 Found 1081 hits with Last Name = 'han' and Initial = 'sy'
Found 1081 hits with Last Name = 'han' and Initial = 'sy' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11542
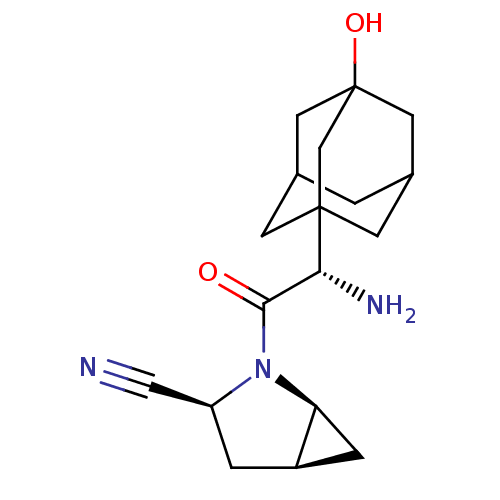
((1S,3S,5S)-2-[(2S)-2-amino-2-(3-hydroxyadamantan-1...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C12CC3CC(CC(O)(C3)C1)C2 |r,TLB:15:16:21:20.13.14,THB:17:16:13:20.21.18,17:18:16.15.22:13,19:18:16.15.22:13,15:14:16.22.17:21| Show InChI InChI=1S/C18H25N3O2/c19-8-13-2-12-3-14(12)21(13)16(22)15(20)17-4-10-1-11(5-17)7-18(23,6-10)9-17/h10-15,23H,1-7,9,20H2/t10?,11?,12-,13+,14+,15-,17?,18?/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| 0.600 | -52.1 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute
| Assay Description
Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea... |
J Med Chem 48: 5025-37 (2005)
Article DOI: 10.1021/jm050261p
BindingDB Entry DOI: 10.7270/Q2FN14DM |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11541
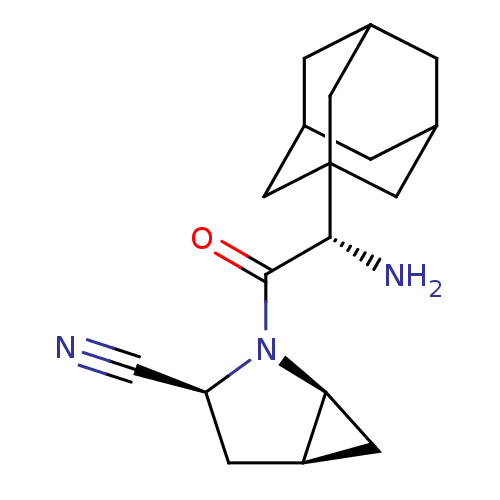
((1S,3S,5S)-2-[(2S)-2-(adamantan-1-yl)-2-aminoacety...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C12CC3CC(CC(C3)C1)C2 |r,TLB:15:16:20:19.13.14,THB:17:16:13:19.20.18,17:18:16.15.21:13,15:14:16.21.17:20| Show InChI InChI=1S/C18H25N3O/c19-9-14-4-13-5-15(13)21(14)17(22)16(20)18-6-10-1-11(7-18)3-12(2-10)8-18/h10-16H,1-8,20H2/t10?,11?,12?,13-,14+,15+,16-,18?/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.900 | -51.1 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute
| Assay Description
Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea... |
J Med Chem 48: 5025-37 (2005)
Article DOI: 10.1021/jm050261p
BindingDB Entry DOI: 10.7270/Q2FN14DM |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11530
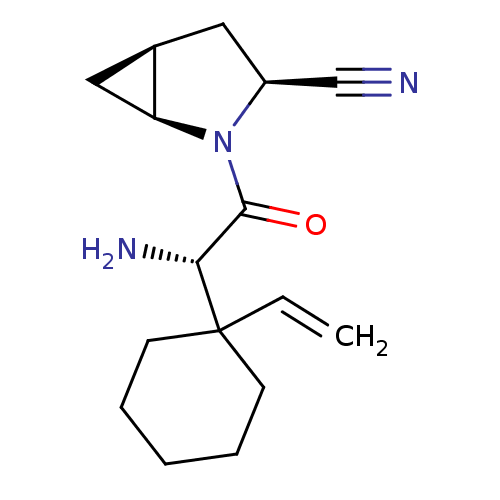
((1S,3S,5S)-2-[(2S)-2-amino-2-(1-ethenylcyclohexyl)...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C1(CCCCC1)C=C |r| Show InChI InChI=1S/C16H23N3O/c1-2-16(6-4-3-5-7-16)14(18)15(20)19-12(10-17)8-11-9-13(11)19/h2,11-14H,1,3-9,18H2/t11-,12+,13+,14-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.40 | -50.0 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute
