| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prothrombin |
|---|
| Ligand | BDBM50377619 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_479759 (CHEMBL934691) |
|---|
| Ki | 0.79±n/a nM |
|---|
| Citation |  Kreutter, KD; Lu, T; Lee, L; Giardino, EC; Patel, S; Huang, H; Xu, G; Fitzgerald, M; Haertlein, BJ; Mohan, V; Crysler, C; Eisennagel, S; Dasgupta, M; McMillan, M; Spurlino, JC; Huebert, ND; Maryanoff, BE; Tomczuk, BE; Damiano, BP; Player, MR Orally efficacious thrombin inhibitors with cyanofluorophenylacetamide as the P2 motif. Bioorg Med Chem Lett18:2865-70 (2008) [PubMed] Article Kreutter, KD; Lu, T; Lee, L; Giardino, EC; Patel, S; Huang, H; Xu, G; Fitzgerald, M; Haertlein, BJ; Mohan, V; Crysler, C; Eisennagel, S; Dasgupta, M; McMillan, M; Spurlino, JC; Huebert, ND; Maryanoff, BE; Tomczuk, BE; Damiano, BP; Player, MR Orally efficacious thrombin inhibitors with cyanofluorophenylacetamide as the P2 motif. Bioorg Med Chem Lett18:2865-70 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prothrombin |
|---|
| Name: | Prothrombin |
|---|
| Synonyms: | Activation peptide fragment 1 | Activation peptide fragment 2 | Coagulation factor II | F2 | Prothrombin precursor | THRB_HUMAN | Thrombin heavy chain | Thrombin light chain |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 70029.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P00734 |
|---|
| Residue: | 622 |
|---|
| Sequence: | MAHVRGLQLPGCLALAALCSLVHSQHVFLAPQQARSLLQRVRRANTFLEEVRKGNLEREC
VEETCSYEEAFEALESSTATDVFWAKYTACETARTPRDKLAACLEGNCAEGLGTNYRGHV
NITRSGIECQLWRSRYPHKPEINSTTHPGADLQENFCRNPDSSTTGPWCYTTDPTVRRQE
CSIPVCGQDQVTVAMTPRSEGSSVNLSPPLEQCVPDRGQQYQGRLAVTTHGLPCLAWASA
QAKALSKHQDFNSAVQLVENFCRNPDGDEEGVWCYVAGKPGDFGYCDLNYCEEAVEEETG
DGLDEDSDRAIEGRTATSEYQTFFNPRTFGSGEADCGLRPLFEKKSLEDKTERELLESYI
DGRIVEGSDAEIGMSPWQVMLFRKSPQELLCGASLISDRWVLTAAHCLLYPPWDKNFTEN
DLLVRIGKHSRTRYERNIEKISMLEKIYIHPRYNWRENLDRDIALMKLKKPVAFSDYIHP
VCLPDRETAASLLQAGYKGRVTGWGNLKETWTANVGKGQPSVLQVVNLPIVERPVCKDST
RIRITDNMFCAGYKPDEGKRGDACEGDSGGPFVMKSPFNNRWYQMGIVSWGEGCDRDGKY
GFYTHVFRLKKWIQKVIDQFGE
|
|
|
|---|
| BDBM50377619 |
|---|
| n/a |
|---|
| Name | BDBM50377619 |
|---|
| Synonyms: | CHEMBL402758 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H22F3N7O2 |
|---|
| Mol. Mass. | 449.4296 |
|---|
| SMILES | [#6]-c1cccc(n1)C(F)(F)[#6]-[#7]-c1ccc(C#N)c(-[#6]-[#6](=O)-[#7]-[#6]-[#6]-[#8]\[#7]=[#6](\[#7])-[#7])c1F |
|---|
| Structure | 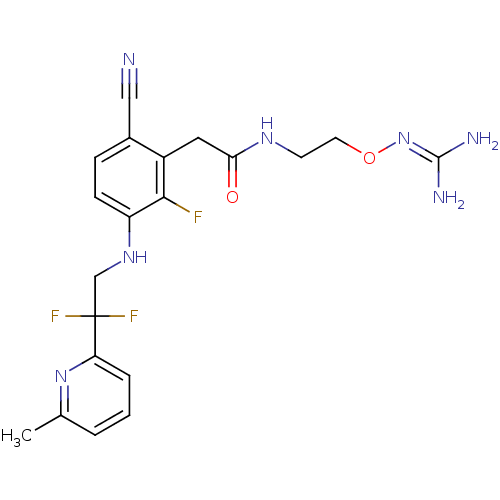
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Kreutter, KD; Lu, T; Lee, L; Giardino, EC; Patel, S; Huang, H; Xu, G; Fitzgerald, M; Haertlein, BJ; Mohan, V; Crysler, C; Eisennagel, S; Dasgupta, M; McMillan, M; Spurlino, JC; Huebert, ND; Maryanoff, BE; Tomczuk, BE; Damiano, BP; Player, MR Orally efficacious thrombin inhibitors with cyanofluorophenylacetamide as the P2 motif. Bioorg Med Chem Lett18:2865-70 (2008) [PubMed] Article
Kreutter, KD; Lu, T; Lee, L; Giardino, EC; Patel, S; Huang, H; Xu, G; Fitzgerald, M; Haertlein, BJ; Mohan, V; Crysler, C; Eisennagel, S; Dasgupta, M; McMillan, M; Spurlino, JC; Huebert, ND; Maryanoff, BE; Tomczuk, BE; Damiano, BP; Player, MR Orally efficacious thrombin inhibitors with cyanofluorophenylacetamide as the P2 motif. Bioorg Med Chem Lett18:2865-70 (2008) [PubMed] Article