| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Acetylcholinesterase |
|---|
| Ligand | BDBM50139053 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1549987 (CHEMBL3755285) |
|---|
| IC50 | 5650±n/a nM |
|---|
| Citation |  Lemes, LF; de Andrade Ramos, G; de Oliveira, AS; da Silva, FM; de Castro Couto, G; da Silva Boni, M; Guimar�es, MJ; Souza, IN; Bartolini, M; Andrisano, V; do Nascimento Nogueira, PC; Silveira, ER; Brand, GD; Soukup, O; Kor�becn�, J; Romeiro, NC; Castro, NG; Bolognesi, ML; Romeiro, LA Cardanol-derived AChE inhibitors: Towards the development of dual binding derivatives for Alzheimer's disease. Eur J Med Chem108:687-700 (2016) [PubMed] Article Lemes, LF; de Andrade Ramos, G; de Oliveira, AS; da Silva, FM; de Castro Couto, G; da Silva Boni, M; Guimar�es, MJ; Souza, IN; Bartolini, M; Andrisano, V; do Nascimento Nogueira, PC; Silveira, ER; Brand, GD; Soukup, O; Kor�becn�, J; Romeiro, NC; Castro, NG; Bolognesi, ML; Romeiro, LA Cardanol-derived AChE inhibitors: Towards the development of dual binding derivatives for Alzheimer's disease. Eur J Med Chem108:687-700 (2016) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Acetylcholinesterase |
|---|
| Name: | Acetylcholinesterase |
|---|
| Synonyms: | ACES_HUMAN | ACHE | Acetylcholinesterase (AChE) | Acetylcholinesterase (human AChE) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 67792.70 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P22303 |
|---|
| Residue: | 614 |
|---|
| Sequence: | MRPPQCLLHTPSLASPLLLLLLWLLGGGVGAEGREDAELLVTVRGGRLRGIRLKTPGGPV
SAFLGIPFAEPPMGPRRFLPPEPKQPWSGVVDATTFQSVCYQYVDTLYPGFEGTEMWNPN
RELSEDCLYLNVWTPYPRPTSPTPVLVWIYGGGFYSGASSLDVYDGRFLVQAERTVLVSM
NYRVGAFGFLALPGSREAPGNVGLLDQRLALQWVQENVAAFGGDPTSVTLFGESAGAASV
GMHLLSPPSRGLFHRAVLQSGAPNGPWATVGMGEARRRATQLAHLVGCPPGGTGGNDTEL
VACLRTRPAQVLVNHEWHVLPQESVFRFSFVPVVDGDFLSDTPEALINAGDFHGLQVLVG
VVKDEGSYFLVYGAPGFSKDNESLISRAEFLAGVRVGVPQVSDLAAEAVVLHYTDWLHPE
DPARLREALSDVVGDHNVVCPVAQLAGRLAAQGARVYAYVFEHRASTLSWPLWMGVPHGY
EIEFIFGIPLDPSRNYTAEEKIFAQRLMRYWANFARTGDPNEPRDPKAPQWPPYTAGAQQ
YVSLDLRPLEVRRGLRAQACAFWNRFLPKLLSATDTLDEAERQWKAEFHRWSSYMVHWKN
QFDHYSKQDRCSDL
|
|
|
|---|
| BDBM50139053 |
|---|
| n/a |
|---|
| Name | BDBM50139053 |
|---|
| Synonyms: | CHEMBL3752227 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H37NO2 |
|---|
| Mol. Mass. | 383.5668 |
|---|
| SMILES | CCN(CCCCCCCCc1cccc(OC)c1)Cc1ccccc1OC |
|---|
| Structure | 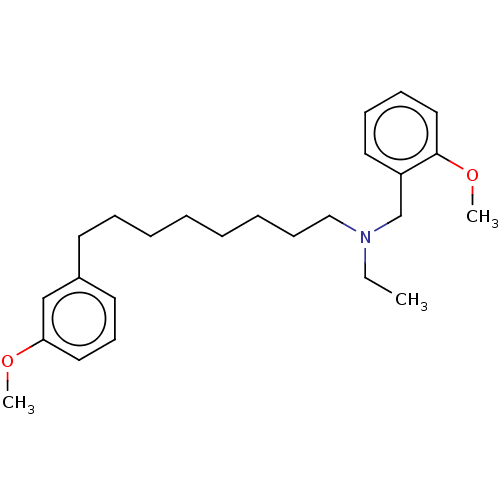
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Lemes, LF; de Andrade Ramos, G; de Oliveira, AS; da Silva, FM; de Castro Couto, G; da Silva Boni, M; Guimar�es, MJ; Souza, IN; Bartolini, M; Andrisano, V; do Nascimento Nogueira, PC; Silveira, ER; Brand, GD; Soukup, O; Kor�becn�, J; Romeiro, NC; Castro, NG; Bolognesi, ML; Romeiro, LA Cardanol-derived AChE inhibitors: Towards the development of dual binding derivatives for Alzheimer's disease. Eur J Med Chem108:687-700 (2016) [PubMed] Article
Lemes, LF; de Andrade Ramos, G; de Oliveira, AS; da Silva, FM; de Castro Couto, G; da Silva Boni, M; Guimar�es, MJ; Souza, IN; Bartolini, M; Andrisano, V; do Nascimento Nogueira, PC; Silveira, ER; Brand, GD; Soukup, O; Kor�becn�, J; Romeiro, NC; Castro, NG; Bolognesi, ML; Romeiro, LA Cardanol-derived AChE inhibitors: Towards the development of dual binding derivatives for Alzheimer's disease. Eur J Med Chem108:687-700 (2016) [PubMed] Article