 Found 403 hits with Last Name = 'davis' and Initial = 'ja'
Found 403 hits with Last Name = 'davis' and Initial = 'ja' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Type-1 angiotensin II receptor
(Homo sapiens (Human)) | BDBM50347563

(CHEMBL1801740)Show SMILES CCc1nc2c(C)cc(CC(C)C)nc2n1[C@H]1CCc2cc(ccc12)-c1ccccc1-c1nnn[nH]1 |r| Show InChI InChI=1S/C29H31N7/c1-5-26-31-27-18(4)15-21(14-17(2)3)30-29(27)36(26)25-13-11-20-16-19(10-12-23(20)25)22-8-6-7-9-24(22)28-32-34-35-33-28/h6-10,12,15-17,25H,5,11,13-14H2,1-4H3,(H,32,33,34,35)/t25-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.690 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Binding affinity to human Angiotensin receptor 1 |
J Med Chem 54: 4219-33 (2011)
Article DOI: 10.1021/jm200409s
BindingDB Entry DOI: 10.7270/Q2SB463J |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50251493

((2S,4S)-1-(2-(1-(4-cyano-3,5-difluorophenyl)piperi...)Show SMILES F[C@H]1C[C@@H](C#N)N(C1)C(=O)CNC1CCN(CC1)c1cc(F)c(C#N)c(F)c1 |r| Show InChI InChI=1S/C19H20F3N5O/c20-12-5-15(8-23)27(11-12)19(28)10-25-13-1-3-26(4-2-13)14-6-17(21)16(9-24)18(22)7-14/h6-7,12-13,15,25H,1-5,10-11H2/t12-,15-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human plasma DPP4 |
Bioorg Med Chem Lett 18: 4087-91 (2008)
Article DOI: 10.1016/j.bmcl.2008.05.101
BindingDB Entry DOI: 10.7270/Q2T153FC |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50251494
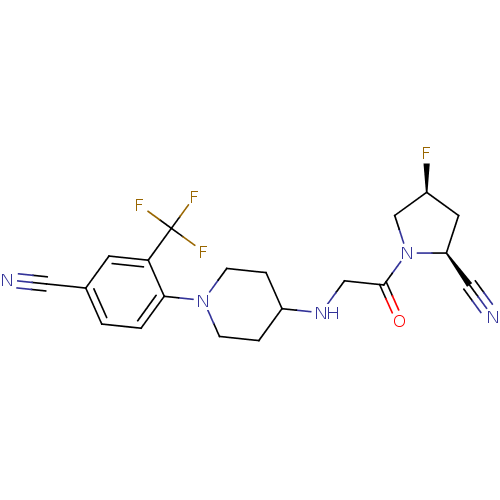
((2S,4S)-1-(2-(1-(4-cyano-2-(trifluoromethyl)phenyl...)Show SMILES F[C@H]1C[C@@H](C#N)N(C1)C(=O)CNC1CCN(CC1)c1ccc(cc1C(F)(F)F)C#N |r| Show InChI InChI=1S/C20H21F4N5O/c21-14-8-16(10-26)29(12-14)19(30)11-27-15-3-5-28(6-4-15)18-2-1-13(9-25)7-17(18)20(22,23)24/h1-2,7,14-16,27H,3-6,8,11-12H2/t14-,16-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human plasma DPP4 |
Bioorg Med Chem Lett 18: 4087-91 (2008)
Article DOI: 10.1016/j.bmcl.2008.05.101
BindingDB Entry DOI: 10.7270/Q2T153FC |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50251495
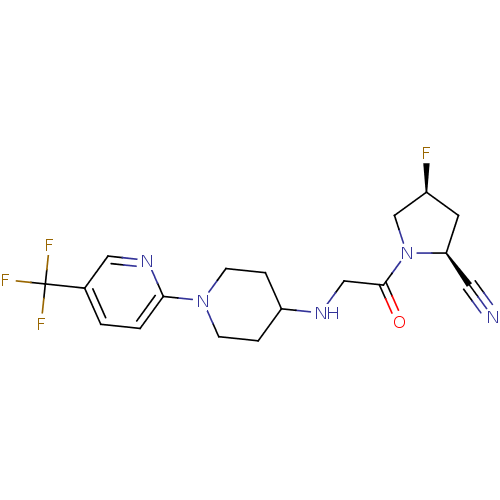
((2S,4S)-4-fluoro-1-(2-(1-(5-(trifluoromethyl)pyrid...)Show SMILES F[C@H]1C[C@@H](C#N)N(C1)C(=O)CNC1CCN(CC1)c1ccc(cn1)C(F)(F)F |r| Show InChI InChI=1S/C18H21F4N5O/c19-13-7-15(8-23)27(11-13)17(28)10-24-14-3-5-26(6-4-14)16-2-1-12(9-25-16)18(20,21)22/h1-2,9,13-15,24H,3-7,10-11H2/t13-,15-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human plasma DPP4 |
Bioorg Med Chem Lett 18: 4087-91 (2008)
Article DOI: 10.1016/j.bmcl.2008.05.101
BindingDB Entry DOI: 10.7270/Q2T153FC |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50251517
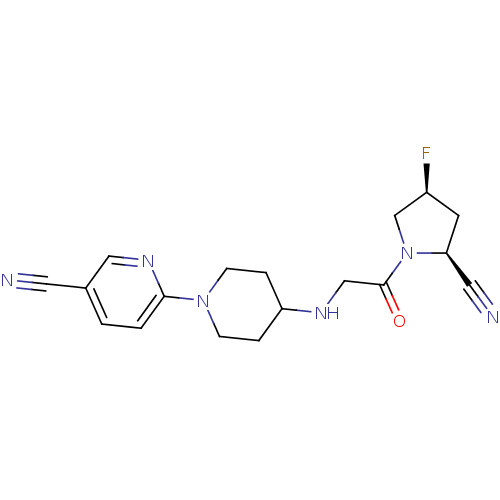
(6-(4-(2-((2S,4S)-2-cyano-4-fluoropyrrolidin-1-yl)-...)Show SMILES F[C@H]1C[C@@H](C#N)N(C1)C(=O)CNC1CCN(CC1)c1ccc(cn1)C#N |r| Show InChI InChI=1S/C18H21FN6O/c19-14-7-16(9-21)25(12-14)18(26)11-22-15-3-5-24(6-4-15)17-2-1-13(8-20)10-23-17/h1-2,10,14-16,22H,3-7,11-12H2/t14-,16-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 31 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human plasma DPP4 |
Bioorg Med Chem Lett 18: 4087-91 (2008)
Article DOI: 10.1016/j.bmcl.2008.05.101
BindingDB Entry DOI: 10.7270/Q2T153FC |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50251518
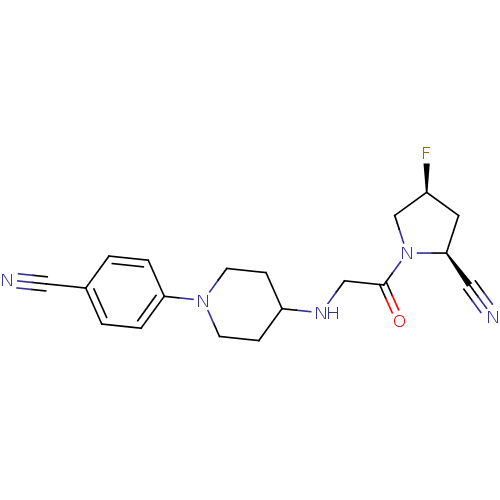
((2S,4S)-1-(2-(1-(4-cyanophenyl)piperidin-4-ylamino...)Show SMILES F[C@H]1C[C@@H](C#N)N(C1)C(=O)CNC1CCN(CC1)c1ccc(cc1)C#N |r| Show InChI InChI=1S/C19H22FN5O/c20-15-9-18(11-22)25(13-15)19(26)12-23-16-5-7-24(8-6-16)17-3-1-14(10-21)2-4-17/h1-4,15-16,18,23H,5-9,12-13H2/t15-,18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 79 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human plasma DPP4 |
Bioorg Med Chem Lett 18: 4087-91 (2008)
Article DOI: 10.1016/j.bmcl.2008.05.101
BindingDB Entry DOI: 10.7270/Q2T153FC |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50363928
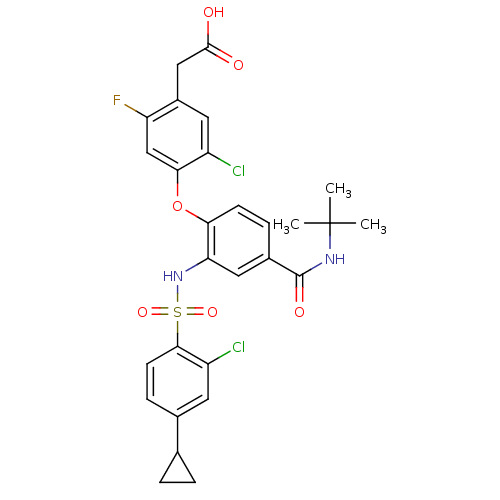
(CHEMBL1951575)Show SMILES CC(C)(C)NC(=O)c1ccc(Oc2cc(F)c(CC(O)=O)cc2Cl)c(NS(=O)(=O)c2ccc(cc2Cl)C2CC2)c1 Show InChI InChI=1S/C28H27Cl2FN2O6S/c1-28(2,3)32-27(36)17-6-8-23(39-24-14-21(31)18(11-19(24)29)13-26(34)35)22(12-17)33-40(37,38)25-9-7-16(10-20(25)30)15-4-5-15/h6-12,14-15,33H,4-5,13H2,1-3H3,(H,32,36)(H,34,35) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen, Inc
Curated by ChEMBL
| Assay Description
Competitive inhibition of CYP2C8 in human liver microsomes assessed as paclitaxel 6-hydroxylation after 20 mins by LC-MS/MS analysis |
Drug Metab Dispos 40: 2239-49 (2012)
Article DOI: 10.1124/dmd.112.047928
BindingDB Entry DOI: 10.7270/Q2Z039WT |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50363928

