| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Leukotriene A-4 hydrolase |
|---|
| Ligand | BDBM50359080 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_789629 (CHEMBL1924714) |
|---|
| IC50 | >100000±n/a nM |
|---|
| Citation |  Stock, NS; Bain, G; Zunic, J; Li, Y; Ziff, J; Roppe, J; Santini, A; Darlington, J; Prodanovich, P; King, CD; Baccei, C; Lee, C; Rong, H; Chapman, C; Broadhead, A; Lorrain, D; Correa, L; Hutchinson, JH; Evans, JF; Prasit, P 5-Lipoxygenase-activating protein (FLAP) inhibitors. Part 4: development of 3-[3-tert-butylsulfanyl-1-[4-(6-ethoxypyridin-3-yl)benzyl]-5-(5-methylpyridin-2-ylmethoxy)-1H-indol-2-yl]-2,2-dimethylpropionic acid (AM803), a potent, oral, once daily FLAP inhibitor. J Med Chem54:8013-29 (2011) [PubMed] Article Stock, NS; Bain, G; Zunic, J; Li, Y; Ziff, J; Roppe, J; Santini, A; Darlington, J; Prodanovich, P; King, CD; Baccei, C; Lee, C; Rong, H; Chapman, C; Broadhead, A; Lorrain, D; Correa, L; Hutchinson, JH; Evans, JF; Prasit, P 5-Lipoxygenase-activating protein (FLAP) inhibitors. Part 4: development of 3-[3-tert-butylsulfanyl-1-[4-(6-ethoxypyridin-3-yl)benzyl]-5-(5-methylpyridin-2-ylmethoxy)-1H-indol-2-yl]-2,2-dimethylpropionic acid (AM803), a potent, oral, once daily FLAP inhibitor. J Med Chem54:8013-29 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Leukotriene A-4 hydrolase |
|---|
| Name: | Leukotriene A-4 hydrolase |
|---|
| Synonyms: | LKHA4_HUMAN | LTA-4 hydrolase | LTA4 | LTA4H | Leukotriene A(4) hydrolase | Leukotriene A-4 hydrolase (LTA4H) | Leukotriene A4 hydrolase |
|---|
| Type: | Hydrolase; metalloprotease |
|---|
| Mol. Mass.: | 69280.41 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Human recombinant LTA4H. |
|---|
| Residue: | 611 |
|---|
| Sequence: | MPEIVDTCSLASPASVCRTKHLHLRCSVDFTRRTLTGTAALTVQSQEDNLRSLVLDTKDL
TIEKVVINGQEVKYALGERQSYKGSPMEISLPIALSKNQEIVIEISFETSPKSSALQWLT
PEQTSGKEHPYLFSQCQAIHCRAILPCQDTPSVKLTYTAEVSVPKELVALMSAIRDGETP
DPEDPSRKIYKFIQKVPIPCYLIALVVGALESRQIGPRTLVWSEKEQVEKSAYEFSETES
MLKIAEDLGGPYVWGQYDLLVLPPSFPYGGMENPCLTFVTPTLLAGDKSLSNVIAHEISH
SWTGNLVTNKTWDHFWLNEGHTVYLERHICGRLFGEKFRHFNALGGWGELQNSVKTFGET
HPFTKLVVDLTDIDPDVAYSSVPYEKGFALLFYLEQLLGGPEIFLGFLKAYVEKFSYKSI
TTDDWKDFLYSYFKDKVDVLNQVDWNAWLYSPGLPPIKPNYDMTLTNACIALSQRWITAK
EDDLNSFNATDLKDLSSHQLNEFLAQTLQRAPLPLGHIKRMQEVYNFNAINNSEIRFRWL
RLCIQSKWEDAIPLALKMATEQGRMKFTRPLFKDLAAFDKSHDQAVRTYQEHKASMHPVT
AMLVGKDLKVD
|
|
|
|---|
| BDBM50359080 |
|---|
| n/a |
|---|
| Name | BDBM50359080 |
|---|
| Synonyms: | CHEMBL1922660 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C38H43N3O4S |
|---|
| Mol. Mass. | 637.831 |
|---|
| SMILES | CCOc1ccc(cn1)-c1ccc(Cn2c(CC(C)(C)C(O)=O)c(SC(C)(C)C)c3cc(OCc4ccc(C)cn4)ccc23)cc1 |
|---|
| Structure | 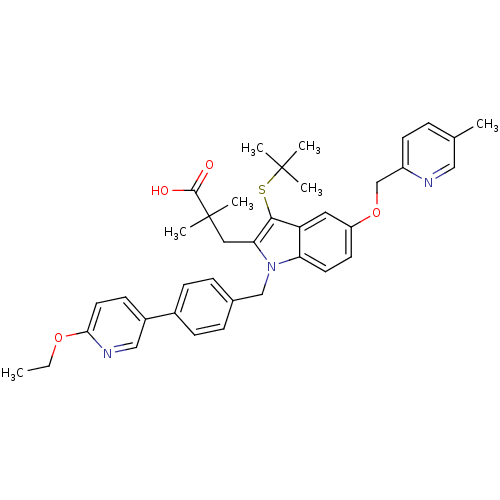
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Stock, NS; Bain, G; Zunic, J; Li, Y; Ziff, J; Roppe, J; Santini, A; Darlington, J; Prodanovich, P; King, CD; Baccei, C; Lee, C; Rong, H; Chapman, C; Broadhead, A; Lorrain, D; Correa, L; Hutchinson, JH; Evans, JF; Prasit, P 5-Lipoxygenase-activating protein (FLAP) inhibitors. Part 4: development of 3-[3-tert-butylsulfanyl-1-[4-(6-ethoxypyridin-3-yl)benzyl]-5-(5-methylpyridin-2-ylmethoxy)-1H-indol-2-yl]-2,2-dimethylpropionic acid (AM803), a potent, oral, once daily FLAP inhibitor. J Med Chem54:8013-29 (2011) [PubMed] Article
Stock, NS; Bain, G; Zunic, J; Li, Y; Ziff, J; Roppe, J; Santini, A; Darlington, J; Prodanovich, P; King, CD; Baccei, C; Lee, C; Rong, H; Chapman, C; Broadhead, A; Lorrain, D; Correa, L; Hutchinson, JH; Evans, JF; Prasit, P 5-Lipoxygenase-activating protein (FLAP) inhibitors. Part 4: development of 3-[3-tert-butylsulfanyl-1-[4-(6-ethoxypyridin-3-yl)benzyl]-5-(5-methylpyridin-2-ylmethoxy)-1H-indol-2-yl]-2,2-dimethylpropionic acid (AM803), a potent, oral, once daily FLAP inhibitor. J Med Chem54:8013-29 (2011) [PubMed] Article