 Found 69 hits with Last Name = 'maiti' and Initial = 'a'
Found 69 hits with Last Name = 'maiti' and Initial = 'a' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Aromatase
(Homo sapiens (Human)) | BDBM29227

(casimiroin analogue, 1p)Show InChI InChI=1S/C14H17NO4/c1-8-6-11(16)15(2)13-9(17-3)7-10(18-4)14(19-5)12(8)13/h6-7H,1-5H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
| Assay Description
Aromatase inhibition was quantified by measuring the fluorescent intensity of fluorescein, the hydrolysis product of dibenzylfluorescein (DBF), by ar... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | |
Aromatase
(Homo sapiens (Human)) | BDBM9460
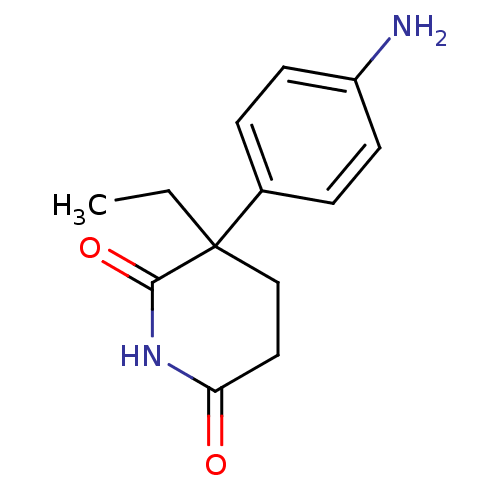
(3-(4-aminophenyl)-3-ethyl-piperidine-2,6-dione | 3...)Show InChI InChI=1S/C13H16N2O2/c1-2-13(8-7-11(16)15-12(13)17)9-3-5-10(14)6-4-9/h3-6H,2,7-8,14H2,1H3,(H,15,16,17) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| n/a | n/a | 270 | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of human aromatase |
J Med Chem 50: 2799-806 (2007)
Article DOI: 10.1021/jm070109i
BindingDB Entry DOI: 10.7270/Q2668F0Z |
More data for this
Ligand-Target Pair | |
Aromatase
(Homo sapiens (Human)) | BDBM9460

(3-(4-aminophenyl)-3-ethyl-piperidine-2,6-dione | 3...)Show InChI InChI=1S/C13H16N2O2/c1-2-13(8-7-11(16)15-12(13)17)9-3-5-10(14)6-4-9/h3-6H,2,7-8,14H2,1H3,(H,15,16,17) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| n/a | n/a | 270 | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
| Assay Description
Aromatase inhibition was quantified by measuring the fluorescent intensity of fluorescein, the hydrolysis product of dibenzylfluorescein (DBF), by ar... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | |
Aromatase
(Homo sapiens (Human)) | BDBM29229

(casimiroin analogue, 1r)Show InChI InChI=1S/C15H19NO5/c1-8-7-9(17)16(2)11-10(8)12(18-3)14(20-5)15(21-6)13(11)19-4/h7H,1-6H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 760 | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
| Assay Description
Aromatase inhibition was quantified by measuring the fluorescent intensity of fluorescein, the hydrolysis product of dibenzylfluorescein (DBF), by ar... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | |
Aromatase
(Homo sapiens (Human)) | BDBM29221
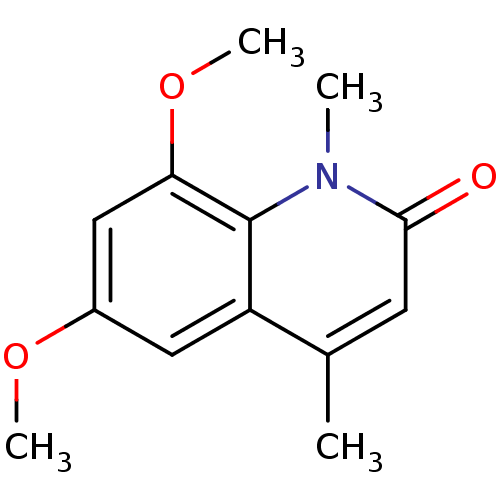
(casimiroin analogue, 1j)Show InChI InChI=1S/C13H15NO3/c1-8-5-12(15)14(2)13-10(8)6-9(16-3)7-11(13)17-4/h5-7H,1-4H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 960 | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
| Assay Description
Aromatase inhibition was quantified by measuring the fluorescent intensity of fluorescein, the hydrolysis product of dibenzylfluorescein (DBF), by ar... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | |
Ribosyldihydronicotinamide dehydrogenase [quinone]
(Homo sapiens (Human)) | BDBM23926

((E)-resveratrol | 5-[(E)-2-(4-hydroxyphenyl)etheny...)Show InChI InChI=1S/C14H12O3/c15-12-5-3-10(4-6-12)1-2-11-7-13(16)9-14(17)8-11/h1-9,15-17H/b2-1+ | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 960 | n/a | n/a | n/a | n/a | 7.5 | 23 |
Purdue University
| Assay Description
The activity of QR2 under steady-state conditions was evaluated on SpectraMax Plus 384 UV/vis spectrophotometer by monitoring the increase in absorba... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Aromatase
(Homo sapiens (Human)) | BDBM29223

(casimiroin analogue, 1l)Show InChI InChI=1S/C13H15NO3/c1-8-7-11(15)14(2)13-10(17-4)6-5-9(16-3)12(8)13/h5-7H,1-4H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 960 | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
| Assay Description
Aromatase inhibition was quantified by measuring the fluorescent intensity of fluorescein, the hydrolysis product of dibenzylfluorescein (DBF), by ar... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | |
Aromatase
(Homo sapiens (Human)) | BDBM29213

