| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Adenosine receptor A2a |
|---|
| Ligand | BDBM50331908 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_687323 (CHEMBL1291999) |
|---|
| Ki | 3509±n/a nM |
|---|
| Citation |  McGuinness, BF; Ho, KK; Stauffer, TM; Rokosz, LL; Mannava, N; Kultgen, SG; Saionz, K; Klon, A; Chen, W; Desai, H; Rogers, WL; Webb, M; Yin, J; Jiang, Y; Li, T; Yan, H; Jing, K; Zhang, S; Majumdar, KK; Srivastava, V; Saha, S Discovery of novel quinolinone adenosine A2B antagonists. Bioorg Med Chem Lett20:7414-20 (2010) [PubMed] Article McGuinness, BF; Ho, KK; Stauffer, TM; Rokosz, LL; Mannava, N; Kultgen, SG; Saionz, K; Klon, A; Chen, W; Desai, H; Rogers, WL; Webb, M; Yin, J; Jiang, Y; Li, T; Yan, H; Jing, K; Zhang, S; Majumdar, KK; Srivastava, V; Saha, S Discovery of novel quinolinone adenosine A2B antagonists. Bioorg Med Chem Lett20:7414-20 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Adenosine receptor A2a |
|---|
| Name: | Adenosine receptor A2a |
|---|
| Synonyms: | A2A adenosine receptor (hA2A) | AA2AR_HUMAN | ADENOSINE A2 | ADENOSINE A2a | ADORA2 | ADORA2A | Adenosine A2A receptor (A2AAR) |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 44716.46 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P29274 |
|---|
| Residue: | 412 |
|---|
| Sequence: | MPIMGSSVYITVELAIAVLAILGNVLVCWAVWLNSNLQNVTNYFVVSLAAADIAVGVLAI
PFAITISTGFCAACHGCLFIACFVLVLTQSSIFSLLAIAIDRYIAIRIPLRYNGLVTGTR
AKGIIAICWVLSFAIGLTPMLGWNNCGQPKEGKNHSQGCGEGQVACLFEDVVPMNYMVYF
NFFACVLVPLLLMLGVYLRIFLAARRQLKQMESQPLPGERARSTLQKEVHAAKSLAIIVG
LFALCWLPLHIINCFTFFCPDCSHAPLWLMYLAIVLSHTNSVVNPFIYAYRIREFRQTFR
KIIRSHVLRQQEPFKAAGTSARVLAAHGSDGEQVSLRLNGHPPGVWANGSAPHPERRPNG
YALGLVSGGSAQESQGNTGLPDVELLSHELKGVCPEPPGLDDPLAQDGAGVS
|
|
|
|---|
| BDBM50331908 |
|---|
| n/a |
|---|
| Name | BDBM50331908 |
|---|
| Synonyms: | CHEMBL1290436 | N-benzyl-2-(2-oxo-4-phenethyl-3-(thiazol-2-yl)-1,2-dihydroquinolin-6-yloxy)acetamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C29H25N3O3S |
|---|
| Mol. Mass. | 495.592 |
|---|
| SMILES | O=C(COc1ccc2[nH]c(=O)c(-c3nccs3)c(CCc3ccccc3)c2c1)NCc1ccccc1 |
|---|
| Structure | 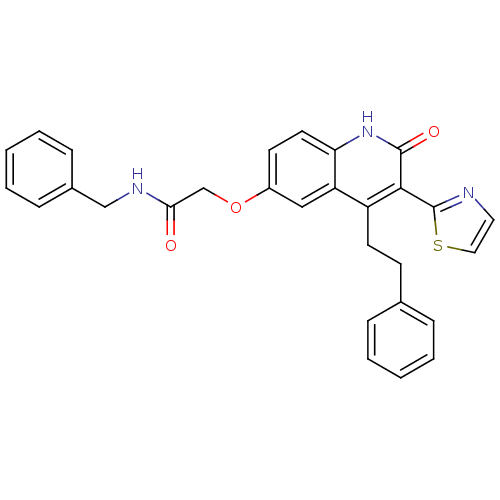
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 McGuinness, BF; Ho, KK; Stauffer, TM; Rokosz, LL; Mannava, N; Kultgen, SG; Saionz, K; Klon, A; Chen, W; Desai, H; Rogers, WL; Webb, M; Yin, J; Jiang, Y; Li, T; Yan, H; Jing, K; Zhang, S; Majumdar, KK; Srivastava, V; Saha, S Discovery of novel quinolinone adenosine A2B antagonists. Bioorg Med Chem Lett20:7414-20 (2010) [PubMed] Article
McGuinness, BF; Ho, KK; Stauffer, TM; Rokosz, LL; Mannava, N; Kultgen, SG; Saionz, K; Klon, A; Chen, W; Desai, H; Rogers, WL; Webb, M; Yin, J; Jiang, Y; Li, T; Yan, H; Jing, K; Zhang, S; Majumdar, KK; Srivastava, V; Saha, S Discovery of novel quinolinone adenosine A2B antagonists. Bioorg Med Chem Lett20:7414-20 (2010) [PubMed] Article