| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Glutaminase kidney isoform, mitochondrial |
|---|
| Ligand | BDBM50238194 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1663491 (CHEMBL4013172) |
|---|
| IC50 | 49±n/a nM |
|---|
| Citation |  Yeh, TK; Kuo, CC; Lee, YZ; Ke, YY; Chu, KF; Hsu, HY; Chang, HY; Liu, YW; Song, JS; Yang, CW; Lin, LM; Sun, M; Wu, SH; Kuo, PC; Shih, C; Chen, CT; Tsou, LK; Lee, SJ Design, Synthesis, and Evaluation of Thiazolidine-2,4-dione Derivatives as a Novel Class of Glutaminase Inhibitors. J Med Chem60:5599-5612 (2017) [PubMed] Article Yeh, TK; Kuo, CC; Lee, YZ; Ke, YY; Chu, KF; Hsu, HY; Chang, HY; Liu, YW; Song, JS; Yang, CW; Lin, LM; Sun, M; Wu, SH; Kuo, PC; Shih, C; Chen, CT; Tsou, LK; Lee, SJ Design, Synthesis, and Evaluation of Thiazolidine-2,4-dione Derivatives as a Novel Class of Glutaminase Inhibitors. J Med Chem60:5599-5612 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Glutaminase kidney isoform, mitochondrial |
|---|
| Name: | Glutaminase kidney isoform, mitochondrial |
|---|
| Synonyms: | GLS | GLS1 | GLSK_HUMAN | Glutaminase 1 | K-glutaminase | KIAA0838 | L-glutamine amidohydrolase |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 73471.89 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | O94925 |
|---|
| Residue: | 669 |
|---|
| Sequence: | MMRLRGSGMLRDLLLRSPAGVSATLRRAQPLVTLCRRPRGGGRPAAGPAAAARLHPWWGG
GGWPAEPLARGLSSSPSEILQELGKGSTHPQPGVSPPAAPAAPGPKDGPGETDAFGNSEG
KELVASGENKIKQGLLPSLEDLLFYTIAEGQEKIPVHKFITALKSTGLRTSDPRLKECMD
MLRLTLQTTSDGVMLDKDLFKKCVQSNIVLLTQAFRRKFVIPDFMSFTSHIDELYESAKK
QSGGKVADYIPQLAKFSPDLWGVSVCTVDGQRHSTGDTKVPFCLQSCVKPLKYAIAVNDL
GTEYVHRYVGKEPSGLRFNKLFLNEDDKPHNPMVNAGAIVVTSLIKQGVNNAEKFDYVMQ
FLNKMAGNEYVGFSNATFQSERESGDRNFAIGYYLKEKKCFPEGTDMVGILDFYFQLCSI
EVTCESASVMAATLANGGFCPITGERVLSPEAVRNTLSLMHSCGMYDFSGQFAFHVGLPA
KSGVAGGILLVVPNVMGMMCWSPPLDKMGNSVKGIHFCHDLVSLCNFHNYDNLRHFAKKL
DPRREGGDQRVKSVINLLFAAYTGDVSALRRFALSAMDMEQRDYDSRTALHVAAAEGHVE
VVKFLLEACKVNPFPKDRWNNTPMDEALHFGHHDVFKILQEYQVQYTPQGDSDNGKENQT
VHKNLDGLL
|
|
|
|---|
| BDBM50238194 |
|---|
| n/a |
|---|
| Name | BDBM50238194 |
|---|
| Synonyms: | CHEMBL4080388 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C32H19N3O4S4 |
|---|
| Mol. Mass. | 637.771 |
|---|
| SMILES | O=C1NC(=O)\C(S1)=C\c1cc(-c2cccs2)n(c1-c1cccs1)-c1ccc(cc1)-c1ccc(\C=C2/SC(=O)NC2=O)cc1 |
|---|
| Structure | 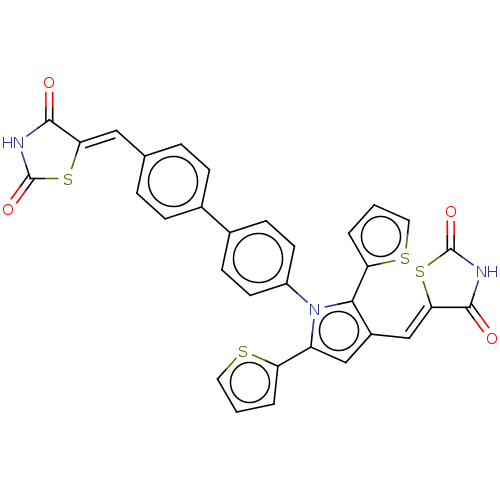
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Yeh, TK; Kuo, CC; Lee, YZ; Ke, YY; Chu, KF; Hsu, HY; Chang, HY; Liu, YW; Song, JS; Yang, CW; Lin, LM; Sun, M; Wu, SH; Kuo, PC; Shih, C; Chen, CT; Tsou, LK; Lee, SJ Design, Synthesis, and Evaluation of Thiazolidine-2,4-dione Derivatives as a Novel Class of Glutaminase Inhibitors. J Med Chem60:5599-5612 (2017) [PubMed] Article
Yeh, TK; Kuo, CC; Lee, YZ; Ke, YY; Chu, KF; Hsu, HY; Chang, HY; Liu, YW; Song, JS; Yang, CW; Lin, LM; Sun, M; Wu, SH; Kuo, PC; Shih, C; Chen, CT; Tsou, LK; Lee, SJ Design, Synthesis, and Evaluation of Thiazolidine-2,4-dione Derivatives as a Novel Class of Glutaminase Inhibitors. J Med Chem60:5599-5612 (2017) [PubMed] Article