

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50346864 CHEMBL1797641::CHEMBL3104337::US8765820, 8
SMILES: NC1CC1c1ccc(NC(=O)C(Cc2ccccc2)NC(=O)OCc2ccccc2)cc1
InChI Key: InChIKey=HDVDLEPVLVTECB-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM50346864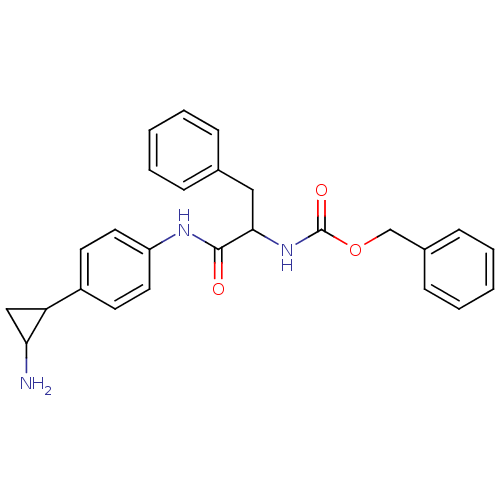 (CHEMBL1797641 | CHEMBL3104337 | US8765820, 8) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Copenhagen Curated by ChEMBL | Assay Description Inhibition of LSD1 | Bioorg Med Chem 19: 3625-36 (2011) Article DOI: 10.1016/j.bmc.2011.01.046 BindingDB Entry DOI: 10.7270/Q23X870S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Amine oxidase (flavin-containing) A (Homo sapiens (Human)) | BDBM50346864 (CHEMBL1797641 | CHEMBL3104337 | US8765820, 8) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto Prefectural University of Medicine Curated by ChEMBL | Assay Description Inhibition of MAO A | J Med Chem 54: 8236-50 (2011) Article DOI: 10.1021/jm201048w BindingDB Entry DOI: 10.7270/Q2D50NFR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM50346864 (CHEMBL1797641 | CHEMBL3104337 | US8765820, 8) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto Prefectural University of Medicine Curated by ChEMBL | Assay Description Inhibition of LSD1 | J Med Chem 54: 8236-50 (2011) Article DOI: 10.1021/jm201048w BindingDB Entry DOI: 10.7270/Q2D50NFR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM50346864 (CHEMBL1797641 | CHEMBL3104337 | US8765820, 8) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sapienza University of Roma Curated by ChEMBL | Assay Description Inhibition of LSD1 (unknown origin) | ACS Med Chem Lett 6: 173-7 (2015) Article DOI: 10.1021/ml500424z BindingDB Entry DOI: 10.7270/Q2833TQM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM50346864 (CHEMBL1797641 | CHEMBL3104337 | US8765820, 8) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | 2.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Universita Degli Studi di Roma “La Sapienza”; Fondazione IEO; Universita Degli Studi di Pavia; Universita Degli Studi di Milano US Patent | Assay Description Human recombinant MAO A and MAO B were expressed in Pichia pastoris and purified as published (Binda C, et al., Proc. Natl. Acad. Sci. USA 100: 9750-... | US Patent US8765820 (2014) BindingDB Entry DOI: 10.7270/Q20P0XPN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Amine oxidase (flavin-containing) A (Homo sapiens (Human)) | BDBM50346864 (CHEMBL1797641 | CHEMBL3104337 | US8765820, 8) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Universita Degli Studi di Roma “La Sapienza”; Fondazione IEO; Universita Degli Studi di Pavia; Universita Degli Studi di Milano US Patent | Assay Description Human recombinant MAO A and MAO B were expressed in Pichia pastoris and purified as published (Binda C, et al., Proc. Natl. Acad. Sci. USA 100: 9750-... | US Patent US8765820 (2014) BindingDB Entry DOI: 10.7270/Q20P0XPN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 2 (LSD2) (Mus musculus (Mouse)) | BDBM50346864 (CHEMBL1797641 | CHEMBL3104337 | US8765820, 8) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | 2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Universita Degli Studi di Roma “La Sapienza”; Fondazione IEO; Universita Degli Studi di Pavia; Universita Degli Studi di Milano US Patent | Assay Description Human recombinant MAO A and MAO B were expressed in Pichia pastoris and purified as published (Binda C, et al., Proc. Natl. Acad. Sci. USA 100: 9750-... | US Patent US8765820 (2014) BindingDB Entry DOI: 10.7270/Q20P0XPN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1B (Homo sapiens (Human)) | BDBM50346864 (CHEMBL1797641 | CHEMBL3104337 | US8765820, 8) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 3.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto Prefectural University of Medicine Curated by ChEMBL | Assay Description Inhibition of LSD2 | J Med Chem 54: 8236-50 (2011) Article DOI: 10.1021/jm201048w BindingDB Entry DOI: 10.7270/Q2D50NFR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Amine oxidase [flavin-containing] B (Homo sapiens (Human)) | BDBM50346864 (CHEMBL1797641 | CHEMBL3104337 | US8765820, 8) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >1.33E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kyoto Prefectural University of Medicine Curated by ChEMBL | Assay Description Inhibition of MAO B | J Med Chem 54: 8236-50 (2011) Article DOI: 10.1021/jm201048w BindingDB Entry DOI: 10.7270/Q2D50NFR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM50346864 (CHEMBL1797641 | CHEMBL3104337 | US8765820, 8) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Utah Curated by ChEMBL | Assay Description Inhibition of human recombinant LSD1 using dimethylated H3K4 peptide as substrate after 1 hr | J Med Chem 56: 9496-508 (2014) Article DOI: 10.1021/jm400870h BindingDB Entry DOI: 10.7270/Q2Z60QJ7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Amine oxidase (flavin-containing) A (Homo sapiens (Human)) | BDBM50346864 (CHEMBL1797641 | CHEMBL3104337 | US8765820, 8) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 320 | n/a | n/a | n/a | n/a | n/a | n/a |
Sapienza University of Roma Curated by ChEMBL | Assay Description Inhibition of human recombinant MAOA expressed in Pichia pastoris incubated for 15 mins prior to substrate addition measured after 30 mins by lumines... | ACS Med Chem Lett 6: 173-7 (2015) Article DOI: 10.1021/ml500424z BindingDB Entry DOI: 10.7270/Q2833TQM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM50346864 (CHEMBL1797641 | CHEMBL3104337 | US8765820, 8) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 150 | n/a | n/a | n/a | n/a | n/a | n/a |
Sapienza University of Roma Curated by ChEMBL | Assay Description Inhibition of human recombinant LSD1 expressed in Escherichia coli using H3-H4 peptide as substrate assessed as H2O2 produced after 15 mins by amplex... | ACS Med Chem Lett 6: 173-7 (2015) Article DOI: 10.1021/ml500424z BindingDB Entry DOI: 10.7270/Q2833TQM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||