| Assay Description
Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea... |
J Med Chem 48: 5025-37 (2005)
Article DOI: 10.1021/jm050261p
BindingDB Entry DOI: 10.7270/Q2FN14DM |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11544
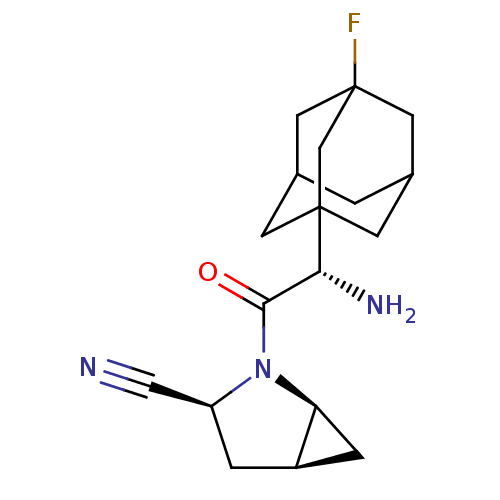
((1S,3S,5S)-2-[(2S)-2-amino-2-(3-fluoroadamantan-1-...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C12CC3CC(CC(F)(C3)C1)C2 |r,TLB:15:16:14.20.13:21,THB:17:16:13:20.21.18,17:18:16.15.22:13,19:18:16.15.22:13,15:14:16.22.17:21| Show InChI InChI=1S/C18H24FN3O/c19-18-6-10-1-11(7-18)5-17(4-10,9-18)15(21)16(23)22-13(8-20)2-12-3-14(12)22/h10-15H,1-7,9,21H2/t10?,11?,12-,13+,14+,15-,17?,18?/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.80 | -49.4 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute
| Assay Description
Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea... |
J Med Chem 48: 5025-37 (2005)
Article DOI: 10.1021/jm050261p
BindingDB Entry DOI: 10.7270/Q2FN14DM |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11543
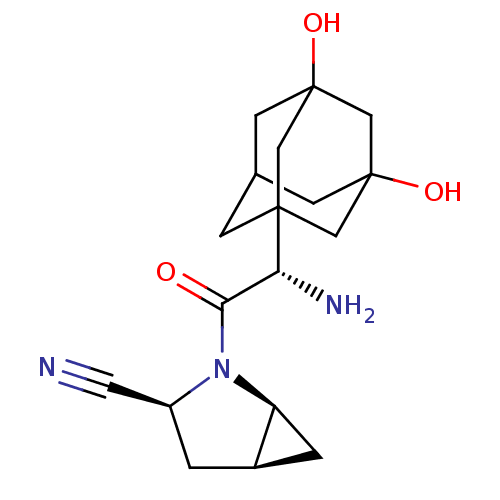
((1S,3S,5S)-2-[(2S)-2-amino-2-(3,5-dihydroxyadamant...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C12CC3CC(O)(CC(O)(C3)C1)C2 |r,TLB:21:14:23:18.22.19,21:19:14.15.13:23,20:19:14.15.13:23,17:16:14.13.21:22,15:16:14.13.21:22,THB:15:14:22:18.23.16| Show InChI InChI=1S/C18H25N3O3/c19-6-12-1-11-2-13(11)21(12)15(22)14(20)16-3-10-4-17(23,7-16)9-18(24,5-10)8-16/h10-14,23-24H,1-5,7-9,20H2/t10?,11-,12+,13+,14-,16?,17?,18?/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.10 | -49.0 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute
| Assay Description
Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea... |
J Med Chem 48: 5025-37 (2005)
Article DOI: 10.1021/jm050261p
BindingDB Entry DOI: 10.7270/Q2FN14DM |