(CHEMBL1951575)Show SMILES CC(C)(C)NC(=O)c1ccc(Oc2cc(F)c(CC(O)=O)cc2Cl)c(NS(=O)(=O)c2ccc(cc2Cl)C2CC2)c1 Show InChI InChI=1S/C28H27Cl2FN2O6S/c1-28(2,3)32-27(36)17-6-8-23(39-24-14-21(31)18(11-19(24)29)13-26(34)35)22(12-17)33-40(37,38)25-9-7-16(10-20(25)30)15-4-5-15/h6-12,14-15,33H,4-5,13H2,1-3H3,(H,32,36)(H,34,35) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen, Inc
Curated by ChEMBL
| Assay Description
Biphasic inhibition of CYP2C8 in human liver microsomes assessed as montelukast 36-hydroxylation after 20 mins by LC-MS/MS analysis |
Drug Metab Dispos 40: 2239-49 (2012)
Article DOI: 10.1124/dmd.112.047928
BindingDB Entry DOI: 10.7270/Q2Z039WT |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50088490

(CHEMBL3526979)Show SMILES CC(C)(C)NC(=O)c1ccc(Oc2cc(F)c(CC(=O)O[C@@H]3O[C@@H]([C@@H](O)[C@H](O)[C@H]3O)C(O)=O)cc2Cl)c(NS(=O)(=O)c2ccc(cc2Cl)C2CC2)c1 |r| Show InChI InChI=1S/C34H35Cl2FN2O12S/c1-34(2,3)38-31(44)17-6-8-23(22(12-17)39-52(47,48)25-9-7-16(10-20(25)36)15-4-5-15)49-24-14-21(37)18(11-19(24)35)13-26(40)50-33-29(43)27(41)28(42)30(51-33)32(45)46/h6-12,14-15,27-30,33,39,41-43H,4-5,13H2,1-3H3,(H,38,44)(H,45,46)/t27-,28-,29+,30-,33+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen, Inc
Curated by ChEMBL
| Assay Description
Linear mixed inhibition of CYP2C8 in human liver microsomes assessed as paclitaxel 6-hydroxylation after 20 mins by LC-MS/MS analysis |
Drug Metab Dispos 40: 2239-49 (2012)
Article DOI: 10.1124/dmd.112.047928
BindingDB Entry DOI: 10.7270/Q2Z039WT |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50363928

(CHEMBL1951575)Show SMILES CC(C)(C)NC(=O)c1ccc(Oc2cc(F)c(CC(O)=O)cc2Cl)c(NS(=O)(=O)c2ccc(cc2Cl)C2CC2)c1 Show InChI InChI=1S/C28H27Cl2FN2O6S/c1-28(2,3)32-27(36)17-6-8-23(39-24-14-21(31)18(11-19(24)29)13-26(34)35)22(12-17)33-40(37,38)25-9-7-16(10-20(25)30)15-4-5-15/h6-12,14-15,33H,4-5,13H2,1-3H3,(H,32,36)(H,34,35) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 6.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen, Inc
Curated by ChEMBL
| Assay Description
Competitive inhibition of CYP2C8 in human liver microsomes assessed as rosiglitazone demethylation after 20 mins by LC-MS/MS analysis |
Drug Metab Dispos 40: 2239-49 (2012)
Article DOI: 10.1124/dmd.112.047928
BindingDB Entry DOI: 10.7270/Q2Z039WT |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50088490

(CHEMBL3526979)Show SMILES CC(C)(C)NC(=O)c1ccc(Oc2cc(F)c(CC(=O)O[C@@H]3O[C@@H]([C@@H](O)[C@H](O)[C@H]3O)C(O)=O)cc2Cl)c(NS(=O)(=O)c2ccc(cc2Cl)C2CC2)c1 |r| Show InChI InChI=1S/C34H35Cl2FN2O12S/c1-34(2,3)38-31(44)17-6-8-23(22(12-17)39-52(47,48)25-9-7-16(10-20(25)36)15-4-5-15)49-24-14-21(37)18(11-19(24)35)13-26(40)50-33-29(43)27(41)28(42)30(51-33)32(45)46/h6-12,14-15,27-30,33,39,41-43H,4-5,13H2,1-3H3,(H,38,44)(H,45,46)/t27-,28-,29+,30-,33+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 6.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen, Inc
Curated by ChEMBL
| Assay Description
Linear mixed inhibition of CYP2C8 in human liver microsomes assessed as rosiglitazone demethylation after 20 mins by LC-MS/MS analysis |
Drug Metab Dispos 40: 2239-49 (2012)
Article DOI: 10.1124/dmd.112.047928
BindingDB Entry DOI: 10.7270/Q2Z039WT |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50088490

(CHEMBL3526979)Show SMILES CC(C)(C)NC(=O)c1ccc(Oc2cc(F)c(CC(=O)O[C@@H]3O[C@@H]([C@@H](O)[C@H](O)[C@H]3O)C(O)=O)cc2Cl)c(NS(=O)(=O)c2ccc(cc2Cl)C2CC2)c1 |r| Show InChI InChI=1S/C34H35Cl2FN2O12S/c1-34(2,3)38-31(44)17-6-8-23(22(12-17)39-52(47,48)25-9-7-16(10-20(25)36)15-4-5-15)49-24-14-21(37)18(11-19(24)35)13-26(40)50-33-29(43)27(41)28(42)30(51-33)32(45)46/h6-12,14-15,27-30,33,39,41-43H,4-5,13H2,1-3H3,(H,38,44)(H,45,46)/t27-,28-,29+,30-,33+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 7.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen, Inc
Curated by ChEMBL
| Assay Description
Biphasic inhibition of CYP2C8 in human liver microsomes assessed as montelukast 36-hydroxylation after 20 mins by LC-MS/MS analysis |
Drug Metab Dispos 40: 2239-49 (2012)
Article DOI: 10.1124/dmd.112.047928
BindingDB Entry DOI: 10.7270/Q2Z039WT |
More data for this
Ligand-Target Pair | |
Type-2 angiotensin II receptor
(Homo sapiens (Human)) | BDBM50347563

