| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Serine/threonine-protein kinase tousled-like 1 |
|---|
| Ligand | BDBM50154291 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1560702 (CHEMBL3778053) |
|---|
| IC50 | <5000±n/a nM |
|---|
| Citation |  Shchekotikhin, AE; Dezhenkova, LG; Tsvetkov, VB; Luzikov, YN; Volodina, YL; Tatarskiy, VV; Kalinina, AA; Treshalin, MI; Treshalina, HM; Romanenko, VI; Kaluzhny, DN; Kubbutat, M; Schols, D; Pommier, Y; Shtil, AA; Preobrazhenskaya, MN Discovery of antitumor anthra[2,3-b]furan-3-carboxamides: Optimization of synthesis and evaluation of antitumor properties. Eur J Med Chem112:114-29 (2016) [PubMed] Article Shchekotikhin, AE; Dezhenkova, LG; Tsvetkov, VB; Luzikov, YN; Volodina, YL; Tatarskiy, VV; Kalinina, AA; Treshalin, MI; Treshalina, HM; Romanenko, VI; Kaluzhny, DN; Kubbutat, M; Schols, D; Pommier, Y; Shtil, AA; Preobrazhenskaya, MN Discovery of antitumor anthra[2,3-b]furan-3-carboxamides: Optimization of synthesis and evaluation of antitumor properties. Eur J Med Chem112:114-29 (2016) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Serine/threonine-protein kinase tousled-like 1 |
|---|
| Name: | Serine/threonine-protein kinase tousled-like 1 |
|---|
| Synonyms: | KIAA0137 | Serine/threonine-protein kinase tousled-like 1 | TLK1 | TLK1_HUMAN |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 86722.30 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | gi_33636698 |
|---|
| Residue: | 766 |
|---|
| Sequence: | MSVQSSSGSLEGPPSWSQLSTSPTPGSAAAARSLLNHTPPSGRPREGAMDELHSLDPRRQ
ELLEARFTGVASGSTGSTGSCSVGAKASTNNESSNHSFGSLGSLSDKESETPEKKQSESS
RGRKRKAENQNESSQGKSIGGRGHKISDYFEYQGGNGSSPVRGIPPAIRSPQNSHSHSTP
SSSVRPNSPSPTALAFGDHPIVQPKQLSFKIIQTDLTMLKLAALESNKIQDLEKKEGRID
DLLRANCDLRRQIDEQQKLLEKYKERLNKCISMSKKLLIEKSTQEKLSSREKSMQDRLRL
GHFTTVRHGASFTEQWTDGFAFQNLVKQQEWVNQQREDIERQRKLLAKRKPPTANNSQAP
STNSEPKQRKNKAVNGAENDPFVRPNLPQLLTLAEYHEQEEIFKLRLGHLKKEEAEIQAE
LERLERVRNLHIRELKRINNEDNSQFKDHPTLNERYLLLHLLGRGGFSEVYKAFDLYEQR
YAAVKIHQLNKSWRDEKKENYHKHACREYRIHKELDHPRIVKLYDYFSLDTDTFCTVLEY
CEGNDLDFYLKQHKLMSEKEARSIVMQIVNALRYLNEIKPPIIHYDLKPGNILLVDGTAC
GEIKITDFGLSKIMDDDSYGVDGMDLTSQGAGTYWYLPPECFVVGKEPPKISNKVDVWSV
GVIFFQCLYGRKPFGHNQSQQDILQENTILKATEVQFPVKPVVSSEAKAFIRRCLAYRKE
DRFDVHQLANDPYLLPHMRRSNSSGNLHMAGLTASPTPPSSSIITY
|
|
|
|---|
| BDBM50154291 |
|---|
| n/a |
|---|
| Name | BDBM50154291 |
|---|
| Synonyms: | CHEMBL3775181 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H22N2O9S |
|---|
| Mol. Mass. | 502.494 |
|---|
| SMILES | CS(O)(=O)=O.Cc1oc2c(O)c3C(=O)c4ccccc4C(=O)c3c(O)c2c1C(=O)N1CC[C@H](N)C1 |r| |
|---|
| Structure | 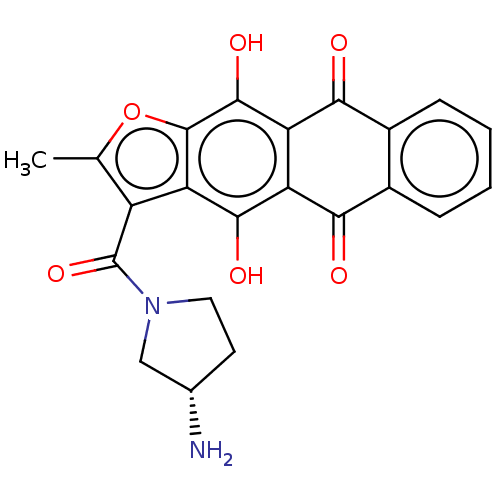
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Shchekotikhin, AE; Dezhenkova, LG; Tsvetkov, VB; Luzikov, YN; Volodina, YL; Tatarskiy, VV; Kalinina, AA; Treshalin, MI; Treshalina, HM; Romanenko, VI; Kaluzhny, DN; Kubbutat, M; Schols, D; Pommier, Y; Shtil, AA; Preobrazhenskaya, MN Discovery of antitumor anthra[2,3-b]furan-3-carboxamides: Optimization of synthesis and evaluation of antitumor properties. Eur J Med Chem112:114-29 (2016) [PubMed] Article
Shchekotikhin, AE; Dezhenkova, LG; Tsvetkov, VB; Luzikov, YN; Volodina, YL; Tatarskiy, VV; Kalinina, AA; Treshalin, MI; Treshalina, HM; Romanenko, VI; Kaluzhny, DN; Kubbutat, M; Schols, D; Pommier, Y; Shtil, AA; Preobrazhenskaya, MN Discovery of antitumor anthra[2,3-b]furan-3-carboxamides: Optimization of synthesis and evaluation of antitumor properties. Eur J Med Chem112:114-29 (2016) [PubMed] Article