| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Tyrosine-protein kinase SYK |
|---|
| Ligand | BDBM50386749 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_827005 (CHEMBL2050069) |
|---|
| IC50 | >10000±n/a nM |
|---|
| Citation |  Li, WW; Wang, XY; Zheng, RL; Yan, HX; Cao, ZX; Zhong, L; Wang, ZR; Ji, P; Yang, LL; Wang, LJ; Xu, Y; Liu, JJ; Yang, J; Zhang, CH; Ma, S; Feng, S; Sun, QZ; Wei, YQ; Yang, SY Discovery of the novel potent and selective FLT3 inhibitor 1-{5-[7-(3- morpholinopropoxy)quinazolin-4-ylthio]-[1,3,4]thiadiazol-2-yl}-3-p-tolylurea and its anti-acute myeloid leukemia (AML) activities in vitro and in vivo. J Med Chem55:3852-66 (2012) [PubMed] Article Li, WW; Wang, XY; Zheng, RL; Yan, HX; Cao, ZX; Zhong, L; Wang, ZR; Ji, P; Yang, LL; Wang, LJ; Xu, Y; Liu, JJ; Yang, J; Zhang, CH; Ma, S; Feng, S; Sun, QZ; Wei, YQ; Yang, SY Discovery of the novel potent and selective FLT3 inhibitor 1-{5-[7-(3- morpholinopropoxy)quinazolin-4-ylthio]-[1,3,4]thiadiazol-2-yl}-3-p-tolylurea and its anti-acute myeloid leukemia (AML) activities in vitro and in vivo. J Med Chem55:3852-66 (2012) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Tyrosine-protein kinase SYK |
|---|
| Name: | Tyrosine-protein kinase SYK |
|---|
| Synonyms: | KSYK_HUMAN | SYK | Spleen Tyrosine Kinase | Spleen tyrosine kinase (SYK) | Tyrosine-protein kinase SYK (Syk) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 72079.99 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P43405 |
|---|
| Residue: | 635 |
|---|
| Sequence: | MASSGMADSANHLPFFFGNITREEAEDYLVQGGMSDGLYLLRQSRNYLGGFALSVAHGRK
AHHYTIERELNGTYAIAGGRTHASPADLCHYHSQESDGLVCLLKKPFNRPQGVQPKTGPF
EDLKENLIREYVKQTWNLQGQALEQAIISQKPQLEKLIATTAHEKMPWFHGKISREESEQ
IVLIGSKTNGKFLIRARDNNGSYALCLLHEGKVLHYRIDKDKTGKLSIPEGKKFDTLWQL
VEHYSYKADGLLRVLTVPCQKIGTQGNVNFGGRPQLPGSHPATWSAGGIISRIKSYSFPK
PGHRKSSPAQGNRQESTVSFNPYEPELAPWAADKGPQREALPMDTEVYESPYADPEEIRP
KEVYLDRKLLTLEDKELGSGNFGTVKKGYYQMKKVVKTVAVKILKNEANDPALKDELLAE
ANVMQQLDNPYIVRMIGICEAESWMLVMEMAELGPLNKYLQQNRHVKDKNIIELVHQVSM
GMKYLEESNFVHRDLAARNVLLVTQHYAKISDFGLSKALRADENYYKAQTHGKWPVKWYA
PECINYYKFSSKSDVWSFGVLMWEAFSYGQKPYRGMKGSEVTAMLEKGERMGCPAGCPRE
MYDLMNLCWTYDVENRPGFAAVELRLRNYYYDVVN
|
|
|
|---|
| BDBM50386749 |
|---|
| n/a |
|---|
| Name | BDBM50386749 |
|---|
| Synonyms: | CHEMBL2046883 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H27N7O3S2 |
|---|
| Mol. Mass. | 537.657 |
|---|
| SMILES | Cc1ccc(NC(=O)Nc2nnc(Sc3ncnc4cc(OCCCN5CCOCC5)ccc34)s2)cc1 |
|---|
| Structure | 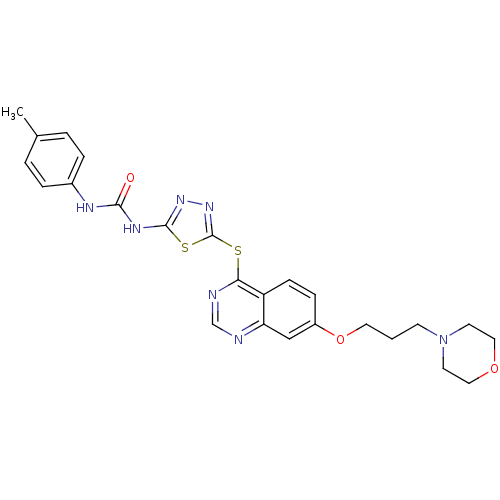
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Li, WW; Wang, XY; Zheng, RL; Yan, HX; Cao, ZX; Zhong, L; Wang, ZR; Ji, P; Yang, LL; Wang, LJ; Xu, Y; Liu, JJ; Yang, J; Zhang, CH; Ma, S; Feng, S; Sun, QZ; Wei, YQ; Yang, SY Discovery of the novel potent and selective FLT3 inhibitor 1-{5-[7-(3- morpholinopropoxy)quinazolin-4-ylthio]-[1,3,4]thiadiazol-2-yl}-3-p-tolylurea and its anti-acute myeloid leukemia (AML) activities in vitro and in vivo. J Med Chem55:3852-66 (2012) [PubMed] Article
Li, WW; Wang, XY; Zheng, RL; Yan, HX; Cao, ZX; Zhong, L; Wang, ZR; Ji, P; Yang, LL; Wang, LJ; Xu, Y; Liu, JJ; Yang, J; Zhang, CH; Ma, S; Feng, S; Sun, QZ; Wei, YQ; Yang, SY Discovery of the novel potent and selective FLT3 inhibitor 1-{5-[7-(3- morpholinopropoxy)quinazolin-4-ylthio]-[1,3,4]thiadiazol-2-yl}-3-p-tolylurea and its anti-acute myeloid leukemia (AML) activities in vitro and in vivo. J Med Chem55:3852-66 (2012) [PubMed] Article