| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2C8 |
|---|
| Ligand | BDBM50397798 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_874592 (CHEMBL2186228) |
|---|
| IC50 | 1000±n/a nM |
|---|
| Citation |  Ashwell, MA; Lapierre, JM; Brassard, C; Bresciano, K; Bull, C; Cornell-Kennon, S; Eathiraj, S; France, DS; Hall, T; Hill, J; Kelleher, E; Khanapurkar, S; Kizer, D; Koerner, S; Link, J; Liu, Y; Makhija, S; Moussa, M; Namdev, N; Nguyen, K; Nicewonger, R; Palma, R; Szwaya, J; Tandon, M; Uppalapati, U; Vensel, D; Volak, LP; Volckova, E; Westlund, N; Wu, H; Yang, RY; Chan, TC Discovery and optimization of a series of 3-(3-phenyl-3H-imidazo[4,5-b]pyridin-2-yl)pyridin-2-amines: orally bioavailable, selective, and potent ATP-independent Akt inhibitors. J Med Chem55:5291-310 (2012) [PubMed] Article Ashwell, MA; Lapierre, JM; Brassard, C; Bresciano, K; Bull, C; Cornell-Kennon, S; Eathiraj, S; France, DS; Hall, T; Hill, J; Kelleher, E; Khanapurkar, S; Kizer, D; Koerner, S; Link, J; Liu, Y; Makhija, S; Moussa, M; Namdev, N; Nguyen, K; Nicewonger, R; Palma, R; Szwaya, J; Tandon, M; Uppalapati, U; Vensel, D; Volak, LP; Volckova, E; Westlund, N; Wu, H; Yang, RY; Chan, TC Discovery and optimization of a series of 3-(3-phenyl-3H-imidazo[4,5-b]pyridin-2-yl)pyridin-2-amines: orally bioavailable, selective, and potent ATP-independent Akt inhibitors. J Med Chem55:5291-310 (2012) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2C8 |
|---|
| Name: | Cytochrome P450 2C8 |
|---|
| Synonyms: | CP2C8_HUMAN | CYP2C8 | CYPIIC8 | Cytochrome P450 2C8 (CYP2C8) | P450 IIC2 | P450 MP-12/MP-20 | P450 form 1 | S-mephenytoin 4-hydroxylase |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 55839.23 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P10632 |
|---|
| Residue: | 490 |
|---|
| Sequence: | MEPFVVLVLCLSFMLLFSLWRQSCRRRKLPPGPTPLPIIGNMLQIDVKDICKSFTNFSKV
YGPVFTVYFGMNPIVVFHGYEAVKEALIDNGEEFSGRGNSPISQRITKGLGIISSNGKRW
KEIRRFSLTTLRNFGMGKRSIEDRVQEEAHCLVEELRKTKASPCDPTFILGCAPCNVICS
VVFQKRFDYKDQNFLTLMKRFNENFRILNSPWIQVCNNFPLLIDCFPGTHNKVLKNVALT
RSYIREKVKEHQASLDVNNPRDFIDCFLIKMEQEKDNQKSEFNIENLVGTVADLFVAGTE
TTSTTLRYGLLLLLKHPEVTAKVQEEIDHVIGRHRSPCMQDRSHMPYTDAVVHEIQRYSD
LVPTGVPHAVTTDTKFRNYLIPKGTTIMALLTSVLHDDKEFPNPNIFDPGHFLDKNGNFK
KSDYFMPFSAGKRICAGEGLARMELFLFLTTILQNFNLKSVDDLKNLNTTAVTKGIVSLP
PSYQICFIPV
|
|
|
|---|
| BDBM50397798 |
|---|
| n/a |
|---|
| Name | BDBM50397798 |
|---|
| Synonyms: | CHEMBL2177836 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C33H26FN7O2 |
|---|
| Mol. Mass. | 571.6036 |
|---|
| SMILES | CC(=O)Nc1cccc(c1)-c1ccc2nc(-c3cccnc3N)n(-c3ccc(CNC(=O)c4cccc(F)c4)cc3)c2n1 |
|---|
| Structure | 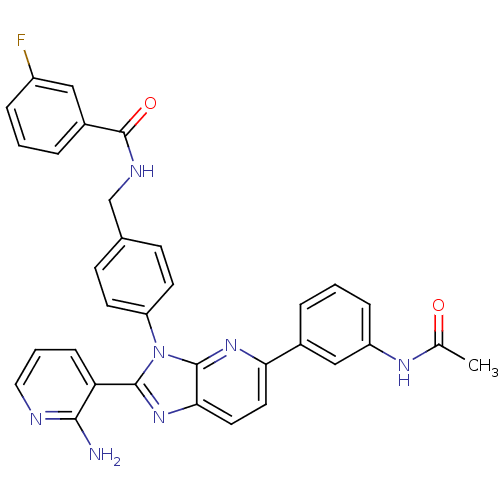
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Ashwell, MA; Lapierre, JM; Brassard, C; Bresciano, K; Bull, C; Cornell-Kennon, S; Eathiraj, S; France, DS; Hall, T; Hill, J; Kelleher, E; Khanapurkar, S; Kizer, D; Koerner, S; Link, J; Liu, Y; Makhija, S; Moussa, M; Namdev, N; Nguyen, K; Nicewonger, R; Palma, R; Szwaya, J; Tandon, M; Uppalapati, U; Vensel, D; Volak, LP; Volckova, E; Westlund, N; Wu, H; Yang, RY; Chan, TC Discovery and optimization of a series of 3-(3-phenyl-3H-imidazo[4,5-b]pyridin-2-yl)pyridin-2-amines: orally bioavailable, selective, and potent ATP-independent Akt inhibitors. J Med Chem55:5291-310 (2012) [PubMed] Article
Ashwell, MA; Lapierre, JM; Brassard, C; Bresciano, K; Bull, C; Cornell-Kennon, S; Eathiraj, S; France, DS; Hall, T; Hill, J; Kelleher, E; Khanapurkar, S; Kizer, D; Koerner, S; Link, J; Liu, Y; Makhija, S; Moussa, M; Namdev, N; Nguyen, K; Nicewonger, R; Palma, R; Szwaya, J; Tandon, M; Uppalapati, U; Vensel, D; Volak, LP; Volckova, E; Westlund, N; Wu, H; Yang, RY; Chan, TC Discovery and optimization of a series of 3-(3-phenyl-3H-imidazo[4,5-b]pyridin-2-yl)pyridin-2-amines: orally bioavailable, selective, and potent ATP-independent Akt inhibitors. J Med Chem55:5291-310 (2012) [PubMed] Article