(CHEMBL1801740)Show SMILES CCc1nc2c(C)cc(CC(C)C)nc2n1[C@H]1CCc2cc(ccc12)-c1ccccc1-c1nnn[nH]1 |r| Show InChI InChI=1S/C29H31N7/c1-5-26-31-27-18(4)15-21(14-17(2)3)30-29(27)36(26)25-13-11-20-16-19(10-12-23(20)25)22-8-6-7-9-24(22)28-32-34-35-33-28/h6-10,12,15-17,25H,5,11,13-14H2,1-4H3,(H,32,33,34,35)/t25-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Binding affinity to human Angiotensin receptor 2 |
J Med Chem 54: 4219-33 (2011)
Article DOI: 10.1021/jm200409s
BindingDB Entry DOI: 10.7270/Q2SB463J |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50363928

(CHEMBL1951575)Show SMILES CC(C)(C)NC(=O)c1ccc(Oc2cc(F)c(CC(O)=O)cc2Cl)c(NS(=O)(=O)c2ccc(cc2Cl)C2CC2)c1 Show InChI InChI=1S/C28H27Cl2FN2O6S/c1-28(2,3)32-27(36)17-6-8-23(39-24-14-21(31)18(11-19(24)29)13-26(34)35)22(12-17)33-40(37,38)25-9-7-16(10-20(25)30)15-4-5-15/h6-12,14-15,33H,4-5,13H2,1-3H3,(H,32,36)(H,34,35) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen, Inc
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 in human liver microsomes using testosterone as substrate after 5 to 30 mins by LC-MS/MS analysis |
Drug Metab Dispos 40: 2239-49 (2012)
Article DOI: 10.1124/dmd.112.047928
BindingDB Entry DOI: 10.7270/Q2Z039WT |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50088490

(CHEMBL3526979)Show SMILES CC(C)(C)NC(=O)c1ccc(Oc2cc(F)c(CC(=O)O[C@@H]3O[C@@H]([C@@H](O)[C@H](O)[C@H]3O)C(O)=O)cc2Cl)c(NS(=O)(=O)c2ccc(cc2Cl)C2CC2)c1 |r| Show InChI InChI=1S/C34H35Cl2FN2O12S/c1-34(2,3)38-31(44)17-6-8-23(22(12-17)39-52(47,48)25-9-7-16(10-20(25)36)15-4-5-15)49-24-14-21(37)18(11-19(24)35)13-26(40)50-33-29(43)27(41)28(42)30(51-33)32(45)46/h6-12,14-15,27-30,33,39,41-43H,4-5,13H2,1-3H3,(H,38,44)(H,45,46)/t27-,28-,29+,30-,33+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen, Inc
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 in human liver microsomes using testosterone as substrate after 5 to 30 mins by LC-MS/MS analysis |
Drug Metab Dispos 40: 2239-49 (2012)
Article DOI: 10.1124/dmd.112.047928
BindingDB Entry DOI: 10.7270/Q2Z039WT |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50363928

(CHEMBL1951575)Show SMILES CC(C)(C)NC(=O)c1ccc(Oc2cc(F)c(CC(O)=O)cc2Cl)c(NS(=O)(=O)c2ccc(cc2Cl)C2CC2)c1 Show InChI InChI=1S/C28H27Cl2FN2O6S/c1-28(2,3)32-27(36)17-6-8-23(39-24-14-21(31)18(11-19(24)29)13-26(34)35)22(12-17)33-40(37,38)25-9-7-16(10-20(25)30)15-4-5-15/h6-12,14-15,33H,4-5,13H2,1-3H3,(H,32,36)(H,34,35) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen, Inc
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 in human liver microsomes using midazolam as substrate after 5 to 30 mins by LC-MS/MS analysis |
Drug Metab Dispos 40: 2239-49 (2012)
Article DOI: 10.1124/dmd.112.047928
BindingDB Entry DOI: 10.7270/Q2Z039WT |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2E1
(Homo sapiens (Human)) | BDBM50088490

(CHEMBL3526979)Show SMILES CC(C)(C)NC(=O)c1ccc(Oc2cc(F)c(CC(=O)O[C@@H]3O[C@@H]([C@@H](O)[C@H](O)[C@H]3O)C(O)=O)cc2Cl)c(NS(=O)(=O)c2ccc(cc2Cl)C2CC2)c1 |r| Show InChI InChI=1S/C34H35Cl2FN2O12S/c1-34(2,3)38-31(44)17-6-8-23(22(12-17)39-52(47,48)25-9-7-16(10-20(25)36)15-4-5-15)49-24-14-21(37)18(11-19(24)35)13-26(40)50-33-29(43)27(41)28(42)30(51-33)32(45)46/h6-12,14-15,27-30,33,39,41-43H,4-5,13H2,1-3H3,(H,38,44)(H,45,46)/t27-,28-,29+,30-,33+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen, Inc
Curated by ChEMBL
| Assay Description
Inhibition of CYP2E1 in human liver microsomes using chlorzoxazone as substrate after 5 to 30 mins by LC-MS/MS analysis |
Drug Metab Dispos 40: 2239-49 (2012)
Article DOI: 10.1124/dmd.112.047928
BindingDB Entry DOI: 10.7270/Q2Z039WT |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2E1
(Homo sapiens (Human)) | BDBM50363928

(CHEMBL1951575)Show SMILES CC(C)(C)NC(=O)c1ccc(Oc2cc(F)c(CC(O)=O)cc2Cl)c(NS(=O)(=O)c2ccc(cc2Cl)C2CC2)c1 Show InChI InChI=1S/C28H27Cl2FN2O6S/c1-28(2,3)32-27(36)17-6-8-23(39-24-14-21(31)18(11-19(24)29)13-26(34)35)22(12-17)33-40(37,38)25-9-7-16(10-20(25)30)15-4-5-15/h6-12,14-15,33H,4-5,13H2,1-3H3,(H,32,36)(H,34,35) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen, Inc
Curated by ChEMBL
| Assay Description
Inhibition of CYP2E1 in human liver microsomes using chlorzoxazone as substrate after 5 to 30 mins by LC-MS/MS analysis |
Drug Metab Dispos 40: 2239-49 (2012)
Article DOI: 10.1124/dmd.112.047928
BindingDB Entry DOI: 10.7270/Q2Z039WT |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50088490

(CHEMBL3526979)Show SMILES CC(C)(C)NC(=O)c1ccc(Oc2cc(F)c(CC(=O)O[C@@H]3O[C@@H]([C@@H](O)[C@H](O)[C@H]3O)C(O)=O)cc2Cl)c(NS(=O)(=O)c2ccc(cc2Cl)C2CC2)c1 |r| Show InChI InChI=1S/C34H35Cl2FN2O12S/c1-34(2,3)38-31(44)17-6-8-23(22(12-17)39-52(47,48)25-9-7-16(10-20(25)36)15-4-5-15)49-24-14-21(37)18(11-19(24)35)13-26(40)50-33-29(43)27(41)28(42)30(51-33)32(45)46/h6-12,14-15,27-30,33,39,41-43H,4-5,13H2,1-3H3,(H,38,44)(H,45,46)/t27-,28-,29+,30-,33+/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen, Inc
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 in human liver microsomes using dextromethorphan as substrate after 5 to 30 mins by LC-MS/MS analysis |
Drug Metab Dispos 40: 2239-49 (2012)
Article DOI: 10.1124/dmd.112.047928
BindingDB Entry DOI: 10.7270/Q2Z039WT |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50363928