(casimiroin analogue, 1b)Show InChI InChI=1S/C12H11NO3/c1-7-5-10(14)13(2)11-8(7)3-4-9-12(11)16-6-15-9/h3-5H,6H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.25E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
| Assay Description
Aromatase inhibition was quantified by measuring the fluorescent intensity of fluorescein, the hydrolysis product of dibenzylfluorescein (DBF), by ar... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | |
Aromatase
(Homo sapiens (Human)) | BDBM50213235
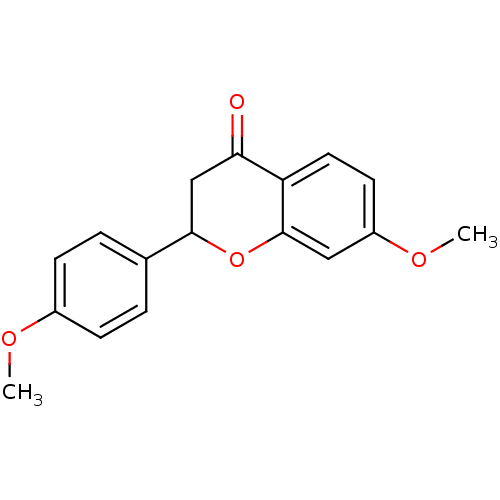
((+/-)-7-methoxy-2-(4-methoxyphenyl)chroman-4-one |...)Show InChI InChI=1S/C17H16O4/c1-19-12-5-3-11(4-6-12)16-10-15(18)14-8-7-13(20-2)9-17(14)21-16/h3-9,16H,10H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.86E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of human aromatase |
J Med Chem 50: 2799-806 (2007)
Article DOI: 10.1021/jm070109i
BindingDB Entry DOI: 10.7270/Q2668F0Z |
More data for this
Ligand-Target Pair | |
Ribosyldihydronicotinamide dehydrogenase [quinone]
(Homo sapiens (Human)) | BDBM29221

(casimiroin analogue, 1j)Show InChI InChI=1S/C13H15NO3/c1-8-5-12(15)14(2)13-10(8)6-9(16-3)7-11(13)17-4/h5-7H,1-4H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 1.90E+3 | n/a | n/a | n/a | n/a | 7.5 | 23 |
Purdue University
| Assay Description
The activity of QR2 under steady-state conditions was evaluated on SpectraMax Plus 384 UV/vis spectrophotometer by monitoring the increase in absorba... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Aromatase
(Homo sapiens (Human)) | BDBM29217

(casimiroin analogue, 1f)Show InChI InChI=1S/C12H13NO2/c1-8-7-11(14)13(2)12-9(8)5-4-6-10(12)15-3/h4-7H,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.02E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
| Assay Description
Aromatase inhibition was quantified by measuring the fluorescent intensity of fluorescein, the hydrolysis product of dibenzylfluorescein (DBF), by ar... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | |
Aromatase
(Homo sapiens (Human)) | BDBM29225

(casimiroin analogue, 1n)Show InChI InChI=1S/C14H17NO4/c1-8-6-11(16)15(2)12-9(8)7-10(17-3)13(18-4)14(12)19-5/h6-7H,1-5H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.72E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
| Assay Description
Aromatase inhibition was quantified by measuring the fluorescent intensity of fluorescein, the hydrolysis product of dibenzylfluorescein (DBF), by ar... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | |
Ornithine decarboxylase
(Homo sapiens (Human)) | BDBM50202558
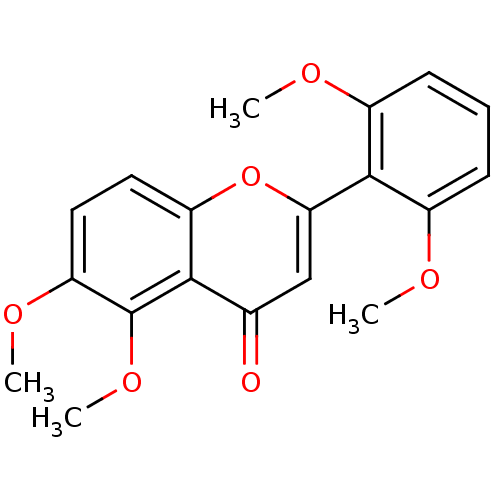
(5,6,2',6'-tetramethoxyflavone | CHEMBL375582)Show InChI InChI=1S/C19H18O6/c1-21-12-6-5-7-13(22-2)18(12)16-10-11(20)17-14(25-16)8-9-15(23-3)19(17)24-4/h5-10H,1-4H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of TPA-induced ODC activity in T24 cells |
J Med Chem 50: 350-5 (2007)
Article DOI: 10.1021/jm060915+
BindingDB Entry DOI: 10.7270/Q2NP2435 |
More data for this
Ligand-Target Pair | |
Aromatase
(Homo sapiens (Human)) | BDBM50213245
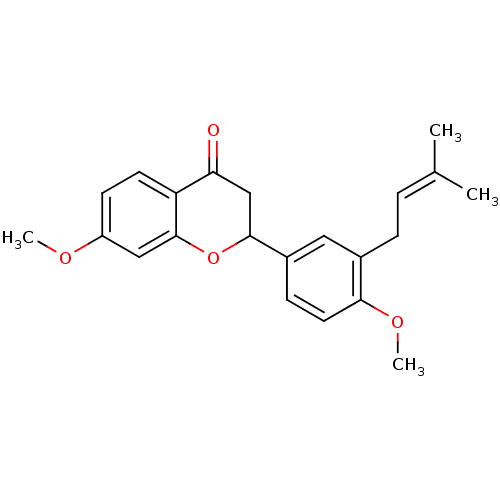
(7-methoxy-2-[4-methoxy-3-(3-methylbut-2-enyl)pheny...)Show SMILES [#6]-[#8]-c1ccc2-[#6](=O)-[#6]-[#6](-[#8]-c2c1)-c1ccc(-[#8]-[#6])c(-[#6]\[#6]=[#6](\[#6])-[#6])c1 Show InChI InChI=1S/C22H24O4/c1-14(2)5-6-15-11-16(7-10-20(15)25-4)21-13-19(23)18-9-8-17(24-3)12-22(18)26-21/h5,7-12,21H,6,13H2,1-4H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.67E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of human aromatase |
J Med Chem 50: 2799-806 (2007)
Article DOI: 10.1021/jm070109i
BindingDB Entry DOI: 10.7270/Q2668F0Z |
More data for this
Ligand-Target Pair | |
Aromatase
(Homo sapiens (Human)) | BDBM29210
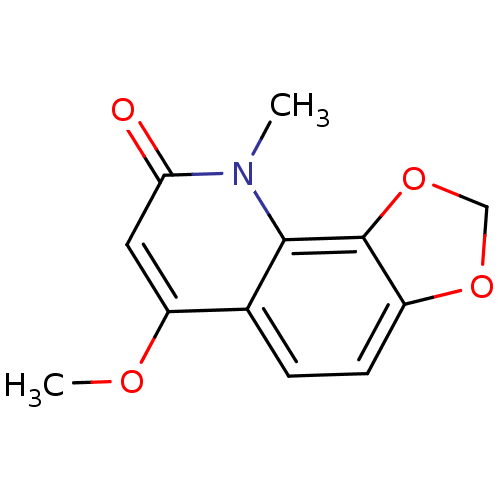
(casimiroin)Show InChI InChI=1S/C12H11NO4/c1-13-10(14)5-9(15-2)7-3-4-8-12(11(7)13)17-6-16-8/h3-5H,6H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 3.92E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
| Assay Description
Aromatase inhibition was quantified by measuring the fluorescent intensity of fluorescein, the hydrolysis product of dibenzylfluorescein (DBF), by ar... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | |
Aromatase
(Homo sapiens (Human)) | BDBM50213239
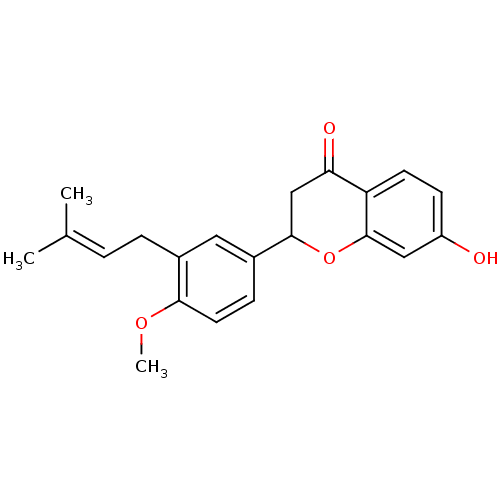
(7-Hydroxy-2-[4'-methoxy-3'-(3-methylbut-2-enyl)phe...)Show SMILES [#6]-[#8]-c1ccc(cc1-[#6]\[#6]=[#6](\[#6])-[#6])-[#6]-1-[#6]-[#6](=O)-c2ccc(-[#8])cc2-[#8]-1 Show InChI InChI=1S/C21H22O4/c1-13(2)4-5-14-10-15(6-9-19(14)24-3)20-12-18(23)17-8-7-16(22)11-21(17)25-20/h4,6-11,20,22H,5,12H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4.08E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of human aromatase |
J Med Chem 50: 2799-806 (2007)
Article DOI: 10.1021/jm070109i
BindingDB Entry DOI: 10.7270/Q2668F0Z |
More data for this
Ligand-Target Pair | |
Ribosyldihydronicotinamide dehydrogenase [quinone]
(Homo sapiens (Human)) | BDBM29223

