

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Cytochrome P450 2C19 | ||
| Ligand | BDBM98282 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_939151 (CHEMBL2329223) | ||
| IC50 | 16300±n/a nM | ||
| Citation |  Jin, M; Wang, J; Kleinberg, A; Kadalbajoo, M; Siu, KW; Cooke, A; Bittner, MA; Yao, Y; Thelemann, A; Ji, Q; Bhagwat, S; Mulvihill, KM; Rechka, JA; Pachter, JA; Crew, AP; Epstein, D; Mulvihill, MJ Discovery of potent, selective and orally bioavailable imidazo[1,5-a]pyrazine derived ACK1 inhibitors. Bioorg Med Chem Lett23:979-84 (2013) [PubMed] Article Jin, M; Wang, J; Kleinberg, A; Kadalbajoo, M; Siu, KW; Cooke, A; Bittner, MA; Yao, Y; Thelemann, A; Ji, Q; Bhagwat, S; Mulvihill, KM; Rechka, JA; Pachter, JA; Crew, AP; Epstein, D; Mulvihill, MJ Discovery of potent, selective and orally bioavailable imidazo[1,5-a]pyrazine derived ACK1 inhibitors. Bioorg Med Chem Lett23:979-84 (2013) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Cytochrome P450 2C19 | |||
| Name: | Cytochrome P450 2C19 | ||
| Synonyms: | (R)-limonene 6-monooxygenase | (S)-limonene 6-monooxygenase | (S)-limonene 7-monooxygenase | CP2CJ_HUMAN | CYP2C19 | CYPIIC17 | CYPIIC19 | Cytochrome P450 2C19 (CYP2C19) | Cytochrome P450 2C19 [I331V] | Cytochrome P450-11A | Cytochrome P450-254C | Fenbendazole monooxygenase (4'-hydroxylating) | Mephenytoin 4-hydroxylase | P450-11A | P450-254C | ||
| Type: | Enzyme | ||
| Mol. Mass.: | 55935.47 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P33261 | ||
| Residue: | 490 | ||
| Sequence: |
| ||
| BDBM98282 | |||
| n/a | |||
| Name | BDBM98282 | ||
| Synonyms: | US8481733, 94 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C24H24N4O3 | ||
| Mol. Mass. | 416.4724 | ||
| SMILES | COc1cc(Oc2ccccc2)ccc1-c1nc([C@H]2C[C@@](C)(O)C2)n2ccnc(N)c12 |r,wU:18.19,20.23,wD:20.22,(2.01,-1.15,;2.01,.39,;.92,1.48,;1.32,2.96,;.23,4.05,;.63,5.54,;2.12,5.94,;3.21,4.85,;4.7,5.25,;5.09,6.74,;4.01,7.83,;2.52,7.43,;-1.25,3.65,;-1.65,2.17,;-.56,1.08,;-.96,-.41,;-.06,-1.65,;-.96,-2.9,;-.56,-4.39,;.77,-5.16,;-0,-6.49,;1.33,-7.26,;-.77,-7.83,;-1.33,-5.72,;-2.43,-2.42,;-3.76,-3.19,;-5.09,-2.42,;-5.09,-.88,;-3.76,-.11,;-3.76,1.43,;-2.43,-.88,)| | ||
| Structure | 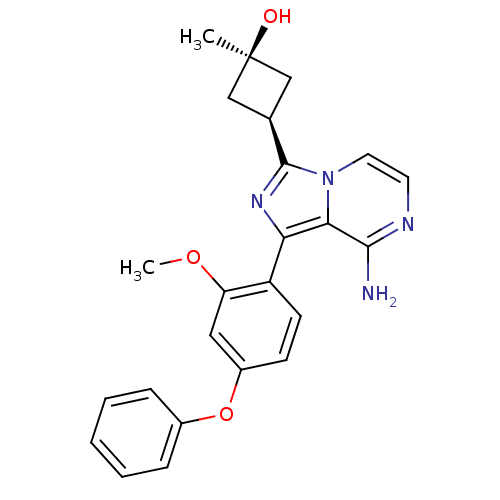
| ||