More data for this
Ligand-Target Pair | |
Kinesin-like protein KIF11
(Homo sapiens (Human)) | BDBM50220180
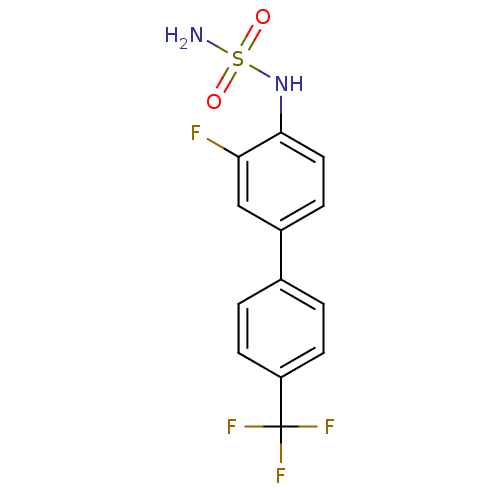
(CHEMBL243669 | N-[3-fluoro-4'-(trifluoromethyl)-4-...)Show InChI InChI=1S/C13H10F4N2O2S/c14-11-7-9(3-6-12(11)19-22(18,20)21)8-1-4-10(5-2-8)13(15,16)17/h1-7,19H,(H2,18,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human KSP motor domain by ATPase assay |
J Med Chem 50: 4939-52 (2007)
Article DOI: 10.1021/jm070435y
BindingDB Entry DOI: 10.7270/Q29023H4 |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11529
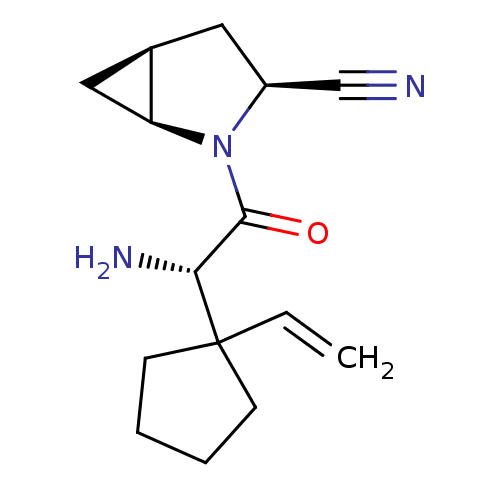
((1S,3S,5S)-2-[(2S)-2-amino-2-(1-ethenylcyclopentyl...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C1(CCCC1)C=C |r| Show InChI InChI=1S/C15H21N3O/c1-2-15(5-3-4-6-15)13(17)14(19)18-11(9-16)7-10-8-12(10)18/h2,10-13H,1,3-8,17H2/t10-,11+,12+,13-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.90 | -47.5 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute
| Assay Description
Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea... |
J Med Chem 48: 5025-37 (2005)
Article DOI: 10.1021/jm050261p
BindingDB Entry DOI: 10.7270/Q2FN14DM |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11535
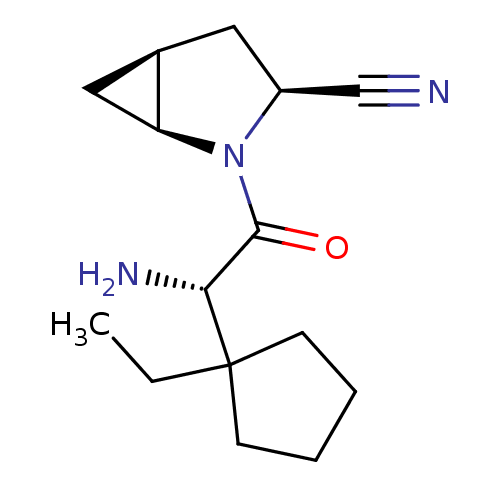
((1S,3S,5S)-2-[(2S)-2-amino-2-(1-ethylcyclopentyl)a...)Show SMILES CCC1(CCCC1)[C@H](N)C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N |r| Show InChI InChI=1S/C15H23N3O/c1-2-15(5-3-4-6-15)13(17)14(19)18-11(9-16)7-10-8-12(10)18/h10-13H,2-8,17H2,1H3/t10-,11+,12+,13-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 5.5 | -46.7 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute
| Assay Description
Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea... |
J Med Chem 48: 5025-37 (2005)
Article DOI: 10.1021/jm050261p
BindingDB Entry DOI: 10.7270/Q2FN14DM |
More data for this
Ligand-Target Pair | |
Kinesin-like protein KIF11
(Homo sapiens (Human)) | BDBM50220182
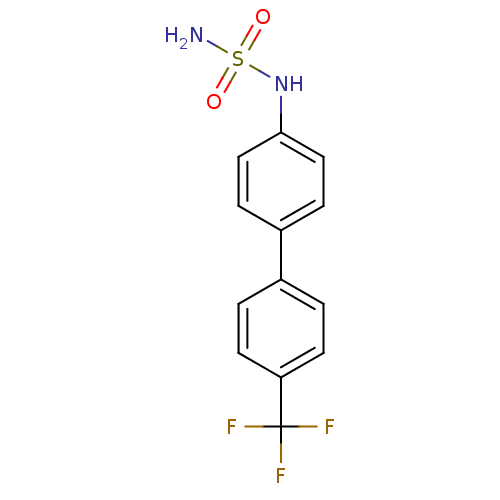