(casimiroin analogue, 1l)Show InChI InChI=1S/C13H15NO3/c1-8-7-11(15)14(2)13-10(17-4)6-5-9(16-3)12(8)13/h5-7H,1-4H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 4.10E+3 | n/a | n/a | n/a | n/a | 7.5 | 23 |
Purdue University
| Assay Description
The activity of QR2 under steady-state conditions was evaluated on SpectraMax Plus 384 UV/vis spectrophotometer by monitoring the increase in absorba... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Aromatase
(Homo sapiens (Human)) | BDBM50213241
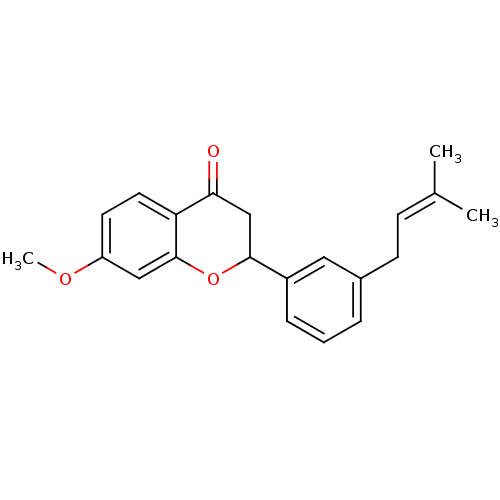
(7-methoxy-2-[3-(3-methylbut-2-enyl)phenyl]chroman-...)Show SMILES [#6]-[#8]-c1ccc2-[#6](=O)-[#6]-[#6](-[#8]-c2c1)-c1cccc(-[#6]\[#6]=[#6](\[#6])-[#6])c1 Show InChI InChI=1S/C21H22O3/c1-14(2)7-8-15-5-4-6-16(11-15)20-13-19(22)18-10-9-17(23-3)12-21(18)24-20/h4-7,9-12,20H,8,13H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4.67E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of human aromatase |
J Med Chem 50: 2799-806 (2007)
Article DOI: 10.1021/jm070109i
BindingDB Entry DOI: 10.7270/Q2668F0Z |
More data for this
Ligand-Target Pair | |
Aromatase
(Homo sapiens (Human)) | BDBM50213236
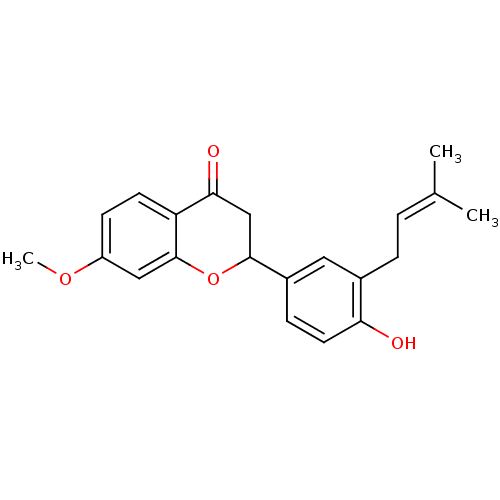
(2-[4-hydroxy-3-(3-methylbut-2-enyl)phenyl]-7-metho...)Show SMILES [#6]-[#8]-c1ccc2-[#6](=O)-[#6]-[#6](-[#8]-c2c1)-c1ccc(-[#8])c(-[#6]\[#6]=[#6](\[#6])-[#6])c1 Show InChI InChI=1S/C21H22O4/c1-13(2)4-5-14-10-15(6-9-18(14)22)20-12-19(23)17-8-7-16(24-3)11-21(17)25-20/h4,6-11,20,22H,5,12H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4.75E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of human aromatase |
J Med Chem 50: 2799-806 (2007)
Article DOI: 10.1021/jm070109i
BindingDB Entry DOI: 10.7270/Q2668F0Z |
More data for this
Ligand-Target Pair | |
Aromatase
(Homo sapiens (Human)) | BDBM29228
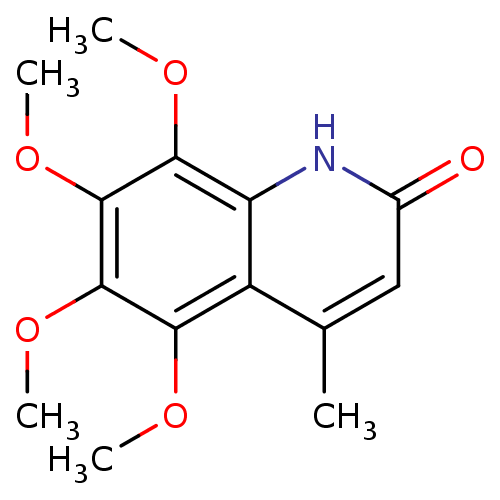
(casimiroin analogue, 1q)Show InChI InChI=1S/C14H17NO5/c1-7-6-8(16)15-10-9(7)11(17-2)13(19-4)14(20-5)12(10)18-3/h6H,1-5H3,(H,15,16) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.68E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
| Assay Description
Aromatase inhibition was quantified by measuring the fluorescent intensity of fluorescein, the hydrolysis product of dibenzylfluorescein (DBF), by ar... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | |
Aromatase
(Homo sapiens (Human)) | BDBM29219

