| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Sphingosine 1-phosphate receptor 1 |
|---|
| Ligand | BDBM50419963 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_809729 (CHEMBL2016148) |
|---|
| EC50 | 1.26±n/a nM |
|---|
| Citation |  Xu, H; Zhang, H; Luan, L; Xu, Y; Li, C; Wang, Y; Han, F; Yang, T; Ren, F; Xiang, JN; Elliott, JD; Zhao, Y; Guo, TB; Lu, H; Zhang, W; Hirst, D; Lindon, M; Lin, X Discovery of thiadiazole amides as potent, S1P3-sparing agonists of sphingosine-1-phosphate 1 (S1P1) receptor. Bioorg Med Chem Lett22:2456-9 (2012) [PubMed] Article Xu, H; Zhang, H; Luan, L; Xu, Y; Li, C; Wang, Y; Han, F; Yang, T; Ren, F; Xiang, JN; Elliott, JD; Zhao, Y; Guo, TB; Lu, H; Zhang, W; Hirst, D; Lindon, M; Lin, X Discovery of thiadiazole amides as potent, S1P3-sparing agonists of sphingosine-1-phosphate 1 (S1P1) receptor. Bioorg Med Chem Lett22:2456-9 (2012) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Sphingosine 1-phosphate receptor 1 |
|---|
| Name: | Sphingosine 1-phosphate receptor 1 |
|---|
| Synonyms: | CHEDG1 | EDG1 | Endothelial differentiation G-protein coupled receptor 1 | S1P receptor | S1P1 | S1PR1 | S1PR1_HUMAN | Sphingosine 1-phosphate receptor 1 (S1PR1) | Sphingosine 1-phosphate receptor Edg-1 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 42836.02 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P21453 |
|---|
| Residue: | 382 |
|---|
| Sequence: | MGPTSVPLVKAHRSSVSDYVNYDIIVRHYNYTGKLNISADKENSIKLTSVVFILICCFII
LENIFVLLTIWKTKKFHRPMYYFIGNLALSDLLAGVAYTANLLLSGATTYKLTPAQWFLR
EGSMFVALSASVFSLLAIAIERYITMLKMKLHNGSNNFRLFLLISACWVISLILGGLPIM
GWNCISALSSCSTVLPLYHKHYILFCTTVFTLLLLSIVILYCRIYSLVRTRSRRLTFRKN
ISKASRSSEKSLALLKTVIIVLSVFIACWAPLFILLLLDVGCKVKTCDILFRAEYFLVLA
VLNSGTNPIIYTLTNKEMRRAFIRIMSCCKCPSGDSAGKFKRPIIAGMEFSRSKSDNSSH
PQKDEGDNPETIMSSGNVNSSS
|
|
|
|---|
| BDBM50419963 |
|---|
| n/a |
|---|
| Name | BDBM50419963 |
|---|
| Synonyms: | CHEMBL2011736 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H23F3N4O5S2 |
|---|
| Mol. Mass. | 556.578 |
|---|
| SMILES | CCCCN(C(=O)c1ccc(cc1)C(F)(F)F)c1nnc(s1)-c1ccc(cc1)S(=O)(=O)NCCC(O)=O |
|---|
| Structure | 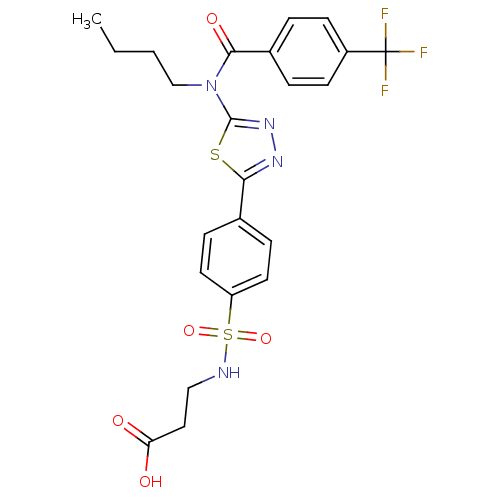
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Xu, H; Zhang, H; Luan, L; Xu, Y; Li, C; Wang, Y; Han, F; Yang, T; Ren, F; Xiang, JN; Elliott, JD; Zhao, Y; Guo, TB; Lu, H; Zhang, W; Hirst, D; Lindon, M; Lin, X Discovery of thiadiazole amides as potent, S1P3-sparing agonists of sphingosine-1-phosphate 1 (S1P1) receptor. Bioorg Med Chem Lett22:2456-9 (2012) [PubMed] Article
Xu, H; Zhang, H; Luan, L; Xu, Y; Li, C; Wang, Y; Han, F; Yang, T; Ren, F; Xiang, JN; Elliott, JD; Zhao, Y; Guo, TB; Lu, H; Zhang, W; Hirst, D; Lindon, M; Lin, X Discovery of thiadiazole amides as potent, S1P3-sparing agonists of sphingosine-1-phosphate 1 (S1P1) receptor. Bioorg Med Chem Lett22:2456-9 (2012) [PubMed] Article