMRILVTLLLDTLPMLGNVLLLCFFV
FFIFGIVGVQLWAGLLRNRCFLDSAFVRNNNLTFLRPYYQTEEGEENPFICSSRRDNGMQ
KCSHIPGRRELRMPCTLGWEAYTQPQAEGVGAARNACINWNQYYNVCRSGDSNPHNGAIN
FDNIGYAWIAIFQVITLEGWVDIMYYVMDAHSFYNFIYFILLIIVGSFFMINLCLVVIAT
QFSETKQRESQLMREQRARHLSNDSTLASFSEPGSCYEELLKYVGHIFRKVKRRSLRLYA
RWQSRWRKKVDPSAVQGQGPGHRQRRAGRHTASVHHLVYHHHHHHHHHYHFSHGSPRRPG
PEPGACDTRLVRAGAPPSPPSPGRGPPDAESVHSIYHADCHIEGPQERARVAHAAATAAA
SLRLATGLGTMNYPTILPSGVGSGKGSTSPGPKGKWAGGPPGTGGHGPLSLNSPDPYEKI
PHVVGEHGLGQAPGHLSGLSVPCPLPSPPAGTLTCELKSCPYCTRALEDPEGELSGSESG
DSDGRGVYEFTQDVRHGDRWDPTRPPRATDTPGPGPGSPQRRAQQRAAPGEPGWMGRLWV
TFSGKLRRIVDSKYFSRGIMMAILVNTLSMGVEYHEQPEELTNALEISNIVFTSMFALEM
LLKLLACGPLGYIRNPYNIFDGIIVVISVWEIVGQADGGLSVLRTFRLLRVLKLVRFLPA
LRRQLVVLVKTMDNVATFCTLLMLFIFIFSILGMHLFGCKFSLKTDTGDTVPDRKNFDSL
LWAIVTVFQILTQEDWNVVLYNGMASTSSWAALYFVALMTFGNYVLFNLLVAILVEGFQA
EGDANRSDTDEDKTSVHFEEDFHKLRELQTTELKMCSLAVTPNGHLEGRGSLSPPLIMCT
AATPMPTPKSSPFLDAAPSLPDSRRGSSSSGDPPLGDQKPPASLRSSPCAPWGPSGAWSS
RRSSWSSLGRAPSLKRRGQCGERESLLSGEGKGSTDDEAEDGRAAPGPRATPLRRAESLD
PRPLRPAALPPTKCRDRDGQVVALPSDFFLRIDSHREDAAELDDDSEDSCCLRLHKVLEP
YKPQWCRSREAWALYLFSPQNRFRVSCQKVITHKMFDHVVLVFIFLNCVTIALERPDIDP
GSTERVFLSVSNYIFTAIFVAEMMVKVVALGLLSGEHAYLQSSWNLLDGLLVLVSLVDIV
VAMASAGGAKILGVLRVLRLLRTLRPLRVISRAPGLKLVVETLISSLRPIGNIVLICCAF
FIIFGILGVQLFKGKFYYCEGPDTRNISTKAQCRAAHYRWVRRKYNFDNLGQALMSLFVL
SSKDGWVNIMYDGLDAVGVDQQPVQNHNPWMLLYFISFLLIVSFFVLNMFVGVVVENFHK
CRQHQEAEEARRREEKRLRRLERRRRSTFPSPEAQRRPYYADYSPTRRSIHSLCTSHYLD
LFITFIICVNVITMSMEHYNQPKSLDEALKYCNYVFTIVFVFEAALKLVAFGFRRFFKDR
WNQLDLAIVLLSLMGITLEEIEMSAALPINPTIIRIMRVLRIARVLKLLKMATGMRALLD
TVVQALPQVGNLGLLFMLLFFIYAALGVELFGRLECSEDNPCEGLSRHATFSNFGMAFLT
LFRVSTGDNWNGIMKDTLRECSREDKHCLSYLPALSPVYFVTFVLVAQFVLVNVVVAVLM
KHLEESNKEAREDAELDAEIELEMAQGPGSARRVDADRPPLPQESPGARDAPNLVARKVS
VSRMLSLPNDSYMFRPVVPASAPHPRPLQEVEMETYGAGTPLGSVASVHSPPAESCASLQ