(casimiroin analogue, 1h)Show InChI InChI=1S/C13H15NO3/c1-8-7-11(15)14(2)12-9(8)5-6-10(16-3)13(12)17-4/h5-7H,1-4H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.76E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
| Assay Description
Aromatase inhibition was quantified by measuring the fluorescent intensity of fluorescein, the hydrolysis product of dibenzylfluorescein (DBF), by ar... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | |
Ribosyldihydronicotinamide dehydrogenase [quinone]
(Homo sapiens (Human)) | BDBM29217

(casimiroin analogue, 1f)Show InChI InChI=1S/C12H13NO2/c1-8-7-11(14)13(2)12-9(8)5-4-6-10(12)15-3/h4-7H,1-3H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.80E+3 | n/a | n/a | n/a | n/a | 7.5 | 23 |
Purdue University
| Assay Description
The activity of QR2 under steady-state conditions was evaluated on SpectraMax Plus 384 UV/vis spectrophotometer by monitoring the increase in absorba... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | |
Ribosyldihydronicotinamide dehydrogenase [quinone]
(Homo sapiens (Human)) | BDBM29226
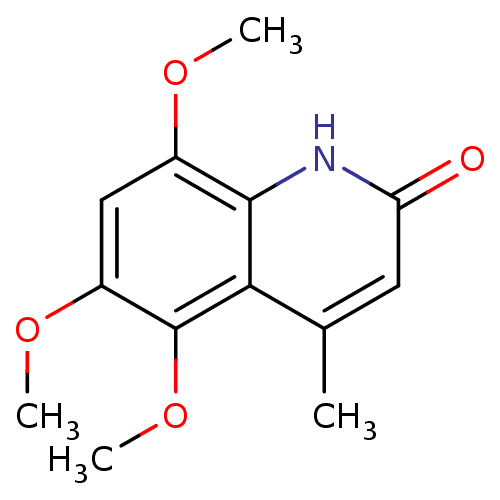
(casimiroin analogue, 1o)Show InChI InChI=1S/C13H15NO4/c1-7-5-10(15)14-12-8(16-2)6-9(17-3)13(18-4)11(7)12/h5-6H,1-4H3,(H,14,15) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 6.00E+3 | n/a | n/a | n/a | n/a | 7.5 | 23 |
Purdue University
| Assay Description
The activity of QR2 under steady-state conditions was evaluated on SpectraMax Plus 384 UV/vis spectrophotometer by monitoring the increase in absorba... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Ribosyldihydronicotinamide dehydrogenase [quinone]
(Homo sapiens (Human)) | BDBM29213

(casimiroin analogue, 1b)Show InChI InChI=1S/C12H11NO3/c1-7-5-10(14)13(2)11-8(7)3-4-9-12(11)16-6-15-9/h3-5H,6H2,1-2H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 6.20E+3 | n/a | n/a | n/a | n/a | 7.5 | 23 |
Purdue University
| Assay Description
The activity of QR2 under steady-state conditions was evaluated on SpectraMax Plus 384 UV/vis spectrophotometer by monitoring the increase in absorba... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Ribosyldihydronicotinamide dehydrogenase [quinone]
(Homo sapiens (Human)) | BDBM29225

