| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Platelet-activating factor receptor |
|---|
| Ligand | BDBM50004633 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_155148 (CHEMBL760189) |
|---|
| IC50 | 4.3±n/a nM |
|---|
| Citation |  Fray, MJ; Bull, DJ; Cooper, K; Parry, MJ; Stefaniak, MH Novel antagonists of platelet-activating factor. 2. Synthesis and structure-activity relationships of potent and long-acting heterofused [1,5]benzodiazepine and [1,4]diazepine derivatives of 2-methyl-1-phenylimidazo[4,5-c]pyridine. J Med Chem38:3524-35 (1995) [PubMed] Fray, MJ; Bull, DJ; Cooper, K; Parry, MJ; Stefaniak, MH Novel antagonists of platelet-activating factor. 2. Synthesis and structure-activity relationships of potent and long-acting heterofused [1,5]benzodiazepine and [1,4]diazepine derivatives of 2-methyl-1-phenylimidazo[4,5-c]pyridine. J Med Chem38:3524-35 (1995) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Platelet-activating factor receptor |
|---|
| Name: | Platelet-activating factor receptor |
|---|
| Synonyms: | PAF-R | PTAFR | PTAFR_CAVPO | Platelet activating factor receptor | Platelet-activating factor receptor |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 39005.63 |
|---|
| Organism: | Cavia porcellus |
|---|
| Description: | n/a |
|---|
| Residue: | 342 |
|---|
| Sequence: | MELNSSSRVDSEFRYTLFPIVYSIIFVLGIIANGYVLWVFARLYPSKKLNEIKIFMVNLT
VADLLFLITLPLWIVYYSNQGNWFLPKFLCNLAGCLFFINTYCSVAFLGVITYNRFQAVK
YPIKTAQATTRKRGIALSLVIWVAIVAAASYFLVMDSTNVVSNKAGSGNITRCFEHYEKG
SKPVLIIHICIVLGFFIVFLLILFCNLVIIHTLLRQPVKQQRNAEVRRRALWMVCTVLAV
FVICFVPHHMVQLPWTLAELGMWPSSNHQAINDAHQVTLCLLSTNCVLDPVIYCFLTKKF
RKHLSEKLNIMRSSQKCSRVTTDTGTEMAIPINHTPVNPIKN
|
|
|
|---|
| BDBM50004633 |
|---|
| n/a |
|---|
| Name | BDBM50004633 |
|---|
| Synonyms: | (S)-4-(2-Chloro-phenyl)-6-methyl-2-[4-(2-methyl-imidazo[4,5-c]pyridin-1-yl)-phenyl]-5-(pyridin-2-ylcarbamoyl)-1,4-dihydro-pyridine-3-carboxylic acid ethyl ester | 4-(2-Chloro-phenyl)-6-methyl-2-[4-(2-methyl-imidazo[4,5-c]pyridin-1-yl)-phenyl]-5-(pyridin-2-ylcarbamoyl)-1,4-dihydro-pyridine-3-carboxylic acid ethyl ester | 4-(2-Chloro-phenyl)-6-methyl-2-[4-(2-methyl-imidazo[4,5-c]pyridin-1-yl)-phenyl]-5-(pyridin-2-ylcarbamoyl)-nicotinic acid ethyl ester | CHEMBL29067 | UK-74505 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C34H29ClN6O3 |
|---|
| Mol. Mass. | 605.085 |
|---|
| SMILES | CCOC(=O)C1=C(N=C(C)C(C1c1ccccc1Cl)C(=O)Nc1ccccn1)c1ccc(cc1)-n1c(C)nc2cnccc12 |t:5,7| |
|---|
| Structure | 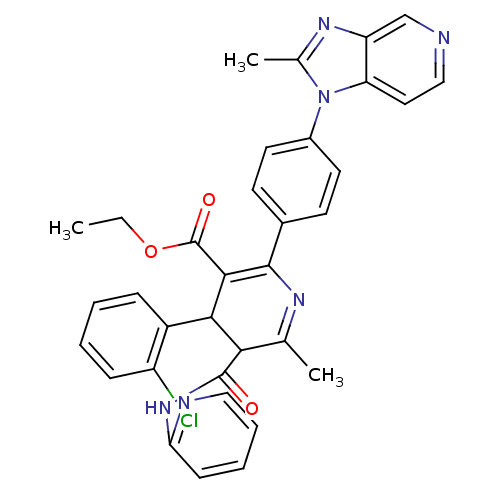
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Fray, MJ; Bull, DJ; Cooper, K; Parry, MJ; Stefaniak, MH Novel antagonists of platelet-activating factor. 2. Synthesis and structure-activity relationships of potent and long-acting heterofused [1,5]benzodiazepine and [1,4]diazepine derivatives of 2-methyl-1-phenylimidazo[4,5-c]pyridine. J Med Chem38:3524-35 (1995) [PubMed]
Fray, MJ; Bull, DJ; Cooper, K; Parry, MJ; Stefaniak, MH Novel antagonists of platelet-activating factor. 2. Synthesis and structure-activity relationships of potent and long-acting heterofused [1,5]benzodiazepine and [1,4]diazepine derivatives of 2-methyl-1-phenylimidazo[4,5-c]pyridine. J Med Chem38:3524-35 (1995) [PubMed]