| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cyclin-dependent kinase 18 [3-474] |
|---|
| Ligand | BDBM4814 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_655513 (CHEMBL1244557) |
|---|
| Kd | 1700±n/a nM |
|---|
| Citation |  Zarrinkar, PP; Gunawardane, RN; Cramer, MD; Gardner, MF; Brigham, D; Belli, B; Karaman, MW; Pratz, KW; Pallares, G; Chao, Q; Sprankle, KG; Patel, HK; Levis, M; Armstrong, RC; James, J; Bhagwat, SS AC220 is a uniquely potent and selective inhibitor of FLT3 for the treatment of acute myeloid leukemia (AML). Blood114:2984-92 (2009) [PubMed] Article Zarrinkar, PP; Gunawardane, RN; Cramer, MD; Gardner, MF; Brigham, D; Belli, B; Karaman, MW; Pratz, KW; Pallares, G; Chao, Q; Sprankle, KG; Patel, HK; Levis, M; Armstrong, RC; James, J; Bhagwat, SS AC220 is a uniquely potent and selective inhibitor of FLT3 for the treatment of acute myeloid leukemia (AML). Blood114:2984-92 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cyclin-dependent kinase 18 [3-474] |
|---|
| Name: | Cyclin-dependent kinase 18 [3-474] |
|---|
| Synonyms: | CDK18 | CDK18_HUMAN | PCTAIRE3 | PCTK3 | Serine/threonine-protein kinase PCTAIRE-3 |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 54193.55 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_774354 |
|---|
| Residue: | 472 |
|---|
| Sequence: | MNKMKNFKRRFSLSVPRTETIEESLAEFTEQFNQLHNRRNENLQLGPLGRDPPQECSTFS
PTDSGEEPGQLSPGVQFQRRQNQRRFSMEDVSKRLSLPMDIRLPQEFLQKLQMESPDLPK
PLSRMSRRASLSDIGFGKLETYVKLDKLGEGTYATVFKGRSKLTENLVALKEIRLEHEEG
APCTAIREVSLLKNLKHANIVTLHDLIHTDRSLTLVFEYLDSDLKQYLDHCGNLMSMHNV
KIFMFQLLRGLAYCHHRKILHRDLKPQNLLINERGELKLADFGLARAKSVPTKTYSNEVV
TLWYRPPDVLLGSTEYSTPIDMWGVGCIHYEMATGRPLFPGSTVKEELHLIFRLLGTPTE
ETWPGVTAFSEFRTYSFPCYLPQPLINHAPRLDTDGIHLLSSLLLYESKSRMSAEAALSH
SYFRSLGERVHQLEDTASIFSLKEIQLQKDPGYRGLAFQQPGRGKNRRQSIF
|
|
|
|---|
| BDBM4814 |
|---|
| n/a |
|---|
| Name | BDBM4814 |
|---|
| Synonyms: | CHEMBL535 | N-[2-(diethylamino)ethyl]-5-[(Z)-(5-fluoro-2-oxo-1,2-dihydro-3H-indol-3-ylidene)methyl]-2,4-dimethyl-1H-pyrrole-3-carboxamide | N-[2-(diethylamino)ethyl]-5-{[(3Z)-5-fluoro-2-oxo-2,3-dihydro-1H-indol-3-ylidene]methyl}-2,4-dimethyl-1H-pyrrole-3-carboxamide | SU11248 | SUNITINIB | SUNITINIB MALATE | US10464902, Sunitinib | US20240058459, Compound Sunit-inib | US9163010, Sunitinib | US9914707, SU11248 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H27FN4O2 |
|---|
| Mol. Mass. | 398.4738 |
|---|
| SMILES | CCN(CC)CCNC(=O)c1c(C)[nH]c(\C=C2/C(=O)Nc3ccc(F)cc23)c1C |
|---|
| Structure | 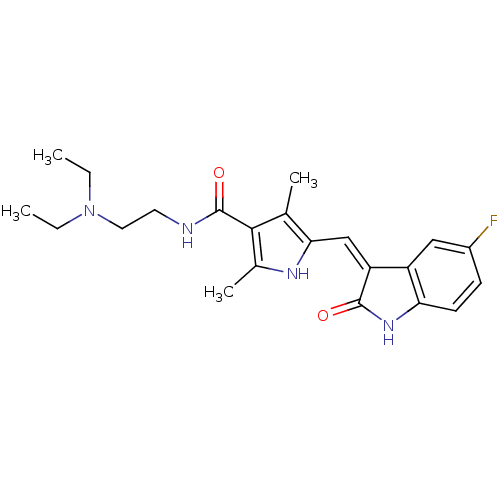
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Zarrinkar, PP; Gunawardane, RN; Cramer, MD; Gardner, MF; Brigham, D; Belli, B; Karaman, MW; Pratz, KW; Pallares, G; Chao, Q; Sprankle, KG; Patel, HK; Levis, M; Armstrong, RC; James, J; Bhagwat, SS AC220 is a uniquely potent and selective inhibitor of FLT3 for the treatment of acute myeloid leukemia (AML). Blood114:2984-92 (2009) [PubMed] Article
Zarrinkar, PP; Gunawardane, RN; Cramer, MD; Gardner, MF; Brigham, D; Belli, B; Karaman, MW; Pratz, KW; Pallares, G; Chao, Q; Sprankle, KG; Patel, HK; Levis, M; Armstrong, RC; James, J; Bhagwat, SS AC220 is a uniquely potent and selective inhibitor of FLT3 for the treatment of acute myeloid leukemia (AML). Blood114:2984-92 (2009) [PubMed] Article