(casimiroin analogue, 1n)Show InChI InChI=1S/C14H17NO4/c1-8-6-11(16)15(2)12-9(8)7-10(17-3)13(18-4)14(12)19-5/h6-7H,1-5H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 7.00E+3 | n/a | n/a | n/a | n/a | 7.5 | 23 |
Purdue University
| Assay Description
The activity of QR2 under steady-state conditions was evaluated on SpectraMax Plus 384 UV/vis spectrophotometer by monitoring the increase in absorba... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Aromatase
(Homo sapiens (Human)) | BDBM50213252
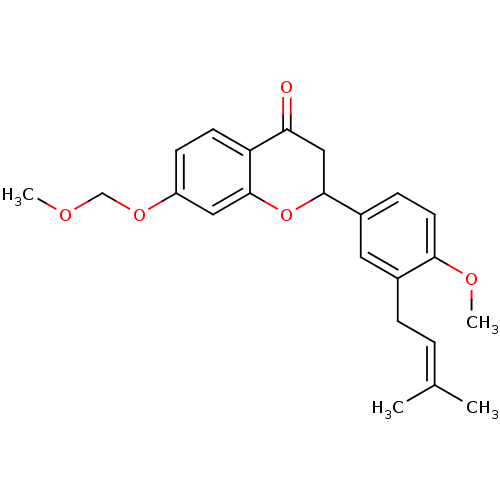
(2-[4-methoxy-3-(3-methylbut-2-enyl)phenyl]-7-(meth...)Show SMILES [#6]-[#8]-[#6]-[#8]-c1ccc2-[#6](=O)-[#6]-[#6](-[#8]-c2c1)-c1ccc(-[#8]-[#6])c(-[#6]\[#6]=[#6](\[#6])-[#6])c1 Show InChI InChI=1S/C23H26O5/c1-15(2)5-6-16-11-17(7-10-21(16)26-4)22-13-20(24)19-9-8-18(27-14-25-3)12-23(19)28-22/h5,7-12,22H,6,13-14H2,1-4H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 7.67E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of human aromatase |
J Med Chem 50: 2799-806 (2007)
Article DOI: 10.1021/jm070109i
BindingDB Entry DOI: 10.7270/Q2668F0Z |
More data for this
Ligand-Target Pair | |
Ribosyldihydronicotinamide dehydrogenase [quinone]
(Homo sapiens (Human)) | BDBM29220
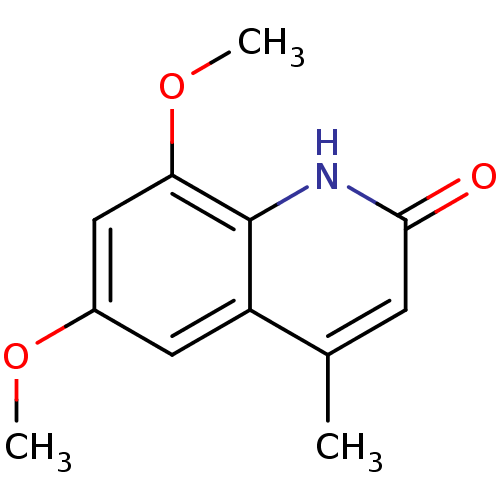
(casimiroin analogue, 1i)Show InChI InChI=1S/C12H13NO3/c1-7-4-11(14)13-12-9(7)5-8(15-2)6-10(12)16-3/h4-6H,1-3H3,(H,13,14) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8.80E+3 | n/a | n/a | n/a | n/a | 7.5 | 23 |
Purdue University
| Assay Description
The activity of QR2 under steady-state conditions was evaluated on SpectraMax Plus 384 UV/vis spectrophotometer by monitoring the increase in absorba... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | |
Ribosyldihydronicotinamide dehydrogenase [quinone]
(Homo sapiens (Human)) | BDBM29219

(casimiroin analogue, 1h)Show InChI InChI=1S/C13H15NO3/c1-8-7-11(15)14(2)12-9(8)5-6-10(16-3)13(12)17-4/h5-7H,1-4H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 9.30E+3 | n/a | n/a | n/a | n/a | 7.5 | 23 |
Purdue University
| Assay Description
The activity of QR2 under steady-state conditions was evaluated on SpectraMax Plus 384 UV/vis spectrophotometer by monitoring the increase in absorba... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Ribosyldihydronicotinamide dehydrogenase [quinone]
(Homo sapiens (Human)) | BDBM29224

(casimiroin analogue, 1m)Show InChI InChI=1S/C13H15NO4/c1-7-5-10(15)14-11-8(7)6-9(16-2)12(17-3)13(11)18-4/h5-6H,1-4H3,(H,14,15) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | 7.5 | 23 |
Purdue University
| Assay Description
The activity of QR2 under steady-state conditions was evaluated on SpectraMax Plus 384 UV/vis spectrophotometer by monitoring the increase in absorba... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Ribosyldihydronicotinamide dehydrogenase [quinone]
(Homo sapiens (Human)) | BDBM29227

(casimiroin analogue, 1p)Show InChI InChI=1S/C14H17NO4/c1-8-6-11(16)15(2)13-9(17-3)7-10(18-4)14(19-5)12(8)13/h6-7H,1-5H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 1.08E+4 | n/a | n/a | n/a | n/a | 7.5 | 23 |
Purdue University
| Assay Description
The activity of QR2 under steady-state conditions was evaluated on SpectraMax Plus 384 UV/vis spectrophotometer by monitoring the increase in absorba... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Ribosyldihydronicotinamide dehydrogenase [quinone]
(Homo sapiens (Human)) | BDBM29222

(casimiroin analogue, 1k)Show InChI InChI=1S/C12H13NO3/c1-7-6-10(14)13-12-9(16-3)5-4-8(15-2)11(7)12/h4-6H,1-3H3,(H,13,14) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 1.08E+4 | n/a | n/a | n/a | n/a | 7.5 | 23 |
Purdue University
| Assay Description
The activity of QR2 under steady-state conditions was evaluated on SpectraMax Plus 384 UV/vis spectrophotometer by monitoring the increase in absorba... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Ribosyldihydronicotinamide dehydrogenase [quinone]
(Homo sapiens (Human)) | BDBM29212

