| Reaction Details |
|---|
 | Report a problem with these data |
| Target | C-C chemokine receptor type 5 |
|---|
| Ligand | BDBM50115528 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_630265 (CHEMBL1106474) |
|---|
| IC50 | 6±n/a nM |
|---|
| Citation |  Rotstein, DM; Melville, CR; Padilla, F; Cournoyer, D; Lee, EK; Lemoine, R; Petersen, AC; Setti, LQ; Wanner, J; Chen, L; Filonova, L; Loughhead, DG; Manka, J; Lin, XF; Gleason, S; Sankuratri, S; Ji, C; Derosier, A; Dioszegi, M; Heilek, G; Jekle, A; Berry, P; Mau, CI; Weller, P Novel hexahydropyrrolo[3,4-c]pyrrole CCR5 antagonists. Bioorg Med Chem Lett20:3116-9 (2010) [PubMed] Article Rotstein, DM; Melville, CR; Padilla, F; Cournoyer, D; Lee, EK; Lemoine, R; Petersen, AC; Setti, LQ; Wanner, J; Chen, L; Filonova, L; Loughhead, DG; Manka, J; Lin, XF; Gleason, S; Sankuratri, S; Ji, C; Derosier, A; Dioszegi, M; Heilek, G; Jekle, A; Berry, P; Mau, CI; Weller, P Novel hexahydropyrrolo[3,4-c]pyrrole CCR5 antagonists. Bioorg Med Chem Lett20:3116-9 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| C-C chemokine receptor type 5 |
|---|
| Name: | C-C chemokine receptor type 5 |
|---|
| Synonyms: | C-C CKR-5 | C-C chemokine receptor type 5 | CC-CKR-5 | CCR-5 | CCR5 | CCR5/mu opioid receptor complex | CCR5_HUMAN | CD_antigen=CD195 | CHEMR13 | CMKBR5 | HIV-1 fusion coreceptor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 40540.21 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P51681 |
|---|
| Residue: | 352 |
|---|
| Sequence: | MDYQVSSPIYDINYYTSEPCQKINVKQIAARLLPPLYSLVFIFGFVGNMLVILILINCKR
LKSMTDIYLLNLAISDLFFLLTVPFWAHYAAAQWDFGNTMCQLLTGLYFIGFFSGIFFII
LLTIDRYLAVVHAVFALKARTVTFGVVTSVITWVVAVFASLPGIIFTRSQKEGLHYTCSS
HFPYSQYQFWKNFQTLKIVILGLVLPLLVMVICYSGILKTLLRCRNEKKRHRAVRLIFTI
MIVYFLFWAPYNIVLLLNTFQEFFGLNNCSSSNRLDQAMQVTETLGMTHCCINPIIYAFV
GEKFRNYLLVFFQKHIAKRFCKCCSIFQQEAPERASSVYTRSTGEQEISVGL
|
|
|
|---|
| BDBM50115528 |
|---|
| n/a |
|---|
| Name | BDBM50115528 |
|---|
| Synonyms: | (Z)-(4-((4-bromophenyl)(ethoxyimino)methyl)-4'-methyl-1,4'-bipiperidin-1'-yl)(2,6-dimethylphenyl)methanone | CHEMBL324643 | {4-[(4-Bromo-phenyl)-ethoxyimino-methyl]-4'-methyl-[1,4']bipiperidinyl-1'-yl}-(2,6-dimethyl-phenyl)-methanone |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C29H38BrN3O2 |
|---|
| Mol. Mass. | 540.535 |
|---|
| SMILES | CCO\N=C(\C1CCN(CC1)C1(C)CCN(CC1)C(=O)c1c(C)cccc1C)c1ccc(Br)cc1 |
|---|
| Structure | 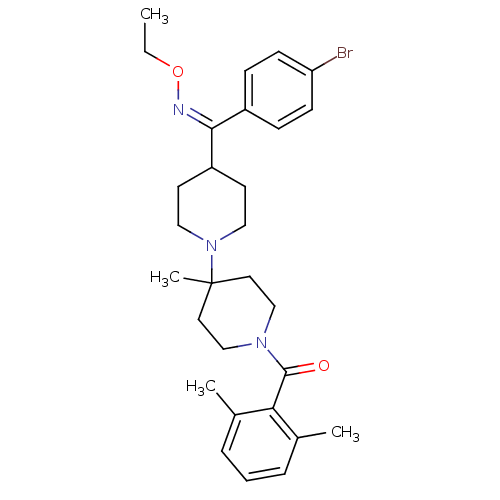
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Rotstein, DM; Melville, CR; Padilla, F; Cournoyer, D; Lee, EK; Lemoine, R; Petersen, AC; Setti, LQ; Wanner, J; Chen, L; Filonova, L; Loughhead, DG; Manka, J; Lin, XF; Gleason, S; Sankuratri, S; Ji, C; Derosier, A; Dioszegi, M; Heilek, G; Jekle, A; Berry, P; Mau, CI; Weller, P Novel hexahydropyrrolo[3,4-c]pyrrole CCR5 antagonists. Bioorg Med Chem Lett20:3116-9 (2010) [PubMed] Article
Rotstein, DM; Melville, CR; Padilla, F; Cournoyer, D; Lee, EK; Lemoine, R; Petersen, AC; Setti, LQ; Wanner, J; Chen, L; Filonova, L; Loughhead, DG; Manka, J; Lin, XF; Gleason, S; Sankuratri, S; Ji, C; Derosier, A; Dioszegi, M; Heilek, G; Jekle, A; Berry, P; Mau, CI; Weller, P Novel hexahydropyrrolo[3,4-c]pyrrole CCR5 antagonists. Bioorg Med Chem Lett20:3116-9 (2010) [PubMed] Article