

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50259746 (4-Hydroxyphenyl)ethyl trans-ferulate::CHEMBL481245::p-Hydroxyphenethyl trans-ferulate (18)::p-hydroxyphenethyl trans-ferulate
SMILES: COc1cc(\C=C\C(=O)OCCc2ccc(O)cc2)ccc1O
InChI Key: InChIKey=JMSFLLZUCIXALN-WEVVVXLNSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Carbonic anhydrase 7 (Homo sapiens (Human)) | BDBM50259746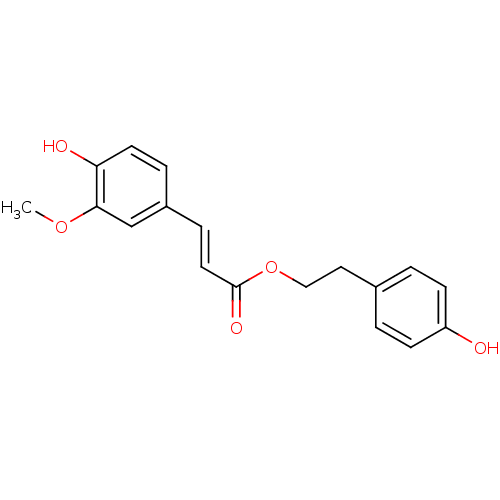 ((4-Hydroxyphenyl)ethyl trans-ferulate | CHEMBL4812...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | PubMed | 620 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze Curated by ChEMBL | Assay Description Inhibition of human carbonic anhydrase 7 preincubated for 15 mins by stopped-flow CO2 hydration assay | Bioorg Med Chem 23: 7181-8 (2015) BindingDB Entry DOI: 10.7270/Q29W0HB2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic Anhydrase VB (Homo sapiens (Human)) | BDBM50259746 ((4-Hydroxyphenyl)ethyl trans-ferulate | CHEMBL4812...) | Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | PubMed | 670 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze Curated by ChEMBL | Assay Description Inhibition of human carbonic anhydrase 5B preincubated for 15 mins by stopped-flow CO2 hydration assay | Bioorg Med Chem 23: 7181-8 (2015) BindingDB Entry DOI: 10.7270/Q29W0HB2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic Anhydrase XIV (Homo sapiens (Human)) | BDBM50259746 ((4-Hydroxyphenyl)ethyl trans-ferulate | CHEMBL4812...) | PDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | PubMed | 720 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze Curated by ChEMBL | Assay Description Inhibition of human carbonic anhydrase 14 preincubated for 15 mins by stopped-flow CO2 hydration assay | Bioorg Med Chem 23: 7181-8 (2015) BindingDB Entry DOI: 10.7270/Q29W0HB2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 6 (CA-VI) (Homo sapiens (Human)) | BDBM50259746 ((4-Hydroxyphenyl)ethyl trans-ferulate | CHEMBL4812...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | PubMed | 770 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze Curated by ChEMBL | Assay Description Inhibition of human carbonic anhydrase 6 preincubated for 15 mins by stopped-flow CO2 hydration assay | Bioorg Med Chem 23: 7181-8 (2015) BindingDB Entry DOI: 10.7270/Q29W0HB2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic Anhydrase VA (Homo sapiens (Human)) | BDBM50259746 ((4-Hydroxyphenyl)ethyl trans-ferulate | CHEMBL4812...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | PubMed | 810 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze Curated by ChEMBL | Assay Description Inhibition of human carbonic anhydrase 5A preincubated for 15 mins by stopped-flow CO2 hydration assay | Bioorg Med Chem 23: 7181-8 (2015) BindingDB Entry DOI: 10.7270/Q29W0HB2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 9 (Homo sapiens (Human)) | BDBM50259746 ((4-Hydroxyphenyl)ethyl trans-ferulate | CHEMBL4812...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | PubMed | 810 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze Curated by ChEMBL | Assay Description Inhibition of human catalytic domain of carbonic anhydrase 9 preincubated for 15 mins by stopped-flow CO2 hydration assay | Bioorg Med Chem 23: 7181-8 (2015) BindingDB Entry DOI: 10.7270/Q29W0HB2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic Anhydrase XIII (Mus musculus (mouse)) | BDBM50259746 ((4-Hydroxyphenyl)ethyl trans-ferulate | CHEMBL4812...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | PubMed | 930 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze Curated by ChEMBL | Assay Description Inhibition of mouse carbonic anhydrase 13 preincubated for 15 mins by stopped-flow CO2 hydration assay | Bioorg Med Chem 23: 7181-8 (2015) BindingDB Entry DOI: 10.7270/Q29W0HB2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 12 (Homo sapiens (Human)) | BDBM50259746 ((4-Hydroxyphenyl)ethyl trans-ferulate | CHEMBL4812...