| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Histone-lysine N-methyltransferase EZH2 |
|---|
| Ligand | BDBM50110364 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1511672 (CHEMBL3607425) |
|---|
| IC50 | 76±n/a nM |
|---|
| Citation |  Gehling, VS; Vaswani, RG; Nasveschuk, CG; Duplessis, M; Iyer, P; Balasubramanian, S; Zhao, F; Good, AC; Campbell, R; Lee, C; Dakin, LA; Cook, AS; Gagnon, A; Harmange, JC; Audia, JE; Cummings, RT; Normant, E; Trojer, P; Albrecht, BK Discovery, design, and synthesis of indole-based EZH2 inhibitors. Bioorg Med Chem Lett25:3644-9 (2015) [PubMed] Article Gehling, VS; Vaswani, RG; Nasveschuk, CG; Duplessis, M; Iyer, P; Balasubramanian, S; Zhao, F; Good, AC; Campbell, R; Lee, C; Dakin, LA; Cook, AS; Gagnon, A; Harmange, JC; Audia, JE; Cummings, RT; Normant, E; Trojer, P; Albrecht, BK Discovery, design, and synthesis of indole-based EZH2 inhibitors. Bioorg Med Chem Lett25:3644-9 (2015) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Histone-lysine N-methyltransferase EZH2 |
|---|
| Name: | Histone-lysine N-methyltransferase EZH2 |
|---|
| Synonyms: | ENX-1 | EZH2 | EZH2_HUMAN | Enhancer of zeste homolog 2 (EZH2) | Histone-lysine N-methyltransferase EZH2 | KMT6 | Lysine N-methyltransferase 6 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 85367.84 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q15910 |
|---|
| Residue: | 746 |
|---|
| Sequence: | MGQTGKKSEKGPVCWRKRVKSEYMRLRQLKRFRRADEVKSMFSSNRQKILERTEILNQEW
KQRRIQPVHILTSVSSLRGTRECSVTSDLDFPTQVIPLKTLNAVASVPIMYSWSPLQQNF
MVEDETVLHNIPYMGDEVLDQDGTFIEELIKNYDGKVHGDRECGFINDEIFVELVNALGQ
YNDDDDDDDGDDPEEREEKQKDLEDHRDDKESRPPRKFPSDKIFEAISSMFPDKGTAEEL
KEKYKELTEQQLPGALPPECTPNIDGPNAKSVQREQSLHSFHTLFCRRCFKYDCFLHPFH
ATPNTYKRKNTETALDNKPCGPQCYQHLEGAKEFAAALTAERIKTPPKRPGGRRRGRLPN
NSSRPSTPTINVLESKDTDSDREAGTETGGENNDKEEEEKKDETSSSSEANSRCQTPIKM
KPNIEPPENVEWSGAEASMFRVLIGTYYDNFCAIARLIGTKTCRQVYEFRVKESSIIAPA
PAEDVDTPPRKKKRKHRLWAAHCRKIQLKKDGSSNHVYNYQPCDHPRQPCDSSCPCVIAQ
NFCEKFCQCSSECQNRFPGCRCKAQCNTKQCPCYLAVRECDPDLCLTCGAADHWDSKNVS
CKNCSIQRGSKKHLLLAPSDVAGWGIFIKDPVQKNEFISEYCGEIISQDEADRRGKVYDK
YMCSFLFNLNNDFVVDATRKGNKIRFANHSVNPNCYAKVMMVNGDHRIGIFAKRAIQTGE
ELFFDYRYSQADALKYVGIEREMEIP
|
|
|
|---|
| BDBM50110364 |
|---|
| n/a |
|---|
| Name | BDBM50110364 |
|---|
| Synonyms: | CHEMBL3605449 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H26N4O2 |
|---|
| Mol. Mass. | 414.4995 |
|---|
| SMILES | CC(c1ccccn1)n1c(C)c(C(=O)NCc2c(C)cc(C)[nH]c2=O)c2ccccc12 |
|---|
| Structure | 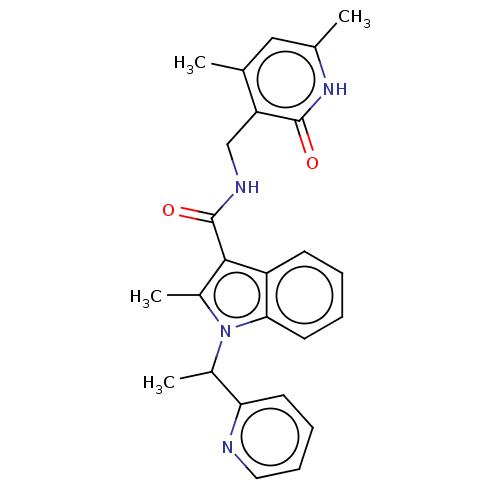
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Gehling, VS; Vaswani, RG; Nasveschuk, CG; Duplessis, M; Iyer, P; Balasubramanian, S; Zhao, F; Good, AC; Campbell, R; Lee, C; Dakin, LA; Cook, AS; Gagnon, A; Harmange, JC; Audia, JE; Cummings, RT; Normant, E; Trojer, P; Albrecht, BK Discovery, design, and synthesis of indole-based EZH2 inhibitors. Bioorg Med Chem Lett25:3644-9 (2015) [PubMed] Article
Gehling, VS; Vaswani, RG; Nasveschuk, CG; Duplessis, M; Iyer, P; Balasubramanian, S; Zhao, F; Good, AC; Campbell, R; Lee, C; Dakin, LA; Cook, AS; Gagnon, A; Harmange, JC; Audia, JE; Cummings, RT; Normant, E; Trojer, P; Albrecht, BK Discovery, design, and synthesis of indole-based EZH2 inhibitors. Bioorg Med Chem Lett25:3644-9 (2015) [PubMed] Article