| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Mitogen-activated protein kinase 14 |
|---|
| Ligand | BDBM50310986 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_618897 (CHEMBL1101788) |
|---|
| IC50 | 55±n/a nM |
|---|
| Citation |  Selness, SR; Devraj, RV; Monahan, JB; Boehm, TL; Walker, JK; Devadas, B; Durley, RC; Kurumbail, R; Shieh, H; Xing, L; Hepperle, M; Rucker, PV; Jerome, KD; Benson, AG; Marrufo, LD; Madsen, HM; Hitchcock, J; Owen, TJ; Christie, L; Promo, MA; Hickory, BS; Alvira, E; Naing, W; Blevis-Bal, R Discovery of N-substituted pyridinones as potent and selective inhibitors of p38 kinase. Bioorg Med Chem Lett19:5851-6 (2009) [PubMed] Article Selness, SR; Devraj, RV; Monahan, JB; Boehm, TL; Walker, JK; Devadas, B; Durley, RC; Kurumbail, R; Shieh, H; Xing, L; Hepperle, M; Rucker, PV; Jerome, KD; Benson, AG; Marrufo, LD; Madsen, HM; Hitchcock, J; Owen, TJ; Christie, L; Promo, MA; Hickory, BS; Alvira, E; Naing, W; Blevis-Bal, R Discovery of N-substituted pyridinones as potent and selective inhibitors of p38 kinase. Bioorg Med Chem Lett19:5851-6 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Mitogen-activated protein kinase 14 |
|---|
| Name: | Mitogen-activated protein kinase 14 |
|---|
| Synonyms: | CSAID-binding protein | CSBP | CSBP1 | CSBP2 | CSPB1 | Cytokine suppressive anti-inflammatory drug-binding protein | MAP kinase 14 | MAP kinase MXI2 | MAP kinase p38 alpha | MAPK 14 | MAPK14 | MAX-interacting protein 2 | MK14_HUMAN | MXI2 | Mitogen-activated protein kinase p38 alpha | SAPK2A | Stress-activated protein kinase 2a | p38 MAP kinase alpha/beta |
|---|
| Type: | Serine/threonine-protein kinase |
|---|
| Mol. Mass.: | 41286.76 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q16539 |
|---|
| Residue: | 360 |
|---|
| Sequence: | MSQERPTFYRQELNKTIWEVPERYQNLSPVGSGAYGSVCAAFDTKTGLRVAVKKLSRPFQ
SIIHAKRTYRELRLLKHMKHENVIGLLDVFTPARSLEEFNDVYLVTHLMGADLNNIVKCQ
KLTDDHVQFLIYQILRGLKYIHSADIIHRDLKPSNLAVNEDCELKILDFGLARHTDDEMT
GYVATRWYRAPEIMLNWMHYNQTVDIWSVGCIMAELLTGRTLFPGTDHIDQLKLILRLVG
TPGAELLKKISSESARNYIQSLTQMPKMNFANVFIGANPLAVDLLEKMLVLDSDKRITAA
QALAHAYFAQYHDPDDEPVADPYDQSFESRDLLIDEWKSLTYDEVISFVPPPLDQEEMES
|
|
|
|---|
| BDBM50310986 |
|---|
| n/a |
|---|
| Name | BDBM50310986 |
|---|
| Synonyms: | CHEMBL1078667 | N-(4-((3-chloro-4-(2,4-difluorobenzyloxy)-6-methyl-2-oxopyridin-1(2H)-yl)methyl)benzyl)-2-hydroxyacetamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H21ClF2N2O4 |
|---|
| Mol. Mass. | 462.874 |
|---|
| SMILES | Cc1cc(OCc2ccc(F)cc2F)c(Cl)c(=O)n1Cc1ccc(CNC(=O)CO)cc1 |
|---|
| Structure | 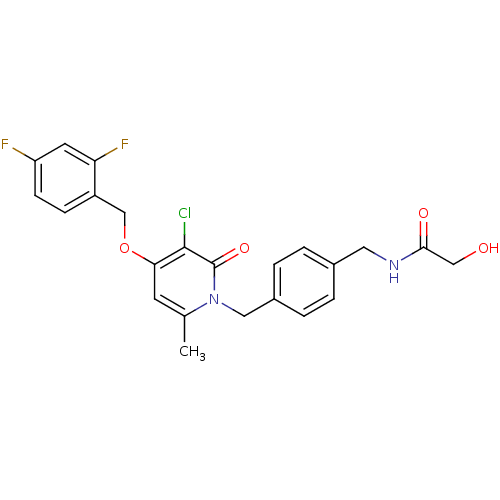
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Selness, SR; Devraj, RV; Monahan, JB; Boehm, TL; Walker, JK; Devadas, B; Durley, RC; Kurumbail, R; Shieh, H; Xing, L; Hepperle, M; Rucker, PV; Jerome, KD; Benson, AG; Marrufo, LD; Madsen, HM; Hitchcock, J; Owen, TJ; Christie, L; Promo, MA; Hickory, BS; Alvira, E; Naing, W; Blevis-Bal, R Discovery of N-substituted pyridinones as potent and selective inhibitors of p38 kinase. Bioorg Med Chem Lett19:5851-6 (2009) [PubMed] Article
Selness, SR; Devraj, RV; Monahan, JB; Boehm, TL; Walker, JK; Devadas, B; Durley, RC; Kurumbail, R; Shieh, H; Xing, L; Hepperle, M; Rucker, PV; Jerome, KD; Benson, AG; Marrufo, LD; Madsen, HM; Hitchcock, J; Owen, TJ; Christie, L; Promo, MA; Hickory, BS; Alvira, E; Naing, W; Blevis-Bal, R Discovery of N-substituted pyridinones as potent and selective inhibitors of p38 kinase. Bioorg Med Chem Lett19:5851-6 (2009) [PubMed] Article