| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Receptor tyrosine-protein kinase erbB-2 |
|---|
| Ligand | BDBM13535 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_655781 (CHEMBL1244825) |
|---|
| Kd | >10000±n/a nM |
|---|
| Citation |  Zarrinkar, PP; Gunawardane, RN; Cramer, MD; Gardner, MF; Brigham, D; Belli, B; Karaman, MW; Pratz, KW; Pallares, G; Chao, Q; Sprankle, KG; Patel, HK; Levis, M; Armstrong, RC; James, J; Bhagwat, SS AC220 is a uniquely potent and selective inhibitor of FLT3 for the treatment of acute myeloid leukemia (AML). Blood114:2984-92 (2009) [PubMed] Article Zarrinkar, PP; Gunawardane, RN; Cramer, MD; Gardner, MF; Brigham, D; Belli, B; Karaman, MW; Pratz, KW; Pallares, G; Chao, Q; Sprankle, KG; Patel, HK; Levis, M; Armstrong, RC; James, J; Bhagwat, SS AC220 is a uniquely potent and selective inhibitor of FLT3 for the treatment of acute myeloid leukemia (AML). Blood114:2984-92 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Receptor tyrosine-protein kinase erbB-2 |
|---|
| Name: | Receptor tyrosine-protein kinase erbB-2 |
|---|
| Synonyms: | 2.7.10.1 | C-erbB-2 | CD_antigen=CD340 | ERBB2 | ERBB2_HUMAN | ErbB-2/ErbB-3 heterodimer | FASN/HER2 | HER-2 Substrate | HER2 | MLN 19 | MLN19 | Metastatic lymph node gene 19 protein | NEU | NGL | Proto-oncogene Neu | Proto-oncogene c-ErbB-2 | Tyrosine kinase-type cell surface receptor HER2 | p185erbB2 |
|---|
| Type: | n/a |
|---|
| Mol. Mass.: | 137894.47 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P04626 |
|---|
| Residue: | 1255 |
|---|
| Sequence: | MELAALCRWGLLLALLPPGAASTQVCTGTDMKLRLPASPETHLDMLRHLYQGCQVVQGNL
ELTYLPTNASLSFLQDIQEVQGYVLIAHNQVRQVPLQRLRIVRGTQLFEDNYALAVLDNG
DPLNNTTPVTGASPGGLRELQLRSLTEILKGGVLIQRNPQLCYQDTILWKDIFHKNNQLA
LTLIDTNRSRACHPCSPMCKGSRCWGESSEDCQSLTRTVCAGGCARCKGPLPTDCCHEQC
AAGCTGPKHSDCLACLHFNHSGICELHCPALVTYNTDTFESMPNPEGRYTFGASCVTACP
YNYLSTDVGSCTLVCPLHNQEVTAEDGTQRCEKCSKPCARVCYGLGMEHLREVRAVTSAN
IQEFAGCKKIFGSLAFLPESFDGDPASNTAPLQPEQLQVFETLEEITGYLYISAWPDSLP
DLSVFQNLQVIRGRILHNGAYSLTLQGLGISWLGLRSLRELGSGLALIHHNTHLCFVHTV
PWDQLFRNPHQALLHTANRPEDECVGEGLACHQLCARGHCWGPGPTQCVNCSQFLRGQEC
VEECRVLQGLPREYVNARHCLPCHPECQPQNGSVTCFGPEADQCVACAHYKDPPFCVARC
PSGVKPDLSYMPIWKFPDEEGACQPCPINCTHSCVDLDDKGCPAEQRASPLTSIISAVVG