(CHEMBL390629 | N-[4'-(trifluoromethyl)-4-biphenyly...)Show InChI InChI=1S/C13H11F3N2O2S/c14-13(15,16)11-5-1-9(2-6-11)10-3-7-12(8-4-10)18-21(17,19)20/h1-8,18H,(H2,17,19,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 6.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human KSP motor domain by ATPase assay |
J Med Chem 50: 4939-52 (2007)
Article DOI: 10.1021/jm070435y
BindingDB Entry DOI: 10.7270/Q29023H4 |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11533
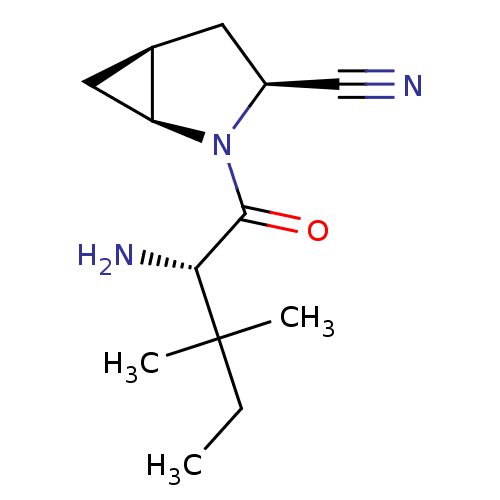
((1S,3S,5S)-2-[(2S)-2-amino-3,3-dimethylpentanoyl]-...)Show SMILES CCC(C)(C)[C@H](N)C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N |r| Show InChI InChI=1S/C13H21N3O/c1-4-13(2,3)11(15)12(17)16-9(7-14)5-8-6-10(8)16/h8-11H,4-6,15H2,1-3H3/t8-,9+,10+,11-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 7.10 | -46.0 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute
| Assay Description
Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea... |
J Med Chem 48: 5025-37 (2005)
Article DOI: 10.1021/jm050261p
BindingDB Entry DOI: 10.7270/Q2FN14DM |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11538
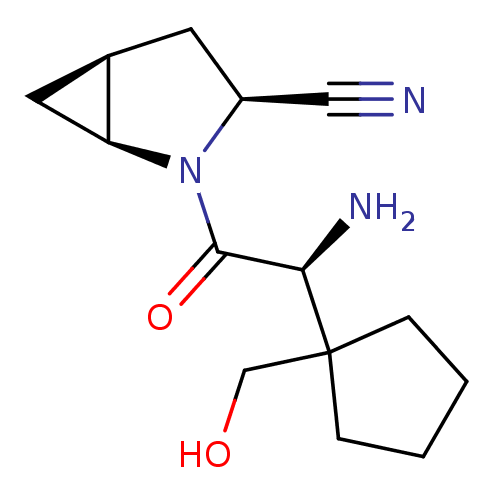
((1S,3S,5S)-2-[(2S)-2-amino-2-[1-(hydroxymethyl)cyc...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C1(CO)CCCC1 |r| Show InChI InChI=1S/C14H21N3O2/c15-7-10-5-9-6-11(9)17(10)13(19)12(16)14(8-18)3-1-2-4-14/h9-12,18H,1-6,8,16H2/t9-,10+,11+,12-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 7.40 | -45.9 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute
| Assay Description
Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea... |
J Med Chem 48: 5025-37 (2005)
Article DOI: 10.1021/jm050261p
BindingDB Entry DOI: 10.7270/Q2FN14DM |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11539
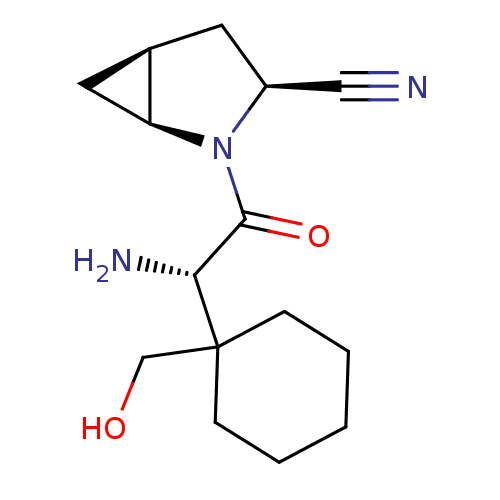