IPLAVSSPARSGEPLHALSPRGTARSPSLSRLLCRQEAVHTDSLEGKIDSPRDTLDPAEP
GEKTPVRPVTQGGSLQSPPRSPRPASVRTRKHTFGQRCVSSRPAAPGGEEAEASDPADEE
VSHITSSACPWQPTAEPHGPEASPVAGGERDLRRLYSVDAQGFLDKPGRADEQWRPSAEL
GSGEPGEAKAWGPEAEPALGARRKKKMSPPCISVEPPAEDEGSARPSAAEGGSTTLRRRT
PSCEATPHRDSLEPTEGSGAGGDPAAKGERWGQASCRAEHLTVPSFAFEPLDLGVPSGDP
FLDGSHSVTPESRASSSGAIVPLEPPESEPPMPVGDPPEKRRGLYLTVPQCPLEKPGSPS
ATPAPGGGADDPV
|
|
|
|---|
| BDBM50322966 |
|---|
| n/a |
|---|
| Name | BDBM50322966 |
|---|
| Synonyms: | 7-(6-chloro-1H-benzo[d]imidazol-2-yl)-2-(3-chloro-4-fluorophenyl)-3-(pyridin-2-yl)-2,7-diazaspiro[3.5]nonan-1-one | CHEMBL1210900 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H20Cl2FN5O |
|---|
| Mol. Mass. | 496.364 |
|---|
| SMILES | Fc1ccc(cc1Cl)N1C(c2ccccn2)C2(CCN(CC2)c2nc3ccc(Cl)cc3[nH]2)C1=O |
|---|
| Structure | 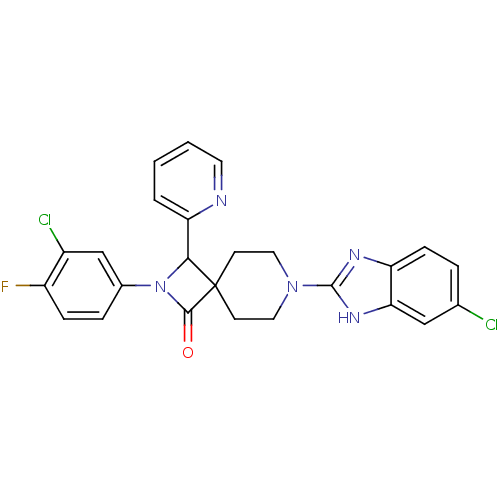
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Smith, EM; Sorota, S; Kim, HM; McKittrick, BA; Nechuta, TL; Bennett, C; Knutson, C; Burnett, DA; Kieselgof, J; Tan, Z; Rindgen, D; Bridal, T; Zhou, X; Jia, YP; Dong, Z; Mullins, D; Zhang, X; Priestley, T; Correll, CC; Tulshian, D; Czarniecki, M; Greenlee, WJ T-type calcium channel blockers: spiro-piperidine azetidines and azetidinones-optimization, design and synthesis. Bioorg Med Chem Lett20:4602-6 (2010) [PubMed] Article
Smith, EM; Sorota, S; Kim, HM; McKittrick, BA; Nechuta, TL; Bennett, C; Knutson, C; Burnett, DA; Kieselgof, J; Tan, Z; Rindgen, D; Bridal, T; Zhou, X; Jia, YP; Dong, Z; Mullins, D; Zhang, X; Priestley, T; Correll, CC; Tulshian, D; Czarniecki, M; Greenlee, WJ T-type calcium channel blockers: spiro-piperidine azetidines and azetidinones-optimization, design and synthesis. Bioorg Med Chem Lett20:4602-6 (2010) [PubMed] Article