(CHEMBL1951575)Show SMILES CC(C)(C)NC(=O)c1ccc(Oc2cc(F)c(CC(O)=O)cc2Cl)c(NS(=O)(=O)c2ccc(cc2Cl)C2CC2)c1 Show InChI InChI=1S/C28H27Cl2FN2O6S/c1-28(2,3)32-27(36)17-6-8-23(39-24-14-21(31)18(11-19(24)29)13-26(34)35)22(12-17)33-40(37,38)25-9-7-16(10-20(25)30)15-4-5-15/h6-12,14-15,33H,4-5,13H2,1-3H3,(H,32,36)(H,34,35) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen, Inc
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 in human liver microsomes using dextromethorphan as substrate after 5 to 30 mins by LC-MS/MS analysis |
Drug Metab Dispos 40: 2239-49 (2012)
Article DOI: 10.1124/dmd.112.047928
BindingDB Entry DOI: 10.7270/Q2Z039WT |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50088490

(CHEMBL3526979)Show SMILES CC(C)(C)NC(=O)c1ccc(Oc2cc(F)c(CC(=O)O[C@@H]3O[C@@H]([C@@H](O)[C@H](O)[C@H]3O)C(O)=O)cc2Cl)c(NS(=O)(=O)c2ccc(cc2Cl)C2CC2)c1 |r| Show InChI InChI=1S/C34H35Cl2FN2O12S/c1-34(2,3)38-31(44)17-6-8-23(22(12-17)39-52(47,48)25-9-7-16(10-20(25)36)15-4-5-15)49-24-14-21(37)18(11-19(24)35)13-26(40)50-33-29(43)27(41)28(42)30(51-33)32(45)46/h6-12,14-15,27-30,33,39,41-43H,4-5,13H2,1-3H3,(H,38,44)(H,45,46)/t27-,28-,29+,30-,33+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen, Inc
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C19 in human liver microsomes using (S)-Mephenytoin as substrate after 5 to 30 mins by LC-MS/MS analysis |
Drug Metab Dispos 40: 2239-49 (2012)
Article DOI: 10.1124/dmd.112.047928
BindingDB Entry DOI: 10.7270/Q2Z039WT |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50363928

(CHEMBL1951575)Show SMILES CC(C)(C)NC(=O)c1ccc(Oc2cc(F)c(CC(O)=O)cc2Cl)c(NS(=O)(=O)c2ccc(cc2Cl)C2CC2)c1 Show InChI InChI=1S/C28H27Cl2FN2O6S/c1-28(2,3)32-27(36)17-6-8-23(39-24-14-21(31)18(11-19(24)29)13-26(34)35)22(12-17)33-40(37,38)25-9-7-16(10-20(25)30)15-4-5-15/h6-12,14-15,33H,4-5,13H2,1-3H3,(H,32,36)(H,34,35) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen, Inc
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C19 in human liver microsomes using (S)-Mephenytoin as substrate after 5 to 30 mins by LC-MS/MS analysis |
Drug Metab Dispos 40: 2239-49 (2012)
Article DOI: 10.1124/dmd.112.047928
BindingDB Entry DOI: 10.7270/Q2Z039WT |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50088490

(CHEMBL3526979)Show SMILES CC(C)(C)NC(=O)c1ccc(Oc2cc(F)c(CC(=O)O[C@@H]3O[C@@H]([C@@H](O)[C@H](O)[C@H]3O)C(O)=O)cc2Cl)c(NS(=O)(=O)c2ccc(cc2Cl)C2CC2)c1 |r| Show InChI InChI=1S/C34H35Cl2FN2O12S/c1-34(2,3)38-31(44)17-6-8-23(22(12-17)39-52(47,48)25-9-7-16(10-20(25)36)15-4-5-15)49-24-14-21(37)18(11-19(24)35)13-26(40)50-33-29(43)27(41)28(42)30(51-33)32(45)46/h6-12,14-15,27-30,33,39,41-43H,4-5,13H2,1-3H3,(H,38,44)(H,45,46)/t27-,28-,29+,30-,33+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen, Inc
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 in human liver microsomes using diclofenac as substrate after 5 to 30 mins by LC-MS/MS analysis |
Drug Metab Dispos 40: 2239-49 (2012)
Article DOI: 10.1124/dmd.112.047928
BindingDB Entry DOI: 10.7270/Q2Z039WT |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50363928

(CHEMBL1951575)Show SMILES CC(C)(C)NC(=O)c1ccc(Oc2cc(F)c(CC(O)=O)cc2Cl)c(NS(=O)(=O)c2ccc(cc2Cl)C2CC2)c1 Show InChI InChI=1S/C28H27Cl2FN2O6S/c1-28(2,3)32-27(36)17-6-8-23(39-24-14-21(31)18(11-19(24)29)13-26(34)35)22(12-17)33-40(37,38)25-9-7-16(10-20(25)30)15-4-5-15/h6-12,14-15,33H,4-5,13H2,1-3H3,(H,32,36)(H,34,35) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen, Inc
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 in human liver microsomes using diclofenac as substrate after 5 to 30 mins by LC-MS/MS analysis |
Drug Metab Dispos 40: 2239-49 (2012)
Article DOI: 10.1124/dmd.112.047928
BindingDB Entry DOI: 10.7270/Q2Z039WT |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2B6
(Homo sapiens (Human)) | BDBM50088490

(CHEMBL3526979)Show SMILES CC(C)(C)NC(=O)c1ccc(Oc2cc(F)c(CC(=O)O[C@@H]3O[C@@H]([C@@H](O)[C@H](O)[C@H]3O)C(O)=O)cc2Cl)c(NS(=O)(=O)c2ccc(cc2Cl)C2CC2)c1 |r| Show InChI InChI=1S/C34H35Cl2FN2O12S/c1-34(2,3)38-31(44)17-6-8-23(22(12-17)39-52(47,48)25-9-7-16(10-20(25)36)15-4-5-15)49-24-14-21(37)18(11-19(24)35)13-26(40)50-33-29(43)27(41)28(42)30(51-33)32(45)46/h6-12,14-15,27-30,33,39,41-43H,4-5,13H2,1-3H3,(H,38,44)(H,45,46)/t27-,28-,29+,30-,33+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen, Inc
Curated by ChEMBL
| Assay Description
Inhibition of CYP2B6 in human liver microsomes using bupropion as substrate after 5 to 30 mins by LC-MS/MS analysis |
Drug Metab Dispos 40: 2239-49 (2012)
Article DOI: 10.1124/dmd.112.047928
BindingDB Entry DOI: 10.7270/Q2Z039WT |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2B6
(Homo sapiens (Human)) | BDBM50363928

(CHEMBL1951575)Show SMILES CC(C)(C)NC(=O)c1ccc(Oc2cc(F)c(CC(O)=O)cc2Cl)c(NS(=O)(=O)c2ccc(cc2Cl)C2CC2)c1 Show InChI InChI=1S/C28H27Cl2FN2O6S/c1-28(2,3)32-27(36)17-6-8-23(39-24-14-21(31)18(11-19(24)29)13-26(34)35)22(12-17)33-40(37,38)25-9-7-16(10-20(25)30)15-4-5-15/h6-12,14-15,33H,4-5,13H2,1-3H3,(H,32,36)(H,34,35) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen, Inc
Curated by ChEMBL
| Assay Description
Inhibition of CYP2B6 in human liver microsomes using bupropion as substrate after 5 to 30 mins by LC-MS/MS analysis |
Drug Metab Dispos 40: 2239-49 (2012)
Article DOI: 10.1124/dmd.112.047928
BindingDB Entry DOI: 10.7270/Q2Z039WT |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50088490