((1S,3S,5S)-2-[(2S)-2-amino-2-[1-(hydroxymethyl)cyc...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C1(CO)CCCCC1 |r| Show InChI InChI=1S/C15H23N3O2/c16-8-11-6-10-7-12(10)18(11)14(20)13(17)15(9-19)4-2-1-3-5-15/h10-13,19H,1-7,9,17H2/t10-,11+,12+,13-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 8 | -45.7 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute
| Assay Description
Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea... |
J Med Chem 48: 5025-37 (2005)
Article DOI: 10.1021/jm050261p
BindingDB Entry DOI: 10.7270/Q2FN14DM |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11531
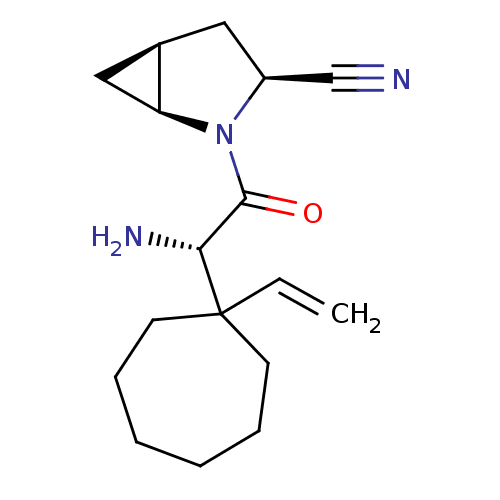
((1S,3S,5S)-2-[(2S)-2-amino-2-(1-ethenylcycloheptyl...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C1(CCCCCC1)C=C |r| Show InChI InChI=1S/C17H25N3O/c1-2-17(7-5-3-4-6-8-17)15(19)16(21)20-13(11-18)9-12-10-14(12)20/h2,12-15H,1,3-10,19H2/t12-,13+,14+,15-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 10 | -45.2 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute
| Assay Description
Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea... |
J Med Chem 48: 5025-37 (2005)
Article DOI: 10.1021/jm050261p
BindingDB Entry DOI: 10.7270/Q2FN14DM |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11532
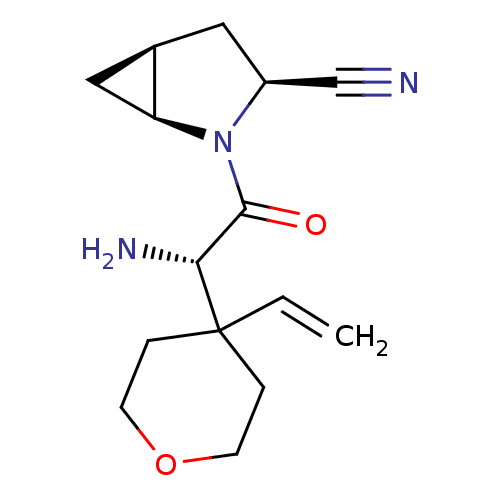
((1S,3S,5S)-2-[(2S)-2-amino-2-(4-ethenyloxan-4-yl)a...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C1(CCOCC1)C=C |r| Show InChI InChI=1S/C15H21N3O2/c1-2-15(3-5-20-6-4-15)13(17)14(19)18-11(9-16)7-10-8-12(10)18/h2,10-13H,1,3-8,17H2/t10-,11+,12+,13-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 10 | -45.2 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute
| Assay Description
Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea... |
J Med Chem 48: 5025-37 (2005)
Article DOI: 10.1021/jm050261p
BindingDB Entry DOI: 10.7270/Q2FN14DM |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11528
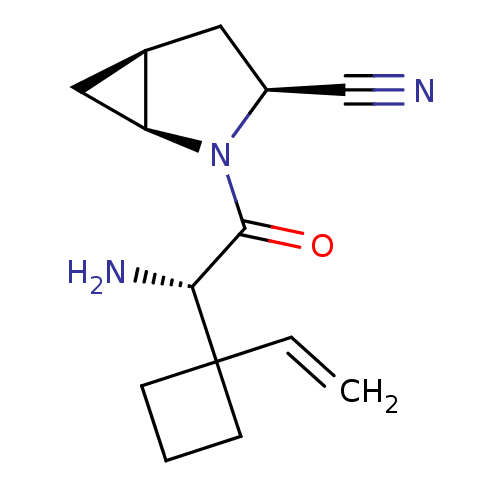
