| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Indoleamine 2,3-dioxygenase 1 |
|---|
| Ligand | BDBM494784 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Enzyme Assay |
|---|
| IC50 | 55.4±n/a nM |
|---|
| Citation |  Liu, K; Achab, A; Biju, P; Cernak, TA; Deng, Y; Fradera, X; Guo, L; Han, Y; He, S; Kozlowski, J; Li, D; Li, G; Pu, Q; Shi, Z; Yu, W; Zhang, H Substituted n′-hydroxycarbamimidoyl-1,2,5-oxadiazole compounds as indoleamine 2,3-dioxygenase (IDO) inhibitors US Patent US10988487 Publication Date 4/27/2021 Liu, K; Achab, A; Biju, P; Cernak, TA; Deng, Y; Fradera, X; Guo, L; Han, Y; He, S; Kozlowski, J; Li, D; Li, G; Pu, Q; Shi, Z; Yu, W; Zhang, H Substituted n′-hydroxycarbamimidoyl-1,2,5-oxadiazole compounds as indoleamine 2,3-dioxygenase (IDO) inhibitors US Patent US10988487 Publication Date 4/27/2021 |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Indoleamine 2,3-dioxygenase 1 |
|---|
| Name: | Indoleamine 2,3-dioxygenase 1 |
|---|
| Synonyms: | I23O1_HUMAN | IDO | IDO-1 | IDO1 | INDO | Indoleamine 2,3-Dioxygenasae (IDO) | Indoleamine 2,3-dioxygenase | Indoleamine-pyrrole 2,3-dioxygenase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 45330.80 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P14902 |
|---|
| Residue: | 403 |
|---|
| Sequence: | MAHAMENSWTISKEYHIDEEVGFALPNPQENLPDFYNDWMFIAKHLPDLIESGQLRERVE
KLNMLSIDHLTDHKSQRLARLVLGCITMAYVWGKGHGDVRKVLPRNIAVPYCQLSKKLEL
PPILVYADCVLANWKKKDPNKPLTYENMDVLFSFRDGDCSKGFFLVSLLVEIAAASAIKV
IPTVFKAMQMQERDTLLKALLEIASCLEKALQVFHQIHDHVNPKAFFSVLRIYLSGWKGN
PQLSDGLVYEGFWEDPKEFAGGSAGQSSVFQCFDVLLGIQQTAGGGHAAQFLQDMRRYMP
PAHRNFLCSLESNPSVREFVLSKGDAGLREAYDACVKALVSLRSYHLQIVTKYILIPASQ
QPKENKTSEDPSKLEAKGTGGTDLMNFLKTVRSTTEKSLLKEG
|
|
|
|---|
| BDBM494784 |
|---|
| n/a |
|---|
| Name | BDBM494784 |
|---|
| Synonyms: | 2-[(4-{N-[(1S)-6-fluoro-2,3- dihydro-1H-inden-1-yl]-N'- hydroxycarbamimidoyl}-1,2,5- oxadiazol-3-yl)sulfanyl]-N-(2- hydroxypropyl)acetamide | US10988487, Example 180 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C17H20FN5O4S |
|---|
| Mol. Mass. | 409.435 |
|---|
| SMILES | CC(O)CNC(=O)CSc1nonc1\C(N[C@H]1CCc2ccc(F)cc12)=N\O |r| |
|---|
| Structure | 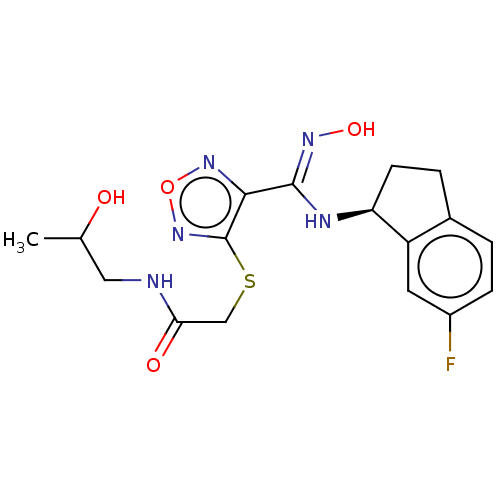
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Liu, K; Achab, A; Biju, P; Cernak, TA; Deng, Y; Fradera, X; Guo, L; Han, Y; He, S; Kozlowski, J; Li, D; Li, G; Pu, Q; Shi, Z; Yu, W; Zhang, H Substituted n′-hydroxycarbamimidoyl-1,2,5-oxadiazole compounds as indoleamine 2,3-dioxygenase (IDO) inhibitors US Patent US10988487 Publication Date 4/27/2021
Liu, K; Achab, A; Biju, P; Cernak, TA; Deng, Y; Fradera, X; Guo, L; Han, Y; He, S; Kozlowski, J; Li, D; Li, G; Pu, Q; Shi, Z; Yu, W; Zhang, H Substituted n′-hydroxycarbamimidoyl-1,2,5-oxadiazole compounds as indoleamine 2,3-dioxygenase (IDO) inhibitors US Patent US10988487 Publication Date 4/27/2021