(CHEMBL3526979)Show SMILES CC(C)(C)NC(=O)c1ccc(Oc2cc(F)c(CC(=O)O[C@@H]3O[C@@H]([C@@H](O)[C@H](O)[C@H]3O)C(O)=O)cc2Cl)c(NS(=O)(=O)c2ccc(cc2Cl)C2CC2)c1 |r| Show InChI InChI=1S/C34H35Cl2FN2O12S/c1-34(2,3)38-31(44)17-6-8-23(22(12-17)39-52(47,48)25-9-7-16(10-20(25)36)15-4-5-15)49-24-14-21(37)18(11-19(24)35)13-26(40)50-33-29(43)27(41)28(42)30(51-33)32(45)46/h6-12,14-15,27-30,33,39,41-43H,4-5,13H2,1-3H3,(H,38,44)(H,45,46)/t27-,28-,29+,30-,33+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen, Inc
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 in human liver microsomes using phenacetin as substrate after 5 to 30 mins by LC-MS/MS analysis |
Drug Metab Dispos 40: 2239-49 (2012)
Article DOI: 10.1124/dmd.112.047928
BindingDB Entry DOI: 10.7270/Q2Z039WT |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50363928

(CHEMBL1951575)Show SMILES CC(C)(C)NC(=O)c1ccc(Oc2cc(F)c(CC(O)=O)cc2Cl)c(NS(=O)(=O)c2ccc(cc2Cl)C2CC2)c1 Show InChI InChI=1S/C28H27Cl2FN2O6S/c1-28(2,3)32-27(36)17-6-8-23(39-24-14-21(31)18(11-19(24)29)13-26(34)35)22(12-17)33-40(37,38)25-9-7-16(10-20(25)30)15-4-5-15/h6-12,14-15,33H,4-5,13H2,1-3H3,(H,32,36)(H,34,35) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen, Inc
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 in human liver microsomes using phenacetin as substrate after 5 to 30 mins by LC-MS/MS analysis |
Drug Metab Dispos 40: 2239-49 (2012)
Article DOI: 10.1124/dmd.112.047928
BindingDB Entry DOI: 10.7270/Q2Z039WT |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50088490

(CHEMBL3526979)Show SMILES CC(C)(C)NC(=O)c1ccc(Oc2cc(F)c(CC(=O)O[C@@H]3O[C@@H]([C@@H](O)[C@H](O)[C@H]3O)C(O)=O)cc2Cl)c(NS(=O)(=O)c2ccc(cc2Cl)C2CC2)c1 |r| Show InChI InChI=1S/C34H35Cl2FN2O12S/c1-34(2,3)38-31(44)17-6-8-23(22(12-17)39-52(47,48)25-9-7-16(10-20(25)36)15-4-5-15)49-24-14-21(37)18(11-19(24)35)13-26(40)50-33-29(43)27(41)28(42)30(51-33)32(45)46/h6-12,14-15,27-30,33,39,41-43H,4-5,13H2,1-3H3,(H,38,44)(H,45,46)/t27-,28-,29+,30-,33+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen, Inc
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 in human liver microsomes using midazolam as substrate after 5 to 30 mins by LC-MS/MS analysis |
Drug Metab Dispos 40: 2239-49 (2012)
Article DOI: 10.1124/dmd.112.047928
BindingDB Entry DOI: 10.7270/Q2Z039WT |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50088489

(CHEMBL3527324)Show SMILES CC(C)(CO)NC(=O)c1ccc(Oc2cc(F)c(CC(O)=O)cc2Cl)c(NS(=O)(=O)c2ccc(cc2Cl)C2CC2)c1 Show InChI InChI=1S/C28H27Cl2FN2O7S/c1-28(2,14-34)32-27(37)17-5-7-23(40-24-13-21(31)18(10-19(24)29)12-26(35)36)22(11-17)33-41(38,39)25-8-6-16(9-20(25)30)15-3-4-15/h5-11,13,15,33-34H,3-4,12,14H2,1-2H3,(H,32,37)(H,35,36) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.17E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen, Inc
Curated by ChEMBL
| Assay Description
Inhibition of recombinant CYP2C8 (unknown origin) treated with AMG 853 |
Drug Metab Dispos 40: 2239-49 (2012)
Article DOI: 10.1124/dmd.112.047928
BindingDB Entry DOI: 10.7270/Q2Z039WT |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2J2
(Homo sapiens (Human)) | BDBM50088489

(CHEMBL3527324)Show SMILES CC(C)(CO)NC(=O)c1ccc(Oc2cc(F)c(CC(O)=O)cc2Cl)c(NS(=O)(=O)c2ccc(cc2Cl)C2CC2)c1 Show InChI InChI=1S/C28H27Cl2FN2O7S/c1-28(2,14-34)32-27(37)17-5-7-23(40-24-13-21(31)18(10-19(24)29)12-26(35)36)22(11-17)33-41(38,39)25-8-6-16(9-20(25)30)15-3-4-15/h5-11,13,15,33-34H,3-4,12,14H2,1-2H3,(H,32,37)(H,35,36) | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.40E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen, Inc
Curated by ChEMBL
| Assay Description
Inhibition of recombinant CYP2J2 (unknown origin) treated with AMG 853 |
Drug Metab Dispos 40: 2239-49 (2012)
Article DOI: 10.1124/dmd.112.047928
BindingDB Entry DOI: 10.7270/Q2Z039WT |
More data for this
Ligand-Target Pair | |
Type-1 angiotensin II receptor
(Homo sapiens (Human)) | BDBM50043280
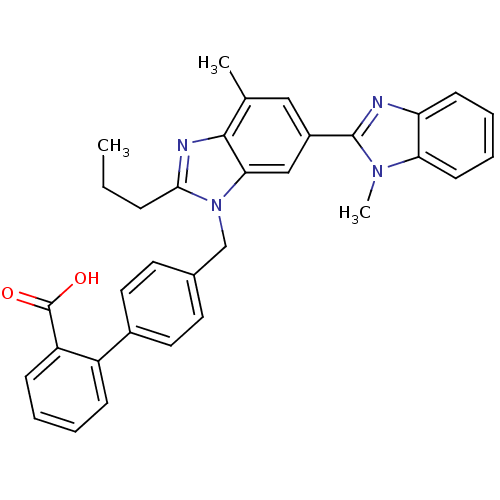
(4'-((1,4'-dimethyl-2'-propyl(2,6'-bi-1H-benzimidaz...)Show SMILES CCCc1nc2c(C)cc(cc2n1Cc1ccc(cc1)-c1ccccc1C(O)=O)-c1nc2ccccc2n1C Show InChI InChI=1S/C33H30N4O2/c1-4-9-30-35-31-21(2)18-24(32-34-27-12-7-8-13-28(27)36(32)3)19-29(31)37(30)20-22-14-16-23(17-15-22)25-10-5-6-11-26(25)33(38)39/h5-8,10-19H,4,9,20H2,1-3H3,(H,38,39) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| n/a | n/a | 0.490 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [125I]Tyr4-Sar1,Ile8-Angiotensin II from human Angiotensin 1 receptor after |
Bioorg Med Chem Lett 23: 767-72 (2013)
Article DOI: 10.1016/j.bmcl.2012.11.088
BindingDB Entry DOI: 10.7270/Q2542PW7 |
More data for this
Ligand-Target Pair | |
Type-1 angiotensin II receptor
(Homo sapiens (Human)) | BDBM50043280