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | PubMed | 8.47E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze Curated by ChEMBL | Assay Description Inhibition of human catalytic domain of carbonic anhydrase 12 preincubated for 15 mins by stopped-flow CO2 hydration assay | Bioorg Med Chem 23: 7181-8 (2015) BindingDB Entry DOI: 10.7270/Q29W0HB2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 4 (Homo sapiens (Human)) | BDBM50259746 ((4-Hydroxyphenyl)ethyl trans-ferulate | CHEMBL4812...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | PubMed | 4.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze Curated by ChEMBL | Assay Description Inhibition of human carbonic anhydrase 4 preincubated for 15 mins by stopped-flow CO2 hydration assay | Bioorg Med Chem 23: 7181-8 (2015) BindingDB Entry DOI: 10.7270/Q29W0HB2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic Anhydrase III (Homo sapiens (Human)) | BDBM50259746 ((4-Hydroxyphenyl)ethyl trans-ferulate | CHEMBL4812...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | PubMed | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze Curated by ChEMBL | Assay Description Inhibition of human carbonic anhydrase 3 preincubated for 15 mins by stopped-flow CO2 hydration assay | Bioorg Med Chem 23: 7181-8 (2015) BindingDB Entry DOI: 10.7270/Q29W0HB2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 2 (Homo sapiens (Human)) | BDBM50259746 ((4-Hydroxyphenyl)ethyl trans-ferulate | CHEMBL4812...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | PubMed | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze Curated by ChEMBL | Assay Description Inhibition of human carbonic anhydrase 2 preincubated for 15 mins by stopped-flow CO2 hydration assay | Bioorg Med Chem 23: 7181-8 (2015) BindingDB Entry DOI: 10.7270/Q29W0HB2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 1 (Homo sapiens (Human)) | BDBM50259746 ((4-Hydroxyphenyl)ethyl trans-ferulate | CHEMBL4812...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | PubMed | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Firenze Curated by ChEMBL | Assay Description Inhibition of human carbonic anhydrase 1 preincubated for 15 mins by stopped-flow CO2 hydration assay | Bioorg Med Chem 23: 7181-8 (2015) BindingDB Entry DOI: 10.7270/Q29W0HB2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hepatocyte growth factor receptor (Homo sapiens (Human)) | BDBM50259746 ((4-Hydroxyphenyl)ethyl trans-ferulate | CHEMBL4812...) | PDB MMDB KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 1.29E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Louisiana at Monroe Curated by ChEMBL | Assay Description Inhibition of recombinant human N-terminal His-tagged cytoplasmic c-MET kinase domain (956 to 1390 residues) phosphorylation expressed in baculovirus... | Eur J Med Chem 118: 299-315 (2016) BindingDB Entry DOI: 10.7270/Q2GT5Q35 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 7 (Homo sapiens (Human)) | BDBM50259746 ((4-Hydroxyphenyl)ethyl trans-ferulate | CHEMBL4812...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 4.76E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Illinois at Chicago Curated by ChEMBL | Assay Description Displacement of [3H]LSD from human recombinant 5HT7 receptor expressed in CHO cells by liquid scintillation counting | J Nat Prod 69: 536-41 (2006) Article DOI: 10.1021/np050301s BindingDB Entry DOI: 10.7270/Q2ST7PM1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Pancreatic lipase (Sus scrofa (Pig)) | BDBM50259746 ((4-Hydroxyphenyl)ethyl trans-ferulate | CHEMBL4812...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | 7.4 | 37 |
Seoul National University | Assay Description Various concentrations of test compound (5, 10, 25, 50 and 100 mM) were dissolved in DMSO (final culture concentration 0.1%) and 4-MU oleate and lipa... | J Enzyme Inhib Med Chem 29: 1-6 (2014) Article DOI: 10.3109/14756366.2012.742517 BindingDB Entry DOI: 10.7270/Q2DJ5DKB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||