| Reaction Details |
|---|
 | Report a problem with these data |
| Target | E3 ubiquitin-protein ligase Mdm2 |
|---|
| Ligand | BDBM50397778 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_951640 (CHEMBL2351554) |
|---|
| IC50 | 360±n/a nM |
|---|
| Citation |  Gonzalez-Lopez de Turiso, F; Sun, D; Rew, Y; Bartberger, MD; Beck, HP; Canon, J; Chen, A; Chow, D; Correll, TL; Huang, X; Julian, LD; Kayser, F; Lo, MC; Long, AM; McMinn, D; Oliner, JD; Osgood, T; Powers, JP; Saiki, AY; Schneider, S; Shaffer, P; Xiao, SH; Yakowec, P; Yan, X; Ye, Q; Yu, D; Zhao, X; Zhou, J; Medina, JC; Olson, SH Rational design and binding mode duality of MDM2-p53 inhibitors. J Med Chem56:4053-70 (2013) [PubMed] Article Gonzalez-Lopez de Turiso, F; Sun, D; Rew, Y; Bartberger, MD; Beck, HP; Canon, J; Chen, A; Chow, D; Correll, TL; Huang, X; Julian, LD; Kayser, F; Lo, MC; Long, AM; McMinn, D; Oliner, JD; Osgood, T; Powers, JP; Saiki, AY; Schneider, S; Shaffer, P; Xiao, SH; Yakowec, P; Yan, X; Ye, Q; Yu, D; Zhao, X; Zhou, J; Medina, JC; Olson, SH Rational design and binding mode duality of MDM2-p53 inhibitors. J Med Chem56:4053-70 (2013) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| E3 ubiquitin-protein ligase Mdm2 |
|---|
| Name: | E3 ubiquitin-protein ligase Mdm2 |
|---|
| Synonyms: | Double minute 2 protein | Double minute 2 protein (HDM2) | E3 ubiquitin-protein ligase Mdm2 (p53-binding protein Mdm2) | Hdm2 | Human Double Minute 2 (HDM2) | MDM2 | MDM2-MDMX | MDM2_HUMAN | p53-Binding Protein MDM2 | p53-binding protein |
|---|
| Type: | Oncoprotein |
|---|
| Mol. Mass.: | 55196.54 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q00987 |
|---|
| Residue: | 491 |
|---|
| Sequence: | MCNTNMSVPTDGAVTTSQIPASEQETLVRPKPLLLKLLKSVGAQKDTYTMKEVLFYLGQY
IMTKRLYDEKQQHIVYCSNDLLGDLFGVPSFSVKEHRKIYTMIYRNLVVVNQQESSDSGT
SVSENRCHLEGGSDQKDLVQELQEEKPSSSHLVSRPSTSSRRRAISETEENSDELSGERQ
RKRHKSDSISLSFDESLALCVIREICCERSSSSESTGTPSNPDLDAGVSEHSGDWLDQDS
VSDQFSVEFEVESLDSEDYSLSEEGQELSDEDDEVYQVTVYQAGESDTDSFEEDPEISLA
DYWKCTSCNEMNPPLPSHCNRCWALRENWLPEDKGKDKGEISEKAKLENSTQAEEGFDVP
DCKKTIVNDSRESCVEENDDKITQASQSQESEDYSQPSTSSSIIYSSQEDVKEFEREETQ
DKEESVESSLPLNAIEPCVICQGRPKNGCIVHGKTGHLMACFTCAKKLKKRNKPCPVCRQ
PIQMIVLTYFP
|
|
|
|---|
| BDBM50397778 |
|---|
| n/a |
|---|
| Name | BDBM50397778 |
|---|
| Synonyms: | CHEMBL2177813 | US9593129, Example 35 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H23Cl2NO3 |
|---|
| Mol. Mass. | 432.34 |
|---|
| SMILES | OC(=O)C[C@H]1C[C@@H]([C@H](N(CC2CC2)C1=O)c1ccc(Cl)cc1)c1cccc(Cl)c1 |r| |
|---|
| Structure | 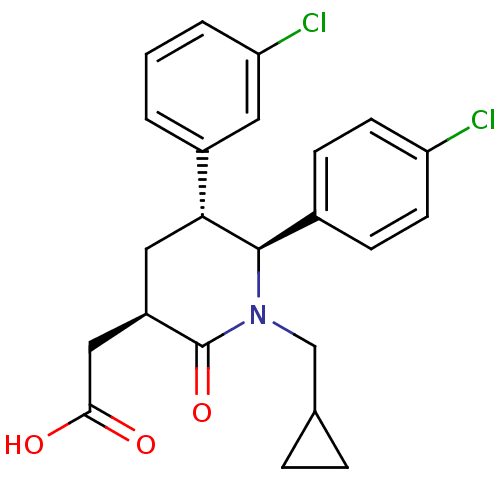
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Gonzalez-Lopez de Turiso, F; Sun, D; Rew, Y; Bartberger, MD; Beck, HP; Canon, J; Chen, A; Chow, D; Correll, TL; Huang, X; Julian, LD; Kayser, F; Lo, MC; Long, AM; McMinn, D; Oliner, JD; Osgood, T; Powers, JP; Saiki, AY; Schneider, S; Shaffer, P; Xiao, SH; Yakowec, P; Yan, X; Ye, Q; Yu, D; Zhao, X; Zhou, J; Medina, JC; Olson, SH Rational design and binding mode duality of MDM2-p53 inhibitors. J Med Chem56:4053-70 (2013) [PubMed] Article
Gonzalez-Lopez de Turiso, F; Sun, D; Rew, Y; Bartberger, MD; Beck, HP; Canon, J; Chen, A; Chow, D; Correll, TL; Huang, X; Julian, LD; Kayser, F; Lo, MC; Long, AM; McMinn, D; Oliner, JD; Osgood, T; Powers, JP; Saiki, AY; Schneider, S; Shaffer, P; Xiao, SH; Yakowec, P; Yan, X; Ye, Q; Yu, D; Zhao, X; Zhou, J; Medina, JC; Olson, SH Rational design and binding mode duality of MDM2-p53 inhibitors. J Med Chem56:4053-70 (2013) [PubMed] Article