

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Tyrosine-protein kinase receptor TYRO3 | ||
| Ligand | BDBM50384584 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_820661 (CHEMBL2037932) | ||
| IC50 | 5.9±n/a nM | ||
| Citation |  Liu, J; Yang, C; Simpson, C; Deryckere, D; Van Deusen, A; Miley, MJ; Kireev, D; Norris-Drouin, J; Sather, S; Hunter, D; Korboukh, VK; Patel, HS; Janzen, WP; Machius, M; Johnson, GL; Earp, HS; Graham, DK; Frye, SV; Wang, X Discovery of Novel Small Molecule Mer Kinase Inhibitors for the Treatment of Pediatric Acute Lymphoblastic Leukemia. ACS Med Chem Lett3:129-134 (2012) [PubMed] Article Liu, J; Yang, C; Simpson, C; Deryckere, D; Van Deusen, A; Miley, MJ; Kireev, D; Norris-Drouin, J; Sather, S; Hunter, D; Korboukh, VK; Patel, HS; Janzen, WP; Machius, M; Johnson, GL; Earp, HS; Graham, DK; Frye, SV; Wang, X Discovery of Novel Small Molecule Mer Kinase Inhibitors for the Treatment of Pediatric Acute Lymphoblastic Leukemia. ACS Med Chem Lett3:129-134 (2012) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Tyrosine-protein kinase receptor TYRO3 | |||
| Name: | Tyrosine-protein kinase receptor TYRO3 | ||
| Synonyms: | BYK | DTK | RSE | SKY | TIF | TYRO3 | TYRO3_HUMAN | Tyrosine-protein kinase SKY | ||
| Type: | Protein | ||
| Mol. Mass.: | 96894.13 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | Q06418 | ||
| Residue: | 890 | ||
| Sequence: |
| ||
| BDBM50384584 | |||
| n/a | |||
| Name | BDBM50384584 | ||
| Synonyms: | CHEMBL2036807 | US9744172, Compound UNC607A | ||
| Type | Small organic molecule | ||
| Emp. Form. | C31H40N8 | ||
| Mol. Mass. | 524.7029 | ||
| SMILES | N[C@H]1CC[C@H](Cn2nc(-c3ccc(cc3)N3CCNCC3)c3cnc(NCCCc4ccccc4)nc23)CC1 |r,wU:4.4,wD:1.0,(41.81,-28.1,;42.85,-26.96,;44.35,-27.28,;45.39,-26.14,;44.91,-24.68,;45.94,-23.54,;45.47,-22.07,;46.38,-20.82,;45.47,-19.56,;45.94,-18.1,;47.45,-17.78,;47.93,-16.32,;46.9,-15.17,;45.38,-15.5,;44.91,-16.96,;47.36,-13.71,;48.87,-13.39,;49.35,-11.94,;48.32,-10.79,;46.81,-11.11,;46.33,-12.57,;43.99,-20.04,;42.66,-19.28,;41.33,-20.05,;41.33,-21.6,;39.99,-22.36,;38.66,-21.59,;38.66,-20.05,;37.33,-19.28,;37.33,-17.74,;38.67,-16.98,;38.67,-15.44,;37.34,-14.67,;36,-15.45,;36,-16.98,;42.66,-22.37,;43.99,-21.6,;43.41,-24.35,;42.38,-25.49,)| | ||
| Structure | 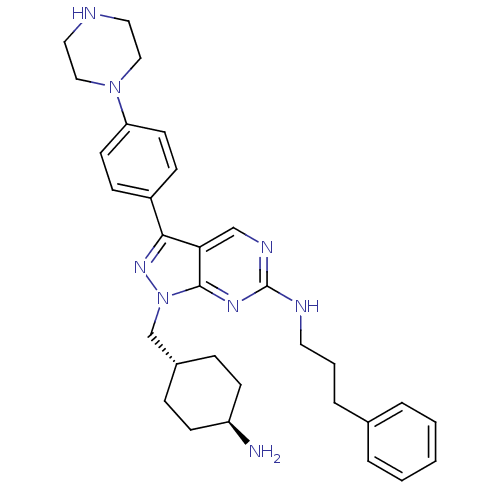
| ||