| Reaction Details |
|---|
 | Report a problem with these data |
| Target | D(1A) dopamine receptor |
|---|
| Ligand | BDBM50004822 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1353324 (CHEMBL3267826) |
|---|
| Ki | 73±n/a nM |
|---|
| Citation |  Liu, Z; Ai, J; Peng, X; Song, Z; Wu, K; Zhang, J; Yao, Q; Chen, Y; Ji, Y; Yang, Y; Geng, M; Zhang, A Novel 2,4-Diarylaminopyrimidine Analogues (DAAPalogues) Showing Potent c-Met/ALK Multikinase Inhibitory Activities. ACS Med Chem Lett5:304-8 (2014) [PubMed] Article Liu, Z; Ai, J; Peng, X; Song, Z; Wu, K; Zhang, J; Yao, Q; Chen, Y; Ji, Y; Yang, Y; Geng, M; Zhang, A Novel 2,4-Diarylaminopyrimidine Analogues (DAAPalogues) Showing Potent c-Met/ALK Multikinase Inhibitory Activities. ACS Med Chem Lett5:304-8 (2014) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| D(1A) dopamine receptor |
|---|
| Name: | D(1A) dopamine receptor |
|---|
| Synonyms: | DA D1 receptor | DOPAMINE D1 | DRD1 | DRD1_HUMAN | Dopamine D1 receptor | Dopamine D1 receptor (D1R) | Dopamine D1A |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 49303.43 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P21728 |
|---|
| Residue: | 446 |
|---|
| Sequence: | MRTLNTSAMDGTGLVVERDFSVRILTACFLSLLILSTLLGNTLVCAAVIRFRHLRSKVTN
FFVISLAVSDLLVAVLVMPWKAVAEIAGFWPFGSFCNIWVAFDIMCSTASILNLCVISVD
RYWAISSPFRYERKMTPKAAFILISVAWTLSVLISFIPVQLSWHKAKPTSPSDGNATSLA
ETIDNCDSSLSRTYAISSSVISFYIPVAIMIVTYTRIYRIAQKQIRRIAALERAAVHAKN
CQTTTGNGKPVECSQPESSFKMSFKRETKVLKTLSVIMGVFVCCWLPFFILNCILPFCGS
GETQPFCIDSNTFDVFVWFGWANSSLNPIIYAFNADFRKAFSTLLGCYRLCPATNNAIET
VSINNNGAAMFSSHHEPRGSISKECNLVYLIPHAVGSSEDLKKEEAAGIARPLEKLSPAL
SVILDYDTDVSLEKIQPITQNGQHPT
|
|
|
|---|
| BDBM50004822 |
|---|
| n/a |
|---|
| Name | BDBM50004822 |
|---|
| Synonyms: | (R)-1-phenyl-2,3,4,5-tetrahydro-1H-benzo[d]azepine-7,8-diol | (R)-7,8-Dihydroxy-1-phenyl-2,3,4,5-tetrahydro-1H-benzo[d]azepinium | 1-Phenyl-2,3,4,5-tetrahydro-1H-benzo[d]azepine-7,8-diol | 1-Phenyl-2,3,4,5-tetrahydro-1H-benzo[d]azepine-7,8-diol(SKF38393) | 7,8-Dihydroxy-1-phenyl-2,3,4,5-tetrahydro-1H-benzo[d]azepinium(R(+)SKF38393) | CHEMBL1467585 | CHEMBL24077 | R(+)SKF 383931-Phenyl-2,3,4,5-tetrahydro-1H-benzo[d]azepine-7,8-diol | SK&F-383933 | SK-38393 | SK-383933 | SKF 38393 | SKF 38393 (+) |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C16H17NO2 |
|---|
| Mol. Mass. | 255.3117 |
|---|
| SMILES | Oc1cc2CCNC[C@H](c3ccccc3)c2cc1O |
|---|
| Structure | 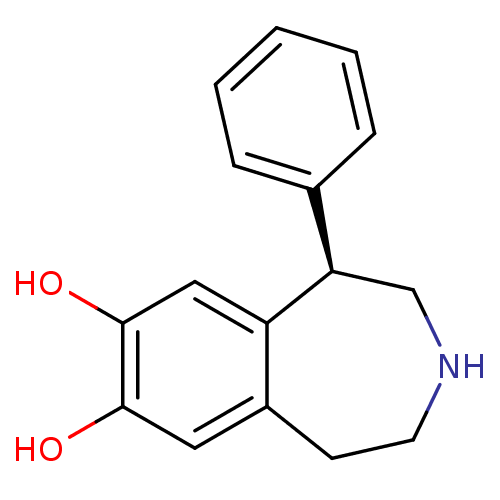
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Liu, Z; Ai, J; Peng, X; Song, Z; Wu, K; Zhang, J; Yao, Q; Chen, Y; Ji, Y; Yang, Y; Geng, M; Zhang, A Novel 2,4-Diarylaminopyrimidine Analogues (DAAPalogues) Showing Potent c-Met/ALK Multikinase Inhibitory Activities. ACS Med Chem Lett5:304-8 (2014) [PubMed] Article
Liu, Z; Ai, J; Peng, X; Song, Z; Wu, K; Zhang, J; Yao, Q; Chen, Y; Ji, Y; Yang, Y; Geng, M; Zhang, A Novel 2,4-Diarylaminopyrimidine Analogues (DAAPalogues) Showing Potent c-Met/ALK Multikinase Inhibitory Activities. ACS Med Chem Lett5:304-8 (2014) [PubMed] Article