| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Glycogen phosphorylase, muscle form |
|---|
| Ligand | BDBM50295860 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1630555 (CHEMBL3873261) |
|---|
| Ki | 76000±n/a nM |
|---|
| Citation |  Kantsadi, AL; Bokor, �; Kun, S; Stravodimos, GA; Chatzileontiadou, DSM; Leonidas, DD; Juh�sz-T�th, �; Szak�cs, A; Batta, G; Docsa, T; Gergely, P; Soms�k, L Synthetic, enzyme kinetic, and protein crystallographic studies of C-�-d-glucopyranosyl pyrroles and imidazoles reveal and explain low nanomolar inhibition of human liver glycogen phosphorylase. Eur J Med Chem123:737-745 (2016) [PubMed] Article Kantsadi, AL; Bokor, �; Kun, S; Stravodimos, GA; Chatzileontiadou, DSM; Leonidas, DD; Juh�sz-T�th, �; Szak�cs, A; Batta, G; Docsa, T; Gergely, P; Soms�k, L Synthetic, enzyme kinetic, and protein crystallographic studies of C-�-d-glucopyranosyl pyrroles and imidazoles reveal and explain low nanomolar inhibition of human liver glycogen phosphorylase. Eur J Med Chem123:737-745 (2016) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Glycogen phosphorylase, muscle form |
|---|
| Name: | Glycogen phosphorylase, muscle form |
|---|
| Synonyms: | Glycogen Phosphorylase (PYGM) | Glycogen phosphorylase a (RMGPa) | Glycogen phosphorylase, muscle form | Myophosphorylase | PYGM | PYGM_RABIT |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 97296.32 |
|---|
| Organism: | Oryctolagus cuniculus (rabbit) |
|---|
| Description: | Phosphorylation of Ser-15 converts phosphorylase B (unphosphorylated) to phosphorylase A. |
|---|
| Residue: | 843 |
|---|
| Sequence: | MSRPLSDQEKRKQISVRGLAGVENVTELKKNFNRHLHFTLVKDRNVATPRDYYFALAHTV
RDHLVGRWIRTQQHYYEKDPKRIYYLSLEFYMGRTLQNTMVNLALENACDEATYQLGLDM
EELEEIEEDAGLGNGGLGRLAACFLDSMATLGLAAYGYGIRYEFGIFNQKICGGWQMEEA
DDWLRYGNPWEKARPEFTLPVHFYGRVEHTSQGAKWVDTQVVLAMPYDTPVPGYRNNVVN
TMRLWSAKAPNDFNLKDFNVGGYIQAVLDRNLAENISRVLYPNDNFFEGKELRLKQEYFV
VAATLQDIIRRFKSSKFGCRDPVRTNFDAFPDKVAIQLNDTHPSLAIPELMRVLVDLERL
DWDKAWEVTVKTCAYTNHTVLPEALERWPVHLLETLLPRHLQIIYEINQRFLNRVAAAFP
GDVDRLRRMSLVEEGAVKRINMAHLCIAGSHAVNGVARIHSEILKKTIFKDFYELEPHKF
QNKTNGITPRRWLVLCNPGLAEIIAERIGEEYISDLDQLRKLLSYVDDEAFIRDVAKVKQ
ENKLKFAAYLEREYKVHINPNSLFDVQVKRIHEYKRQLLNCLHVITLYNRIKKEPNKFVV
PRTVMIGGKAAPGYHMAKMIIKLITAIGDVVNHDPVVGDRLRVIFLENYRVSLAEKVIPA
ADLSEQISTAGTEASGTGNMKFMLNGALTIGTMDGANVEMAEEAGEENFFIFGMRVEDVD
RLDQRGYNAQEYYDRIPELRQIIEQLSSGFFSPKQPDLFKDIVNMLMHHDRFKVFADYEE
YVKCQERVSALYKNPREWTRMVIRNIATSGKFSSDRTIAQYAREIWGVEPSRQRLPAPDE
KIP
|
|
|
|---|
| BDBM50295860 |
|---|
| n/a |
|---|
| Name | BDBM50295860 |
|---|
| Synonyms: | (2R,3R,4S,5S,6R)-2-(benzo[d]thiazol-2-yl)-6-(hydroxymethyl)tetrahydro-2H-pyran-3,4,5-triol | CHEMBL559793 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C13H15NO5S |
|---|
| Mol. Mass. | 297.327 |
|---|
| SMILES | OC[C@H]1O[C@H]([C@H](O)[C@@H](O)[C@@H]1O)c1nc2ccccc2s1 |r| |
|---|
| Structure | 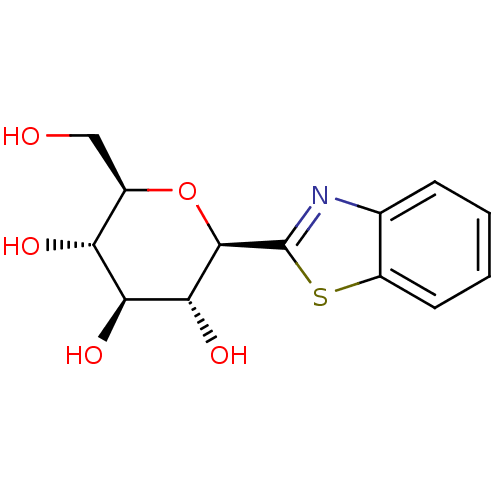
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Kantsadi, AL; Bokor, �; Kun, S; Stravodimos, GA; Chatzileontiadou, DSM; Leonidas, DD; Juh�sz-T�th, �; Szak�cs, A; Batta, G; Docsa, T; Gergely, P; Soms�k, L Synthetic, enzyme kinetic, and protein crystallographic studies of C-�-d-glucopyranosyl pyrroles and imidazoles reveal and explain low nanomolar inhibition of human liver glycogen phosphorylase. Eur J Med Chem123:737-745 (2016) [PubMed] Article
Kantsadi, AL; Bokor, �; Kun, S; Stravodimos, GA; Chatzileontiadou, DSM; Leonidas, DD; Juh�sz-T�th, �; Szak�cs, A; Batta, G; Docsa, T; Gergely, P; Soms�k, L Synthetic, enzyme kinetic, and protein crystallographic studies of C-�-d-glucopyranosyl pyrroles and imidazoles reveal and explain low nanomolar inhibition of human liver glycogen phosphorylase. Eur J Med Chem123:737-745 (2016) [PubMed] Article