((1S,3S,5S)-2-[(2S)-2-amino-2-(1-ethenylcyclobutyl)...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C1(CCC1)C=C |r| Show InChI InChI=1S/C14H19N3O/c1-2-14(4-3-5-14)12(16)13(18)17-10(8-15)6-9-7-11(9)17/h2,9-12H,1,3-7,16H2/t9-,10+,11+,12-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 12 | -44.8 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute
| Assay Description
Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea... |
J Med Chem 48: 5025-37 (2005)
Article DOI: 10.1021/jm050261p
BindingDB Entry DOI: 10.7270/Q2FN14DM |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11536
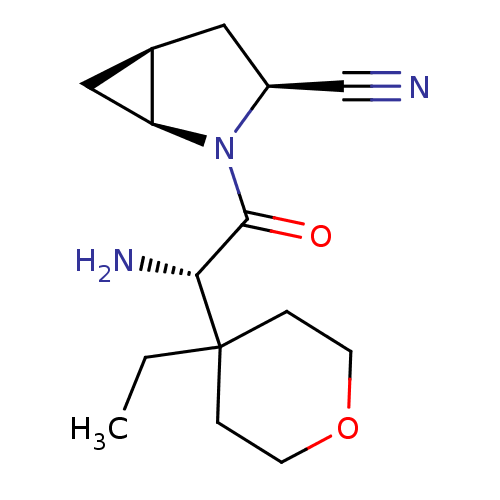
((1S,3S,5S)-2-[(2S)-2-amino-2-(4-ethyloxan-4-yl)ace...)Show SMILES CCC1(CCOCC1)[C@H](N)C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N |r| Show InChI InChI=1S/C15H23N3O2/c1-2-15(3-5-20-6-4-15)13(17)14(19)18-11(9-16)7-10-8-12(10)18/h10-13H,2-8,17H2,1H3/t10-,11+,12+,13-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 21 | -43.4 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute
| Assay Description
Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea... |
J Med Chem 48: 5025-37 (2005)
Article DOI: 10.1021/jm050261p
BindingDB Entry DOI: 10.7270/Q2FN14DM |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11527
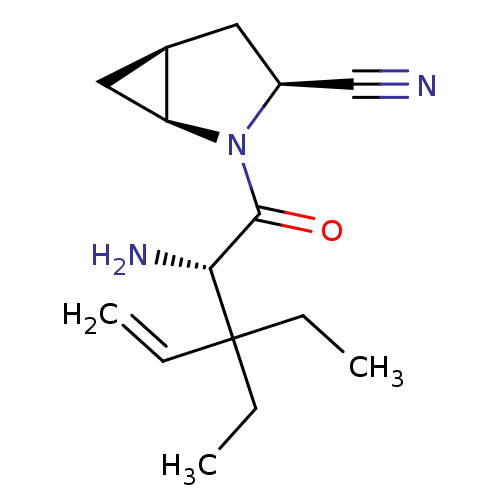
((1S,3S,5S)-2-[(2S)-2-amino-3,3-diethylpent-4-enoyl...)Show SMILES CCC(CC)(C=C)[C@H](N)C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N |r| Show InChI InChI=1S/C15H23N3O/c1-4-15(5-2,6-3)13(17)14(19)18-11(9-16)7-10-8-12(10)18/h4,10-13H,1,5-8,17H2,2-3H3/t10-,11+,12+,13-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 25 | -43.0 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute
| Assay Description
Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea... |
J Med Chem 48: 5025-37 (2005)
Article DOI: 10.1021/jm050261p
BindingDB Entry DOI: 10.7270/Q2FN14DM |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11534
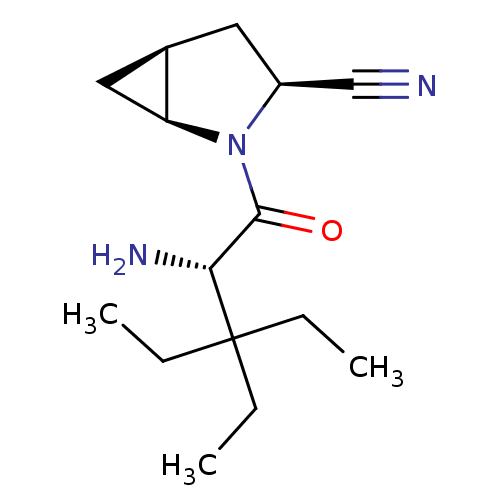