(casimiroin analogue, 1a)Show InChI InChI=1S/C11H9NO3/c1-6-4-9(13)12-10-7(6)2-3-8-11(10)15-5-14-8/h2-4H,5H2,1H3,(H,12,13) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 1.08E+4 | n/a | n/a | n/a | n/a | 7.5 | 23 |
Purdue University
| Assay Description
The activity of QR2 under steady-state conditions was evaluated on SpectraMax Plus 384 UV/vis spectrophotometer by monitoring the increase in absorba... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Ribosyldihydronicotinamide dehydrogenase [quinone]
(Homo sapiens (Human)) | BDBM9019
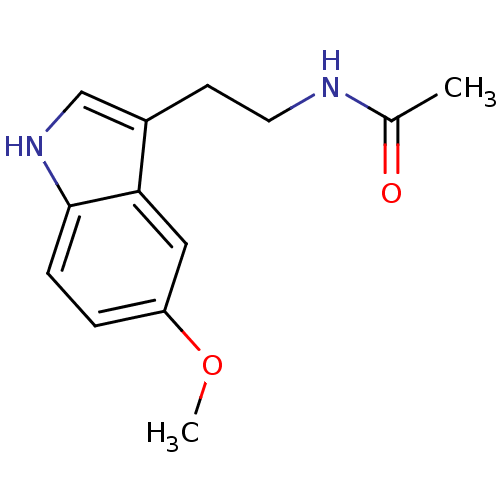
(CHEMBL45 | Melatonin | N-[2-(5-methoxy-1H-indol-3-...)Show InChI InChI=1S/C13H16N2O2/c1-9(16)14-6-5-10-8-15-13-4-3-11(17-2)7-12(10)13/h3-4,7-8,15H,5-6H2,1-2H3,(H,14,16) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 1.13E+4 | n/a | n/a | n/a | n/a | 7.5 | 23 |
Purdue University
| Assay Description
The activity of QR2 under steady-state conditions was evaluated on SpectraMax Plus 384 UV/vis spectrophotometer by monitoring the increase in absorba... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Aromatase
(Homo sapiens (Human)) | BDBM50370985
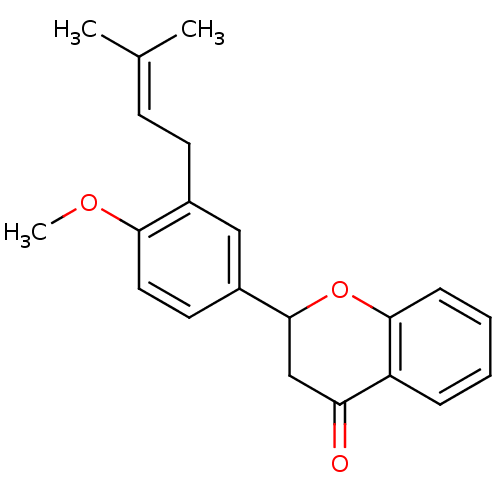
(CHEMBL1162951)Show SMILES [#6]-[#8]-c1ccc(cc1-[#6]\[#6]=[#6](\[#6])-[#6])-[#6]-1-[#6]-[#6](=O)-c2ccccc2-[#8]-1 Show InChI InChI=1S/C21H22O3/c1-14(2)8-9-15-12-16(10-11-19(15)23-3)21-13-18(22)17-6-4-5-7-20(17)24-21/h4-8,10-12,21H,9,13H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.21E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of human aromatase |
J Med Chem 50: 2799-806 (2007)
Article DOI: 10.1021/jm070109i
BindingDB Entry DOI: 10.7270/Q2668F0Z |
More data for this
Ligand-Target Pair | |
Aromatase
(Homo sapiens (Human)) | BDBM50213234

(7-(methoxymethoxy)-2-[4-(methoxymethoxy)-3-(3-meth...)Show SMILES [#6]-[#8]-[#6]-[#8]-c1ccc2-[#6](=O)-[#6]-[#6](-[#8]-c2c1)-c1ccc(-[#8]-[#6]-[#8]-[#6])c(-[#6]\[#6]=[#6](\[#6])-[#6])c1 Show InChI InChI=1S/C24H28O6/c1-16(2)5-6-17-11-18(7-10-22(17)29-15-27-4)23-13-21(25)20-9-8-19(28-14-26-3)12-24(20)30-23/h5,7-12,23H,6,13-15H2,1-4H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of human aromatase |
J Med Chem 50: 2799-806 (2007)
Article DOI: 10.1021/jm070109i
BindingDB Entry DOI: 10.7270/Q2668F0Z |
More data for this
Ligand-Target Pair | |
Aromatase
(Homo sapiens (Human)) | BDBM29226

(casimiroin analogue, 1o)Show InChI InChI=1S/C13H15NO4/c1-7-5-10(15)14-12-8(16-2)6-9(17-3)13(18-4)11(7)12/h5-6H,1-4H3,(H,14,15) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.48E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
| Assay Description
Aromatase inhibition was quantified by measuring the fluorescent intensity of fluorescein, the hydrolysis product of dibenzylfluorescein (DBF), by ar... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | |
Ribosyldihydronicotinamide dehydrogenase [quinone]
(Homo sapiens (Human)) | BDBM29215
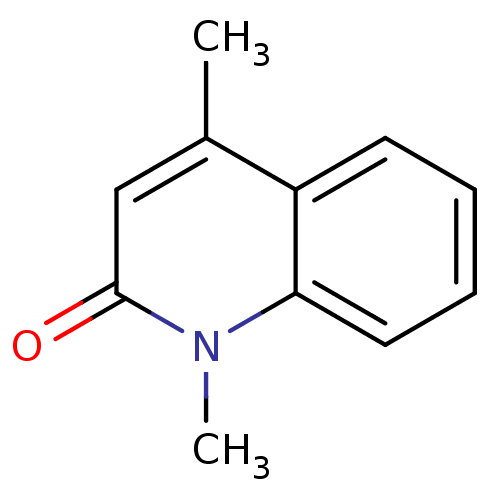
(casimiroin analogue, 1d)Show InChI InChI=1S/C11H11NO/c1-8-7-11(13)12(2)10-6-4-3-5-9(8)10/h3-7H,1-2H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 1.82E+4 | n/a | n/a | n/a | n/a | 7.5 | 23 |
Purdue University
| Assay Description
The activity of QR2 under steady-state conditions was evaluated on SpectraMax Plus 384 UV/vis spectrophotometer by monitoring the increase in absorba... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Aromatase
(Homo sapiens (Human)) | BDBM29224

