| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Delta-type opioid receptor |
|---|
| Ligand | BDBM50216132 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_439881 (CHEMBL890202) |
|---|
| Ki | 7620±n/a nM |
|---|
| Citation |  Simpson, DS; Katavic, PL; Lozama, A; Harding, WW; Parrish, D; Deschamps, JR; Dersch, CM; Partilla, JS; Rothman, RB; Navarro, H; Prisinzano, TE Synthetic studies of neoclerodane diterpenes from Salvia divinorum: preparation and opioid receptor activity of salvinicin analogues. J Med Chem50:3596-603 (2007) [PubMed] Article Simpson, DS; Katavic, PL; Lozama, A; Harding, WW; Parrish, D; Deschamps, JR; Dersch, CM; Partilla, JS; Rothman, RB; Navarro, H; Prisinzano, TE Synthetic studies of neoclerodane diterpenes from Salvia divinorum: preparation and opioid receptor activity of salvinicin analogues. J Med Chem50:3596-603 (2007) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Delta-type opioid receptor |
|---|
| Name: | Delta-type opioid receptor |
|---|
| Synonyms: | D-OR-1 | DOR-1 | Delta opioid receptor | Delta-type opioid receptor (Delta) | OPIATE Delta | OPRD | OPRD1 | OPRD_HUMAN | OPRK1 | opioid receptor, delta 1 |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 40382.98 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Competition binding assays were carried out using membrane preparations from transfected HN9.10 cells that constitutively expressed the delta opioid receptor. |
|---|
| Residue: | 372 |
|---|
| Sequence: | MEPAPSAGAELQPPLFANASDAYPSACPSAGANASGPPGARSASSLALAIAITALYSAVC
AVGLLGNVLVMFGIVRYTKMKTATNIYIFNLALADALATSTLPFQSAKYLMETWPFGELL
CKAVLSIDYYNMFTSIFTLTMMSVDRYIAVCHPVKALDFRTPAKAKLINICIWVLASGVG
VPIMVMAVTRPRDGAVVCMLQFPSPSWYWDTVTKICVFLFAFVVPILIITVCYGLMLLRL
RSVRLLSGSKEKDRSLRRITRMVLVVVGAFVVCWAPIHIFVIVWTLVDIDRRDPLVVAAL
HLCIALGYANSSLNPVLYAFLDENFKRCFRQLCRKPCGRPDPSSFSRAREATARERVTAC
TPSDGPGGGAAA
|
|
|
|---|
| BDBM50216132 |
|---|
| n/a |
|---|
| Name | BDBM50216132 |
|---|
| Synonyms: | (2S,4aR,6aR,7R,9S,10aS,10bR)-methyl 9-acetoxy-2-(2-bromofuran-3-yl)-6a,10b-dimethyl-4,10-dioxo-dodecahydro-1H-benzo[f]isochromene-7-carboxylate | (2S,4aR,6aR,7R,9S,10aS,10bR)-methyl 9-acetoxy-2-(2-bromofuran-3-yl)-6a,10b-dimethyl-4,10-dioxododecahydro-1H-benzo[f]isochromene-7-carboxylate | CHEMBL390935 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H27BrO8 |
|---|
| Mol. Mass. | 511.36 |
|---|
| SMILES | COC(=O)[C@@H]1C[C@H](OC(C)=O)C(=O)[C@H]2[C@@]1(C)CC[C@H]1C(=O)O[C@@H](C[C@]21C)c1ccoc1Br |r| |
|---|
| Structure | 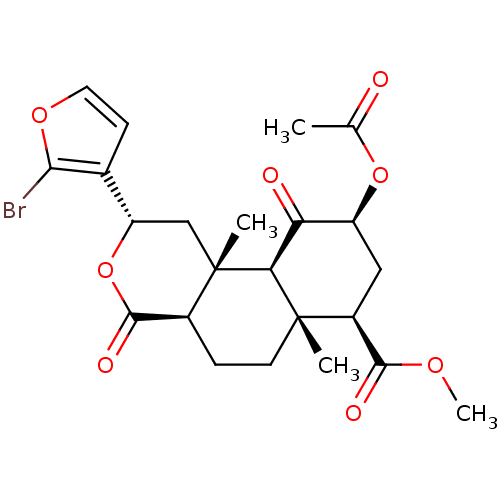
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Simpson, DS; Katavic, PL; Lozama, A; Harding, WW; Parrish, D; Deschamps, JR; Dersch, CM; Partilla, JS; Rothman, RB; Navarro, H; Prisinzano, TE Synthetic studies of neoclerodane diterpenes from Salvia divinorum: preparation and opioid receptor activity of salvinicin analogues. J Med Chem50:3596-603 (2007) [PubMed] Article
Simpson, DS; Katavic, PL; Lozama, A; Harding, WW; Parrish, D; Deschamps, JR; Dersch, CM; Partilla, JS; Rothman, RB; Navarro, H; Prisinzano, TE Synthetic studies of neoclerodane diterpenes from Salvia divinorum: preparation and opioid receptor activity of salvinicin analogues. J Med Chem50:3596-603 (2007) [PubMed] Article