((1S,3S,5S)-2-[(2S)-2-amino-3,3-diethylpentanoyl]-2...)Show SMILES CCC(CC)(CC)[C@H](N)C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N |r| Show InChI InChI=1S/C15H25N3O/c1-4-15(5-2,6-3)13(17)14(19)18-11(9-16)7-10-8-12(10)18/h10-13H,4-8,17H2,1-3H3/t10-,11+,12+,13-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 31 | -42.4 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute
| Assay Description
Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea... |
J Med Chem 48: 5025-37 (2005)
Article DOI: 10.1021/jm050261p
BindingDB Entry DOI: 10.7270/Q2FN14DM |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11537
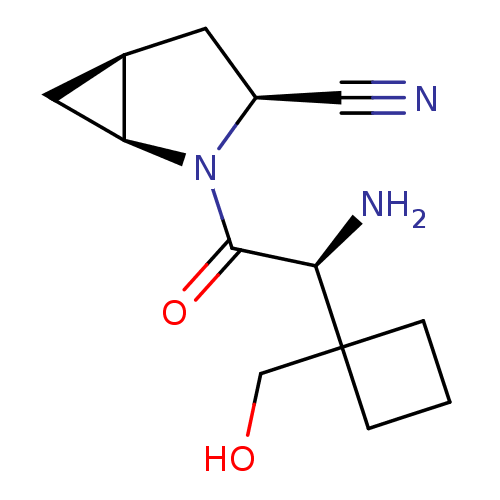
((1S,3S,5S)-2-[(2S)-2-amino-2-[1-(hydroxymethyl)cyc...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C1(CO)CCC1 |r| Show InChI InChI=1S/C13H19N3O2/c14-6-9-4-8-5-10(8)16(9)12(18)11(15)13(7-17)2-1-3-13/h8-11,17H,1-5,7,15H2/t8-,9+,10+,11-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 42 | -41.7 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute
| Assay Description
Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea... |
J Med Chem 48: 5025-37 (2005)
Article DOI: 10.1021/jm050261p
BindingDB Entry DOI: 10.7270/Q2FN14DM |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11525
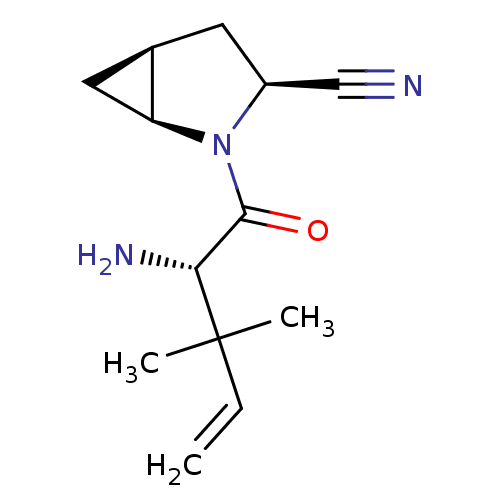
((1S,3S,5S)-2-[(2S)-2-amino-3,3-dimethylpent-4-enoy...)Show SMILES CC(C)(C=C)[C@H](N)C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N |r| Show InChI InChI=1S/C13H19N3O/c1-4-13(2,3)11(15)12(17)16-9(7-14)5-8-6-10(8)16/h4,8-11H,1,5-6,15H2,2-3H3/t8-,9+,10+,11-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 57 | -40.9 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute
| Assay Description
Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea... |
J Med Chem 48: 5025-37 (2005)
Article DOI: 10.1021/jm050261p
BindingDB Entry DOI: 10.7270/Q2FN14DM |
More data for this
Ligand-Target Pair | |
Lysosomal acid glucosylceramidase
(Homo sapiens (Human)) | BDBM50438750
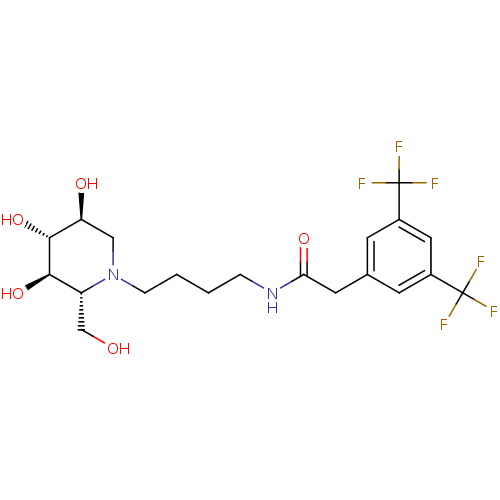