(4'-((1,4'-dimethyl-2'-propyl(2,6'-bi-1H-benzimidaz...)Show SMILES CCCc1nc2c(C)cc(cc2n1Cc1ccc(cc1)-c1ccccc1C(O)=O)-c1nc2ccccc2n1C Show InChI InChI=1S/C33H30N4O2/c1-4-9-30-35-31-21(2)18-24(32-34-27-12-7-8-13-28(27)36(32)3)19-29(31)37(30)20-22-14-16-23(17-15-22)25-10-5-6-11-26(25)33(38)39/h5-8,10-19H,4,9,20H2,1-3H3,(H,38,39) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| n/a | n/a | 0.490 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [125I]Tyr4-Sar1,Ile8-Angiotensin II from human Angiotensin 1 receptor after 60 mins by scintillation counting |
J Med Chem 54: 4219-33 (2011)
Article DOI: 10.1021/jm200409s
BindingDB Entry DOI: 10.7270/Q2SB463J |
More data for this
Ligand-Target Pair | |
Type-1 angiotensin II receptor
(Homo sapiens (Human)) | BDBM50347563

(CHEMBL1801740)Show SMILES CCc1nc2c(C)cc(CC(C)C)nc2n1[C@H]1CCc2cc(ccc12)-c1ccccc1-c1nnn[nH]1 |r| Show InChI InChI=1S/C29H31N7/c1-5-26-31-27-18(4)15-21(14-17(2)3)30-29(27)36(26)25-13-11-20-16-19(10-12-23(20)25)22-8-6-7-9-24(22)28-32-34-35-33-28/h6-10,12,15-17,25H,5,11,13-14H2,1-4H3,(H,32,33,34,35)/t25-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [125I]Tyr4-Sar1,Ile8-Angiotensin II from human Angiotensin 1 receptor after 60 mins by scintillation counting |
J Med Chem 54: 4219-33 (2011)
Article DOI: 10.1021/jm200409s
BindingDB Entry DOI: 10.7270/Q2SB463J |
More data for this
Ligand-Target Pair | |
Type-1 angiotensin II receptor
(Homo sapiens (Human)) | BDBM50347563

(CHEMBL1801740)Show SMILES CCc1nc2c(C)cc(CC(C)C)nc2n1[C@H]1CCc2cc(ccc12)-c1ccccc1-c1nnn[nH]1 |r| Show InChI InChI=1S/C29H31N7/c1-5-26-31-27-18(4)15-21(14-17(2)3)30-29(27)36(26)25-13-11-20-16-19(10-12-23(20)25)22-8-6-7-9-24(22)28-32-34-35-33-28/h6-10,12,15-17,25H,5,11,13-14H2,1-4H3,(H,32,33,34,35)/t25-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [125I]Tyr4-Sar1,Ile8-Angiotensin II from human Angiotensin 1 receptor after |
Bioorg Med Chem Lett 23: 767-72 (2013)
Article DOI: 10.1016/j.bmcl.2012.11.088
BindingDB Entry DOI: 10.7270/Q2542PW7 |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50367254

(ENALAPRILAT)Show SMILES C[C@H](N[C@@H](CCc1ccccc1)C(O)=O)C(=O)N1CCC[C@H]1C(O)=O |r| Show InChI InChI=1S/C18H24N2O5/c1-12(16(21)20-11-5-8-15(20)18(24)25)19-14(17(22)23)10-9-13-6-3-2-4-7-13/h2-4,6-7,12,14-15,19H,5,8-11H2,1H3,(H,22,23)(H,24,25)/t12-,14-,15-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibitory activity against human renin inhibition (at pH 7.4) |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Type-1 angiotensin II receptor
(Homo sapiens (Human)) | BDBM50009718

(2-Ethyl-5,7-dimethyl-3-[2'-(1H-tetrazol-5-yl)-biph...)Show SMILES CCc1nc2c(C)cc(C)nc2n1Cc1ccc(cc1)-c1ccccc1-c1nnn[nH]1 Show InChI InChI=1S/C24H23N7/c1-4-21-26-22-15(2)13-16(3)25-24(22)31(21)14-17-9-11-18(12-10-17)19-7-5-6-8-20(19)23-27-29-30-28-23/h5-13H,4,14H2,1-3H3,(H,27,28,29,30) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [125I]Tyr4-Sar1,Ile8-Angiotensin II from human Angiotensin 1 receptor after 60 mins by scintillation counting |
J Med Chem 54: 4219-33 (2011)
Article DOI: 10.1021/jm200409s
BindingDB Entry DOI: 10.7270/Q2SB463J |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM33294
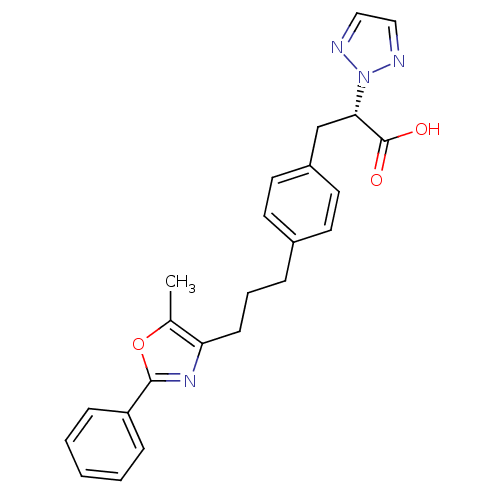
(phenylpropanoic acid derivative, 17j)Show SMILES Cc1oc(nc1CCCc1ccc(C[C@@H](C(O)=O)n2nccn2)cc1)-c1ccccc1 |r| Show InChI InChI=1S/C24H24N4O3/c1-17-21(27-23(31-17)20-7-3-2-4-8-20)9-5-6-18-10-12-19(13-11-18)16-22(24(29)30)28-25-14-15-26-28/h2-4,7-8,10-15,22H,5-6,9,16H2,1H3,(H,29,30)/t22-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 3 | n/a | 13 | n/a | n/a | 7.4 | 23 |
Pfizer
| Assay Description
Human 6His-PPAR ligand-binding domain was added to the mixture containing radioligand and test compound, followed by yttrium silicate polylysine SPA ... |
Bioorg Med Chem 17: 7113-25 (2009)
Article DOI: 10.1016/j.bmc.2009.09.001
BindingDB Entry DOI: 10.7270/Q2BP0158 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Type-1 angiotensin II receptor
(Homo sapiens (Human)) | BDBM50347564
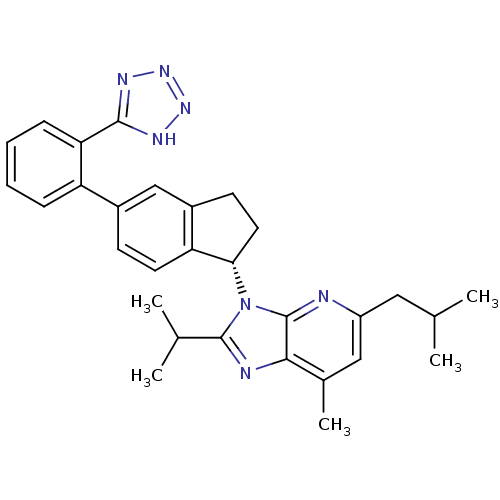
(CHEMBL1801741)Show SMILES CC(C)Cc1cc(C)c2nc(C(C)C)n([C@H]3CCc4cc(ccc34)-c3ccccc3-c3nnn[nH]3)c2n1 |r| Show InChI InChI=1S/C30H33N7/c1-17(2)14-22-15-19(5)27-30(31-22)37(29(32-27)18(3)4)26-13-11-21-16-20(10-12-24(21)26)23-8-6-7-9-25(23)28-33-35-36-34-28/h6-10,12,15-18,26H,11,13-14H2,1-5H3,(H,33,34,35,36)/t26-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [125I]Tyr4-Sar1,Ile8-Angiotensin II from human Angiotensin 1 receptor after 60 mins by scintillation counting |
J Med Chem 54: 4219-33 (2011)
Article DOI: 10.1021/jm200409s
BindingDB Entry DOI: 10.7270/Q2SB463J |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM33293