(casimiroin analogue, 1m)Show InChI InChI=1S/C13H15NO4/c1-7-5-10(15)14-11-8(7)6-9(16-2)12(17-3)13(11)18-4/h5-6H,1-4H3,(H,14,15) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.85E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
| Assay Description
Aromatase inhibition was quantified by measuring the fluorescent intensity of fluorescein, the hydrolysis product of dibenzylfluorescein (DBF), by ar... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | |
Ribosyldihydronicotinamide dehydrogenase [quinone]
(Homo sapiens (Human)) | BDBM29216
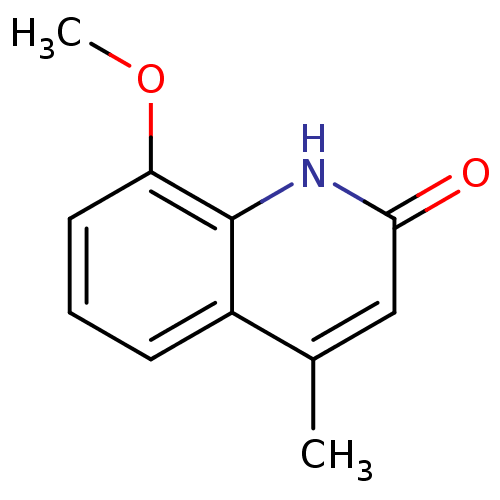
(casimiroin analogue, 1e)Show InChI InChI=1S/C11H11NO2/c1-7-6-10(13)12-11-8(7)4-3-5-9(11)14-2/h3-6H,1-2H3,(H,12,13) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 2.41E+4 | n/a | n/a | n/a | n/a | 7.5 | 23 |
Purdue University
| Assay Description
The activity of QR2 under steady-state conditions was evaluated on SpectraMax Plus 384 UV/vis spectrophotometer by monitoring the increase in absorba... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Aromatase
(Homo sapiens (Human)) | BDBM50213243
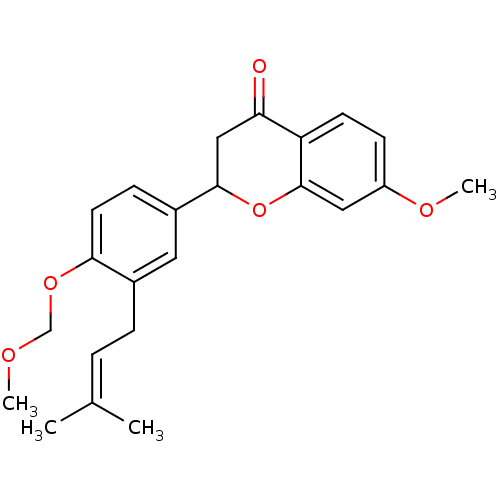
(7-methoxy-2-[4-(methoxymethoxy)-3-(3-methylbut-2-e...)Show SMILES [#6]-[#8]-[#6]-[#8]-c1ccc(cc1-[#6]\[#6]=[#6](\[#6])-[#6])-[#6]-1-[#6]-[#6](=O)-c2ccc(-[#8]-[#6])cc2-[#8]-1 Show InChI InChI=1S/C23H26O5/c1-15(2)5-6-16-11-17(7-10-21(16)27-14-25-3)22-13-20(24)19-9-8-18(26-4)12-23(19)28-22/h5,7-12,22H,6,13-14H2,1-4H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.51E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of human aromatase |
J Med Chem 50: 2799-806 (2007)
Article DOI: 10.1021/jm070109i
BindingDB Entry DOI: 10.7270/Q2668F0Z |
More data for this
Ligand-Target Pair | |
Aromatase
(Homo sapiens (Human)) | BDBM29222

(casimiroin analogue, 1k)Show InChI InChI=1S/C12H13NO3/c1-7-6-10(14)13-12-9(16-3)5-4-8(15-2)11(7)12/h4-6H,1-3H3,(H,13,14) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.68E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
| Assay Description
Aromatase inhibition was quantified by measuring the fluorescent intensity of fluorescein, the hydrolysis product of dibenzylfluorescein (DBF), by ar... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | |
Aromatase
(Homo sapiens (Human)) | BDBM50213238

(7-methoxy-2-[4-(3-methylbut-2-enyloxy)phenyl]chrom...)Show SMILES [#6]-[#8]-c1ccc2-[#6](=O)-[#6]-[#6](-[#8]-c2c1)-c1ccc(-[#8]-[#6]\[#6]=[#6](\[#6])-[#6])cc1 Show InChI InChI=1S/C21H22O4/c1-14(2)10-11-24-16-6-4-15(5-7-16)20-13-19(22)18-9-8-17(23-3)12-21(18)25-20/h4-10,12,20H,11,13H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.87E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of human aromatase |
J Med Chem 50: 2799-806 (2007)
Article DOI: 10.1021/jm070109i
BindingDB Entry DOI: 10.7270/Q2668F0Z |
More data for this
Ligand-Target Pair | |
Ribosyldihydronicotinamide dehydrogenase [quinone]
(Homo sapiens (Human)) | BDBM29218

(casimiroin analogue, 1g)Show InChI InChI=1S/C12H13NO3/c1-7-6-10(14)13-11-8(7)4-5-9(15-2)12(11)16-3/h4-6H,1-3H3,(H,13,14) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.93E+4 | n/a | n/a | n/a | n/a | 7.5 | 23 |
Purdue University
| Assay Description
The activity of QR2 under steady-state conditions was evaluated on SpectraMax Plus 384 UV/vis spectrophotometer by monitoring the increase in absorba... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | |
Aromatase
(Homo sapiens (Human)) | BDBM29218

(casimiroin analogue, 1g)Show InChI InChI=1S/C12H13NO3/c1-7-6-10(14)13-11-8(7)4-5-9(15-2)12(11)16-3/h4-6H,1-3H3,(H,13,14) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.15E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
| Assay Description
Aromatase inhibition was quantified by measuring the fluorescent intensity of fluorescein, the hydrolysis product of dibenzylfluorescein (DBF), by ar... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | |
Aromatase
(Homo sapiens (Human)) | BDBM50213251