ILLVVVLGVVFGILIKRRQQKIRKYTMRRLLQETELVEPLTPSGAMPNQAQMRILKETEL
RKVKVLGSGAFGTVYKGIWIPDGENVKIPVAIKVLRENTSPKANKEILDEAYVMAGVGSP
YVSRLLGICLTSTVQLVTQLMPYGCLLDHVRENRGRLGSQDLLNWCMQIAKGMSYLEDVR
LVHRDLAARNVLVKSPNHVKITDFGLARLLDIDETEYHADGGKVPIKWMALESILRRRFT
HQSDVWSYGVTVWELMTFGAKPYDGIPAREIPDLLEKGERLPQPPICTIDVYMIMVKCWM
IDSECRPRFRELVSEFSRMARDPQRFVVIQNEDLGPASPLDSTFYRSLLEDDDMGDLVDA
EEYLVPQQGFFCPDPAPGAGGMVHHRHRSSSTRSGGGDLTLGLEPSEEEAPRSPLAPSEG
AGSDVFDGDLGMGAAKGLQSLPTHDPSPLQRYSEDPTVPLPSETDGYVAPLTCSPQPEYV
NQPDVRPQPPSPREGPLPAARPAGATLERPKTLSPGKNGVVKDVFAFGGAVENPEYLTPQ
GGAAPQPHPPPAFSPAFDNLYYWDQDPPERGAPPSTFKGTPTAENPEYLGLDVPV
|
|
|
|---|
| BDBM13535 |
|---|
| n/a |
|---|
| Name | BDBM13535 |
|---|
| Synonyms: | 4-[6-methoxy-7-(3-piperidin-1-ylpropoxy)quinazolin-4-yl]-N-(4-propan-2-yloxyphenyl)piperazine-1-carboxamide | 4-[6-methoxy-7-(3-piperidin-1-ylpropoxy)quinazolin-4-yl]-N-[4-(1-methylethoxy)phenyl]piperazine-1-carboxamide | 4-[6-methoxy-7-[3-(1-piperidinyl)propoxy]-4-quinazolinyl]-N-(4-propan-2-yloxyphenyl)-1-piperazinecarboxamide | 4-{6-methoxy-7-[3-(piperidin-1-yl)propoxy]quinazolin-4-yl}-N-[4-(propan-2-yloxy)phenyl]piperazine-1-carboxamide | CHEMBL124660 | MLN-518 | MLN518 | N-(4-isopropoxyphenyl)-4-[6-methoxy-7-(3-piperidinopropoxy)quinazolin-4-yl]piperazine-1-carboxamide | cid_3038522 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C31H42N6O4 |
|---|
| Mol. Mass. | 562.703 |
|---|
| SMILES | COc1cc2c(ncnc2cc1OCCCN1CCCCC1)N1CCN(CC1)C(=O)Nc1ccc(OC(C)C)cc1 |
|---|
| Structure | 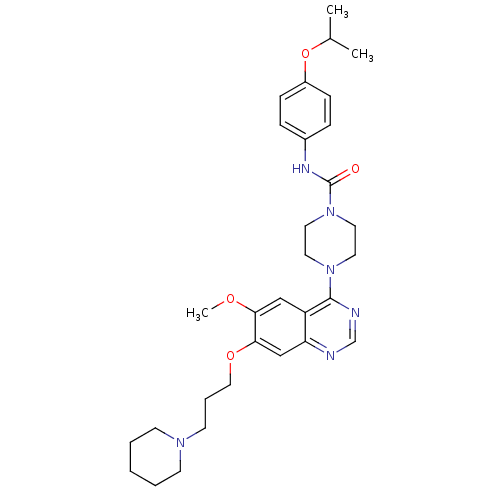
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Zarrinkar, PP; Gunawardane, RN; Cramer, MD; Gardner, MF; Brigham, D; Belli, B; Karaman, MW; Pratz, KW; Pallares, G; Chao, Q; Sprankle, KG; Patel, HK; Levis, M; Armstrong, RC; James, J; Bhagwat, SS AC220 is a uniquely potent and selective inhibitor of FLT3 for the treatment of acute myeloid leukemia (AML). Blood114:2984-92 (2009) [PubMed] Article
Zarrinkar, PP; Gunawardane, RN; Cramer, MD; Gardner, MF; Brigham, D; Belli, B; Karaman, MW; Pratz, KW; Pallares, G; Chao, Q; Sprankle, KG; Patel, HK; Levis, M; Armstrong, RC; James, J; Bhagwat, SS AC220 is a uniquely potent and selective inhibitor of FLT3 for the treatment of acute myeloid leukemia (AML). Blood114:2984-92 (2009) [PubMed] Article