(phenylpropanoic acid derivative, 17i | racemic)Show SMILES Cc1oc(nc1CCCc1ccc(CC(C(O)=O)n2nccn2)cc1)-c1ccccc1 Show InChI InChI=1S/C24H24N4O3/c1-17-21(27-23(31-17)20-7-3-2-4-8-20)9-5-6-18-10-12-19(13-11-18)16-22(24(29)30)28-25-14-15-26-28/h2-4,7-8,10-15,22H,5-6,9,16H2,1H3,(H,29,30) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 5 | n/a | 15 | n/a | n/a | 7.4 | 23 |
Pfizer
| Assay Description
Human 6His-PPAR ligand-binding domain was added to the mixture containing radioligand and test compound, followed by yttrium silicate polylysine SPA ... |
Bioorg Med Chem 17: 7113-25 (2009)
Article DOI: 10.1016/j.bmc.2009.09.001
BindingDB Entry DOI: 10.7270/Q2BP0158 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Type-1 angiotensin II receptor
(Homo sapiens (Human)) | BDBM50426788
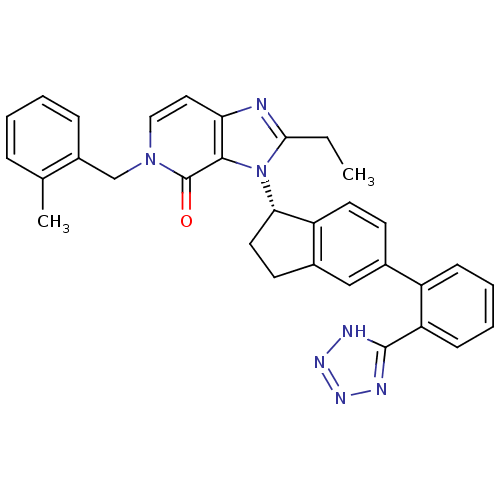
(CHEMBL2322444)Show SMILES CCc1nc2ccn(Cc3ccccc3C)c(=O)c2n1[C@H]1CCc2cc(ccc12)-c1ccccc1-c1nnn[nH]1 |r| Show InChI InChI=1S/C32H29N7O/c1-3-29-33-27-16-17-38(19-23-9-5-4-8-20(23)2)32(40)30(27)39(29)28-15-13-22-18-21(12-14-25(22)28)24-10-6-7-11-26(24)31-34-36-37-35-31/h4-12,14,16-18,28H,3,13,15,19H2,1-2H3,(H,34,35,36,37)/t28-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [125I]Tyr4-Sar1,Ile8-Angiotensin II from human Angiotensin 1 receptor after |
Bioorg Med Chem Lett 23: 767-72 (2013)
Article DOI: 10.1016/j.bmcl.2012.11.088
BindingDB Entry DOI: 10.7270/Q2542PW7 |
More data for this
Ligand-Target Pair | |
Type-1 angiotensin II receptor
(Homo sapiens (Human)) | BDBM50347565
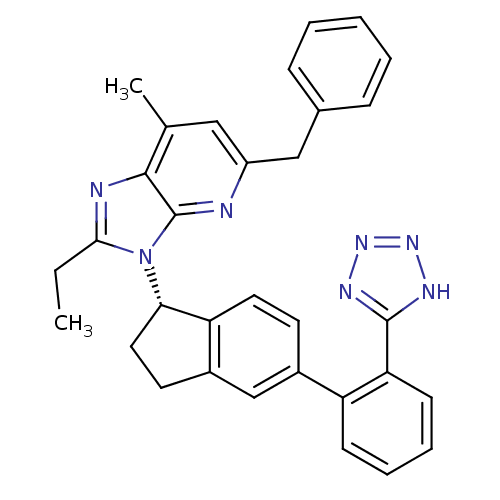
(CHEMBL1801743)Show SMILES CCc1nc2c(C)cc(Cc3ccccc3)nc2n1[C@H]1CCc2cc(ccc12)-c1ccccc1-c1nnn[nH]1 |r| Show InChI InChI=1S/C32H29N7/c1-3-29-34-30-20(2)17-24(18-21-9-5-4-6-10-21)33-32(30)39(29)28-16-14-23-19-22(13-15-26(23)28)25-11-7-8-12-27(25)31-35-37-38-36-31/h4-13,15,17,19,28H,3,14,16,18H2,1-2H3,(H,35,36,37,38)/t28-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [125I]Tyr4-Sar1,Ile8-Angiotensin II from human Angiotensin 1 receptor after 60 mins by scintillation counting |
J Med Chem 54: 4219-33 (2011)
Article DOI: 10.1021/jm200409s
BindingDB Entry DOI: 10.7270/Q2SB463J |
More data for this
Ligand-Target Pair | |
Type-1 angiotensin II receptor
(Homo sapiens (Human)) | BDBM50426777
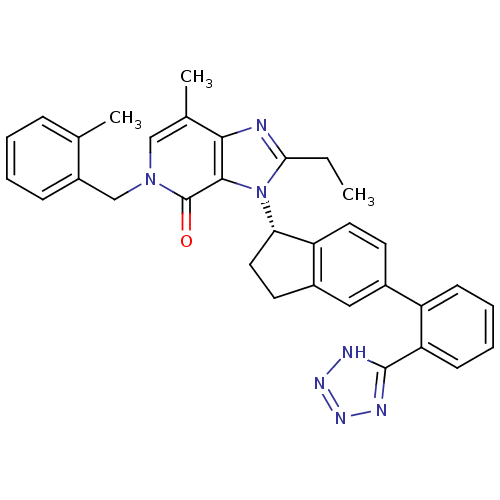
(CHEMBL2322175)Show SMILES CCc1nc2c(C)cn(Cc3ccccc3C)c(=O)c2n1[C@H]1CCc2cc(ccc12)-c1ccccc1-c1nnn[nH]1 |r| Show InChI InChI=1S/C33H31N7O/c1-4-29-34-30-21(3)18-39(19-24-10-6-5-9-20(24)2)33(41)31(30)40(29)28-16-14-23-17-22(13-15-26(23)28)25-11-7-8-12-27(25)32-35-37-38-36-32/h5-13,15,17-18,28H,4,14,16,19H2,1-3H3,(H,35,36,37,38)/t28-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [125I]Tyr4-Sar1,Ile8-Angiotensin II from human Angiotensin 1 receptor after |
Bioorg Med Chem Lett 23: 767-72 (2013)
Article DOI: 10.1016/j.bmcl.2012.11.088
BindingDB Entry DOI: 10.7270/Q2542PW7 |
More data for this
Ligand-Target Pair | |
Type-1 angiotensin II receptor
(Homo sapiens (Human)) | BDBM50347566
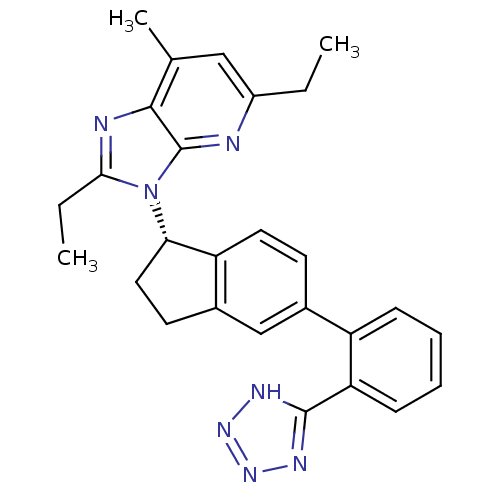
(CHEMBL1801738)Show SMILES CCc1nc2c(C)cc(CC)nc2n1[C@H]1CCc2cc(ccc12)-c1ccccc1-c1nnn[nH]1 |r| Show InChI InChI=1S/C27H27N7/c1-4-19-14-16(3)25-27(28-19)34(24(5-2)29-25)23-13-11-18-15-17(10-12-21(18)23)20-8-6-7-9-22(20)26-30-32-33-31-26/h6-10,12,14-15,23H,4-5,11,13H2,1-3H3,(H,30,31,32,33)/t23-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [125I]Tyr4-Sar1,Ile8-Angiotensin II from human Angiotensin 1 receptor after 60 mins by scintillation counting |
J Med Chem 54: 4219-33 (2011)
Article DOI: 10.1021/jm200409s
BindingDB Entry DOI: 10.7270/Q2SB463J |
More data for this
Ligand-Target Pair | |
Type-1 angiotensin II receptor
(Homo sapiens (Human)) | BDBM50426776
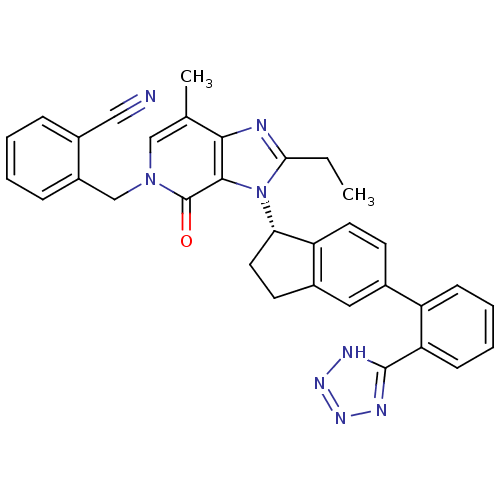
(CHEMBL2322176)Show SMILES CCc1nc2c(C)cn(Cc3ccccc3C#N)c(=O)c2n1[C@H]1CCc2cc(ccc12)-c1ccccc1-c1nnn[nH]1 |r| Show InChI InChI=1S/C33H28N8O/c1-3-29-35-30-20(2)18-40(19-24-9-5-4-8-23(24)17-34)33(42)31(30)41(29)28-15-13-22-16-21(12-14-26(22)28)25-10-6-7-11-27(25)32-36-38-39-37-32/h4-12,14,16,18,28H,3,13,15,19H2,1-2H3,(H,36,37,38,39)/t28-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [125I]Tyr4-Sar1,Ile8-Angiotensin II from human Angiotensin 1 receptor after |
Bioorg Med Chem Lett 23: 767-72 (2013)
Article DOI: 10.1016/j.bmcl.2012.11.088
BindingDB Entry DOI: 10.7270/Q2542PW7 |
More data for this
Ligand-Target Pair | |
Type-1 angiotensin II receptor
(Homo sapiens (Human)) | BDBM50347567

