| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Neprilysin |
|---|
| Ligand | BDBM50083382 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_144468 (CHEMBL755533) |
|---|
| Ki | 5.4±n/a nM |
|---|
| Citation |  David, C; Bischoff, L; Meudal, H; Mothé, A; De Mota, N; DaNascimento, S; Llorens-Cortes, C; Fournié-Zaluski, MC; Roques, BP Investigation of subsite preferences in aminopeptidase A (EC 3.4.11.7) led to the design of the first highly potent and selective inhibitors of this enzyme. J Med Chem42:5197-211 (2000) [PubMed] David, C; Bischoff, L; Meudal, H; Mothé, A; De Mota, N; DaNascimento, S; Llorens-Cortes, C; Fournié-Zaluski, MC; Roques, BP Investigation of subsite preferences in aminopeptidase A (EC 3.4.11.7) led to the design of the first highly potent and selective inhibitors of this enzyme. J Med Chem42:5197-211 (2000) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Neprilysin |
|---|
| Name: | Neprilysin |
|---|
| Synonyms: | Atriopeptidase | CD10 antigen | CD_antigen=CD10 | Enkephalinase | MME | NEP | NEP_RABIT | Neutral Endopeptidase | Neutral endopeptidase 24.11 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 85570.75 |
|---|
| Organism: | Oryctolagus cuniculus (rabbit) |
|---|
| Description: | NEP was purified to homogeneity from rabbit kidney. |
|---|
| Residue: | 750 |
|---|
| Sequence: | MGRSESQMDITDINTPKPKKKQRWTPLEISLSVLVLLLTVIAVTMIALYATYDDGICKSS
DCIKSAARLIQNMDATAEPCTDFFKYACGGWLKRNVIPETSSRYSNFDILRDELEVILKD
VLQEPKTEDIVAVQKAKTLYRSCVNETAIDSRGGQPLLKLLPDVYGWPVATQNWEQTYGT
SWSAEKSIAQLNSNYGKKVLINFFVGTDDKNSMNHIIHIDQPRLGLPSRDYYECTGIYKE
ACTAYVDFMIAVAKLIRQEEGLPIDENQISVEMNKVMELEKEIANATTKSEDRNDPMLLY
NKMTLAQIQNNFSLEINGKPFSWSNFTNEIMSTVNINIPNEEDVVVYAPEYLIKLKPILT
KYFPRDFQNLFSWRFIMDLVSSLSRTYKDSRNAFRKALYGTTSESATWRRCANYVNGNME
NAVGRLYVEAAFAGESKHVVEDLIAQIREVFIQTLDDLTWMDAETKKKAEEKALAIKERI
GYPDDIVSNDNKLNNEYLELNYKEDEYFENIIQNLKFSQSKQLKKLREKVDKDEWITGAA
IVNAFYSSGRNQIVFPAGILQPPFFSAQQSNSLNYGGIGMVIGHEITHGFDDNGRNFNKD
GDLVDWWTQQSANNFKEQSQCMVYQYGNFSWDLAGGQHLNGINTLGENIADNGGIGQAYR
AYQNYVKKNGEEKLLPGIDLNHKQLFFLNFAQVWCGTYRPEYAVNSIKTDVHSPGNFRII
GSLQNSVEFSEAFQCPKNSYMNPEKKCRVW
|
|
|
|---|
| BDBM50083382 |
|---|
| n/a |
|---|
| Name | BDBM50083382 |
|---|
| Synonyms: | 1-{[1-(1,2-Dicarboxy-ethylcarbamoyl)-2-methyl-butylcarbamoyl]-mercapto-methyl}-3-phosphono-propyl-ammonium | CHEMBL356858 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C15H28N3O9PS |
|---|
| Mol. Mass. | 457.436 |
|---|
| SMILES | CC[C@@H](C)[C@H](NC(=O)[C@H](S)[C@H]([NH3+])CCP(O)([O-])=O)C(=O)N[C@@H](CC(O)=O)C(O)=O |
|---|
| Structure | 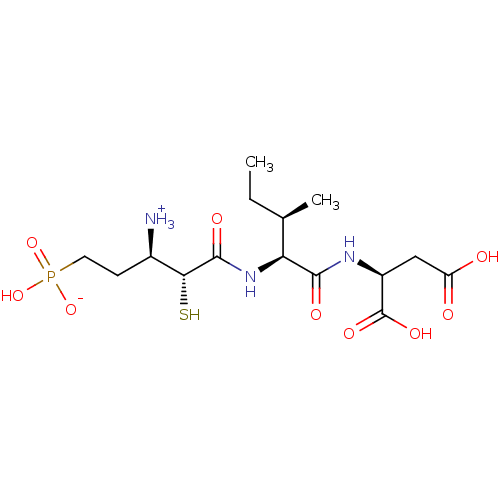
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 David, C; Bischoff, L; Meudal, H; Mothé, A; De Mota, N; DaNascimento, S; Llorens-Cortes, C; Fournié-Zaluski, MC; Roques, BP Investigation of subsite preferences in aminopeptidase A (EC 3.4.11.7) led to the design of the first highly potent and selective inhibitors of this enzyme. J Med Chem42:5197-211 (2000) [PubMed]
David, C; Bischoff, L; Meudal, H; Mothé, A; De Mota, N; DaNascimento, S; Llorens-Cortes, C; Fournié-Zaluski, MC; Roques, BP Investigation of subsite preferences in aminopeptidase A (EC 3.4.11.7) led to the design of the first highly potent and selective inhibitors of this enzyme. J Med Chem42:5197-211 (2000) [PubMed]