

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Macrophage colony-stimulating factor 1 receptor | ||
| Ligand | BDBM50321372 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_640283 (CHEMBL1174608) | ||
| IC50 | 2.5±n/a nM | ||
| Citation |  Wilson, KJ; Illig, CR; Chen, J; Wall, MJ; Ballentine, SK; DesJarlais, RL; Chen, Y; Schubert, C; Donatelli, R; Petrounia, I; Crysler, CS; Molloy, CJ; Chaikin, MA; Manthey, CL; Player, MR; Tomczuk, BE; Meegalla, SK Reducing ion channel activity in a series of 4-heterocyclic arylamide FMS inhibitors. Bioorg Med Chem Lett20:3925-9 (2010) [PubMed] Article Wilson, KJ; Illig, CR; Chen, J; Wall, MJ; Ballentine, SK; DesJarlais, RL; Chen, Y; Schubert, C; Donatelli, R; Petrounia, I; Crysler, CS; Molloy, CJ; Chaikin, MA; Manthey, CL; Player, MR; Tomczuk, BE; Meegalla, SK Reducing ion channel activity in a series of 4-heterocyclic arylamide FMS inhibitors. Bioorg Med Chem Lett20:3925-9 (2010) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Macrophage colony-stimulating factor 1 receptor | |||
| Name: | Macrophage colony-stimulating factor 1 receptor | ||
| Synonyms: | CSF1R | CSF1R_HUMAN | FMS | Fms proto-oncogene | Macrophage colony stimulating factor receptor | Macrophage colony-stimulating factor 1 receptor (FMS) | Macrophage colony-stimulating factor 1 receptor (c-FMS) | Macrophage colony-stimulating factor 1 receptor (cFMS) | Proto-oncogene c-Fms | Tyrosine Kinase CSF1-R | c-fms | cFMS | ||
| Type: | Protein | ||
| Mol. Mass.: | 107979.48 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P07333 | ||
| Residue: | 972 | ||
| Sequence: |
| ||
| BDBM50321372 | |||
| n/a | |||
| Name | BDBM50321372 | ||
| Synonyms: | 4-cyano-N-(4-(2-(cyanoimino)hexahydropyrimidin-5-yl)-2-(4,4-dimethylcyclohex-1-enyl)phenyl)-1H-imidazole-2-carboxamide | CHEMBL1170997 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C24H26N8O | ||
| Mol. Mass. | 442.5162 | ||
| SMILES | CC1(C)CCC(=CC1)c1cc(ccc1NC(=O)c1ncc([nH]1)C#N)C1CNC(NC1)=NC#N |c:5,(11.18,-14.71,;11.96,-16.05,;12.71,-14.71,;10.62,-16.82,;10.63,-18.36,;11.95,-19.12,;13.29,-18.35,;13.29,-16.82,;11.95,-20.65,;10.63,-21.42,;10.62,-22.96,;11.96,-23.73,;13.29,-22.96,;13.29,-21.42,;14.6,-20.61,;15.94,-21.38,;15.94,-22.91,;17.26,-20.6,;18.67,-21.23,;19.7,-20.08,;18.92,-18.75,;17.42,-19.08,;19.53,-17.35,;20.15,-15.93,;9.3,-23.73,;7.97,-22.95,;6.62,-23.7,;6.63,-25.24,;7.96,-26.02,;9.3,-25.26,;5.3,-26.01,;3.96,-25.24,;2.63,-24.47,)| | ||
| Structure | 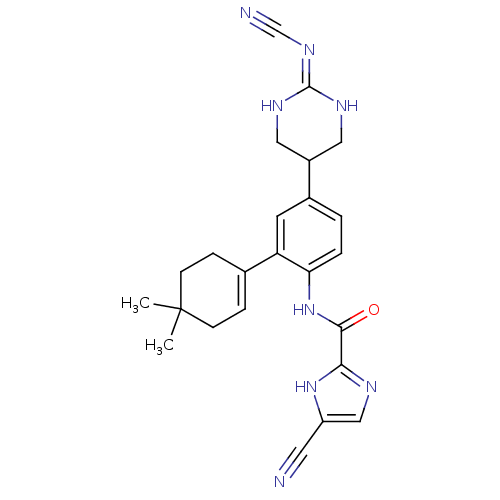
| ||