(CHEMBL1801712)Show SMILES CCc1nc2c(C)cc(C)nc2n1[C@H]1CCc2cc(ccc12)-c1ccccc1-c1nnn[nH]1 |r| Show InChI InChI=1S/C26H25N7/c1-4-23-28-24-15(2)13-16(3)27-26(24)33(23)22-12-10-18-14-17(9-11-20(18)22)19-7-5-6-8-21(19)25-29-31-32-30-25/h5-9,11,13-14,22H,4,10,12H2,1-3H3,(H,29,30,31,32)/t22-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 7.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [125I]Tyr4-Sar1,Ile8-Angiotensin II from human Angiotensin 1 receptor after 60 mins by scintillation counting |
J Med Chem 54: 4219-33 (2011)
Article DOI: 10.1021/jm200409s
BindingDB Entry DOI: 10.7270/Q2SB463J |
More data for this
Ligand-Target Pair | |
Type-1 angiotensin II receptor
(Homo sapiens (Human)) | BDBM50347568
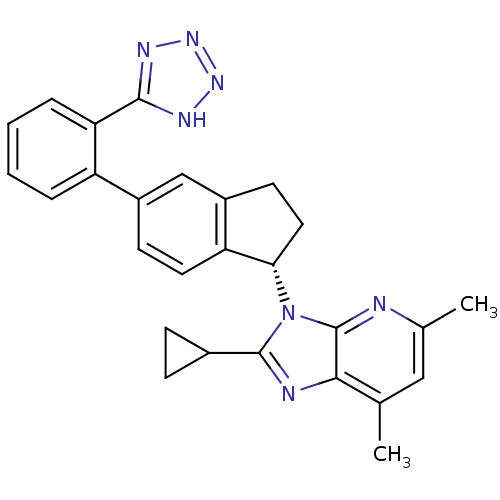
(CHEMBL1801735)Show SMILES Cc1cc(C)c2nc(C3CC3)n([C@H]3CCc4cc(ccc34)-c3ccccc3-c3nnn[nH]3)c2n1 |r| Show InChI InChI=1S/C27H25N7/c1-15-13-16(2)28-27-24(15)29-26(17-7-8-17)34(27)23-12-10-19-14-18(9-11-21(19)23)20-5-3-4-6-22(20)25-30-32-33-31-25/h3-6,9,11,13-14,17,23H,7-8,10,12H2,1-2H3,(H,30,31,32,33)/t23-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [125I]Tyr4-Sar1,Ile8-Angiotensin II from human Angiotensin 1 receptor after 60 mins by scintillation counting |
J Med Chem 54: 4219-33 (2011)
Article DOI: 10.1021/jm200409s
BindingDB Entry DOI: 10.7270/Q2SB463J |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50242264

(CHEMBL4086264)Show SMILES CC[C@H](N[C@@H](CCc1ccccc1)C(O)=O)C(=O)N1C[C@H](C[C@H]1C(O)=O)NC(=O)C[C@H](N)Cc1cc(F)c(F)cc1F |r| Show InChI InChI=1S/C29H35F3N4O6/c1-2-23(35-24(28(39)40)9-8-16-6-4-3-5-7-16)27(38)36-15-19(13-25(36)29(41)42)34-26(37)12-18(33)10-17-11-21(31)22(32)14-20(17)30/h3-7,11,14,18-19,23-25,35H,2,8-10,12-13,15,33H2,1H3,(H,34,37)(H,39,40)(H,41,42)/t18-,19+,23+,24+,25+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 8.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibition of human plasma ACE |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Rattus norvegicus) | BDBM50367254

(ENALAPRILAT)Show SMILES C[C@H](N[C@@H](CCc1ccccc1)C(O)=O)C(=O)N1CCC[C@H]1C(O)=O |r| Show InChI InChI=1S/C18H24N2O5/c1-12(16(21)20-11-5-8-15(20)18(24)25)19-14(17(22)23)10-9-13-6-3-2-4-7-13/h2-4,6-7,12,14-15,19H,5,8-11H2,1H3,(H,22,23)(H,24,25)/t12-,14-,15-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Ranbaxy Laboratories Limited
Curated by ChEMBL
| Assay Description
Inhibition of Wistar rat plasma ACE |
Bioorg Med Chem Lett 27: 2313-2318 (2017)
Article DOI: 10.1016/j.bmcl.2017.04.036
BindingDB Entry DOI: 10.7270/Q2ZC858S |
More data for this
Ligand-Target Pair | |
Type-1 angiotensin II receptor
(Homo sapiens (Human)) | BDBM50347569

(CHEMBL1801734)Show SMILES CC(C)c1nc2c(C)cc(C)nc2n1[C@H]1CCc2cc(ccc12)-c1ccccc1-c1nnn[nH]1 |r| Show InChI InChI=1S/C27H27N7/c1-15(2)26-29-24-16(3)13-17(4)28-27(24)34(26)23-12-10-19-14-18(9-11-21(19)23)20-7-5-6-8-22(20)25-30-32-33-31-25/h5-9,11,13-15,23H,10,12H2,1-4H3,(H,30,31,32,33)/t23-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 10.2 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [125I]Tyr4-Sar1,Ile8-Angiotensin II from human Angiotensin 1 receptor after 60 mins by scintillation counting |
J Med Chem 54: 4219-33 (2011)
Article DOI: 10.1021/jm200409s
BindingDB Entry DOI: 10.7270/Q2SB463J |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data