((rac)-7-hydroxy-2-(4-hydroxy-3-(3-methylbut-2-enyl...)Show SMILES [#6]\[#6](-[#6])=[#6]\[#6]-c1cc(ccc1-[#8])-[#6]-1-[#6]-[#6](=O)-c2ccc(-[#8])cc2-[#8]-1 Show InChI InChI=1S/C20H20O4/c1-12(2)3-4-13-9-14(5-8-17(13)22)19-11-18(23)16-7-6-15(21)10-20(16)24-19/h3,5-10,19,21-22H,4,11H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of human aromatase |
J Med Chem 50: 2799-806 (2007)
Article DOI: 10.1021/jm070109i
BindingDB Entry DOI: 10.7270/Q2668F0Z |
More data for this
Ligand-Target Pair | |
Ribosyldihydronicotinamide dehydrogenase [quinone]
(Homo sapiens (Human)) | BDBM29210

(casimiroin)Show InChI InChI=1S/C12H11NO4/c1-13-10(14)5-9(15-2)7-3-4-8-12(11(7)13)17-6-16-8/h3-5H,6H2,1-2H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
| MMDB
PDB
Article
PubMed
| n/a | n/a | 5.41E+4 | n/a | n/a | n/a | n/a | 7.5 | 23 |
Purdue University
| Assay Description
The activity of QR2 under steady-state conditions was evaluated on SpectraMax Plus 384 UV/vis spectrophotometer by monitoring the increase in absorba... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Aromatase
(Homo sapiens (Human)) | BDBM50213248

(3-hydroxy-1-(2-hydroxy-4-methoxyphenyl)-3-[4-metho...)Show SMILES [#6]-[#8]-c1ccc(-[#6](=O)-[#6]-[#6](-[#8])-c2ccc(-[#8]-[#6])c(-[#6]\[#6]=[#6](\[#6])-[#6])c2)c(-[#8])c1 Show InChI InChI=1S/C22H26O5/c1-14(2)5-6-16-11-15(7-10-22(16)27-4)19(23)13-21(25)18-9-8-17(26-3)12-20(18)24/h5,7-12,19,23-24H,6,13H2,1-4H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 5.47E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of human aromatase |
J Med Chem 50: 2799-806 (2007)
Article DOI: 10.1021/jm070109i
BindingDB Entry DOI: 10.7270/Q2668F0Z |
More data for this
Ligand-Target Pair | |
Aromatase
(Homo sapiens (Human)) | BDBM50213240
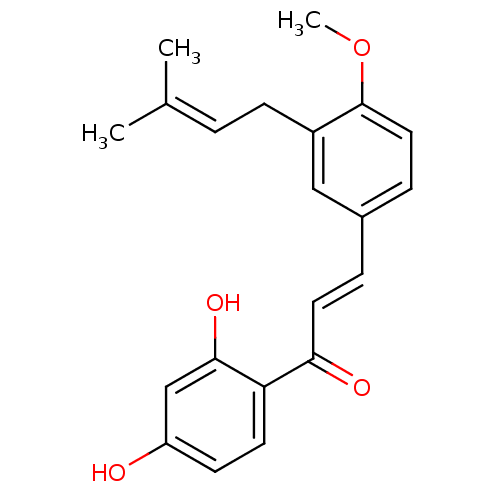
(1-(2,4-Dihydroxyphenyl)-3-[4-methoxy-3-(3-methylbu...)Show SMILES [#6]-[#8]-c1ccc(\[#6]=[#6]\[#6](=O)-c2ccc(-[#8])cc2-[#8])cc1-[#6]\[#6]=[#6](\[#6])-[#6] Show InChI InChI=1S/C21H22O4/c1-14(2)4-7-16-12-15(6-11-21(16)25-3)5-10-19(23)18-9-8-17(22)13-20(18)24/h4-6,8-13,22,24H,7H2,1-3H3/b10-5+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 8.29E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
Curated by ChEMBL
| Assay Description
Inhibition of human aromatase |
J Med Chem 50: 2799-806 (2007)
Article DOI: 10.1021/jm070109i
BindingDB Entry DOI: 10.7270/Q2668F0Z |
More data for this
Ligand-Target Pair | |
Aromatase
(Homo sapiens (Human)) | BDBM29220

(casimiroin analogue, 1i)Show InChI InChI=1S/C12H13NO3/c1-7-4-11(14)13-12-9(7)5-8(15-2)6-10(12)16-3/h4-6H,1-3H3,(H,13,14) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >9.13E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
| Assay Description
Aromatase inhibition was quantified by measuring the fluorescent intensity of fluorescein, the hydrolysis product of dibenzylfluorescein (DBF), by ar... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | |
Aromatase
(Homo sapiens (Human)) | BDBM29230
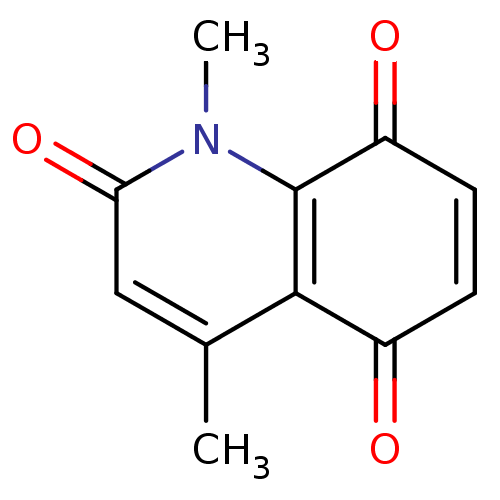
(quinone, 12)Show InChI InChI=1S/C11H9NO3/c1-6-5-9(15)12(2)11-8(14)4-3-7(13)10(6)11/h3-5H,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >9.84E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Purdue University
| Assay Description
Aromatase inhibition was quantified by measuring the fluorescent intensity of fluorescein, the hydrolysis product of dibenzylfluorescein (DBF), by ar... |
J Med Chem 52: 1873-84 (2009)
Article DOI: 10.1021/jm801335z
BindingDB Entry DOI: 10.7270/Q2NP22RV |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data