(CHEMBL2414884)Show SMILES OC[C@@H]1[C@@H](O)[C@H](O)[C@@H](O)CN1CCCCNC(=O)Cc1cc(cc(c1)C(F)(F)F)C(F)(F)F |r| Show InChI InChI=1S/C20H26F6N2O5/c21-19(22,23)12-5-11(6-13(8-12)20(24,25)26)7-16(31)27-3-1-2-4-28-9-15(30)18(33)17(32)14(28)10-29/h5-6,8,14-15,17-18,29-30,32-33H,1-4,7,9-10H2,(H,27,31)/t14-,15+,17-,18-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 71 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genomics Research Center
Curated by ChEMBL
| Assay Description
Competitive inhibition of human beta-glucocerebrosidase by Lineweaver-Burk double reciprocal plot method |
Bioorg Med Chem 21: 5021-8 (2013)
Article DOI: 10.1016/j.bmc.2013.06.054
BindingDB Entry DOI: 10.7270/Q2XG9SJX |
More data for this
Ligand-Target Pair | |
Kinesin-like protein KIF11
(Homo sapiens (Human)) | BDBM50220169
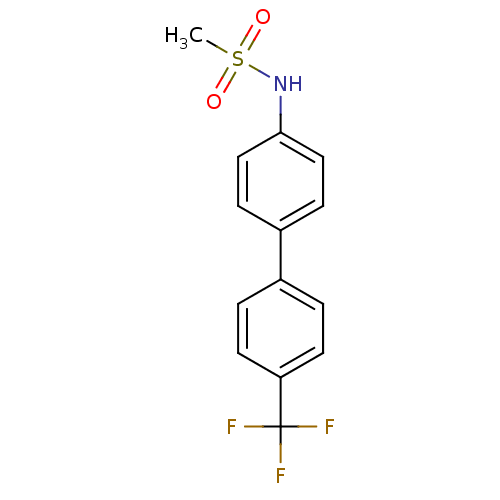
(CHEMBL244933 | N-[4'-(trifluoromethyl)-4-biphenyly...)Show InChI InChI=1S/C14H12F3NO2S/c1-21(19,20)18-13-8-4-11(5-9-13)10-2-6-12(7-3-10)14(15,16)17/h2-9,18H,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human KSP motor domain by ATPase assay |
J Med Chem 50: 4939-52 (2007)
Article DOI: 10.1021/jm070435y
BindingDB Entry DOI: 10.7270/Q29023H4 |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM11540
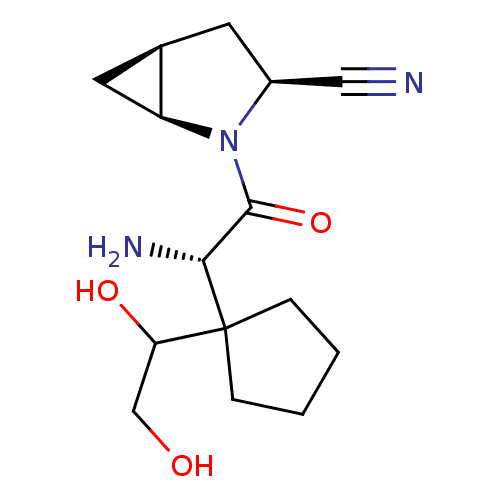
((1S,3S,5S)-2-[(2S)-2-amino-2-[1-(1,2-dihydroxyethy...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C1(CCCC1)C(O)CO |r| Show InChI InChI=1S/C15H23N3O3/c16-7-10-5-9-6-11(9)18(10)14(21)13(17)15(12(20)8-19)3-1-2-4-15/h9-13,19-20H,1-6,8,17H2/t9-,10+,11+,12?,13-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 143 | -38.7 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute
| Assay Description
Inhibition of human DPP-IV activity was measured under steady-state conditions by following the absorbance increase at 405 nm upon the substrate clea... |
J Med Chem 48: 5025-37 (2005)
Article DOI: 10.1021/jm050261p
BindingDB Entry DOI: 10.7270/Q2FN14DM |
More data for this
Ligand-Target Pair | |
Lysosomal acid glucosylceramidase
(Homo sapiens (Human)) | BDBM50438754
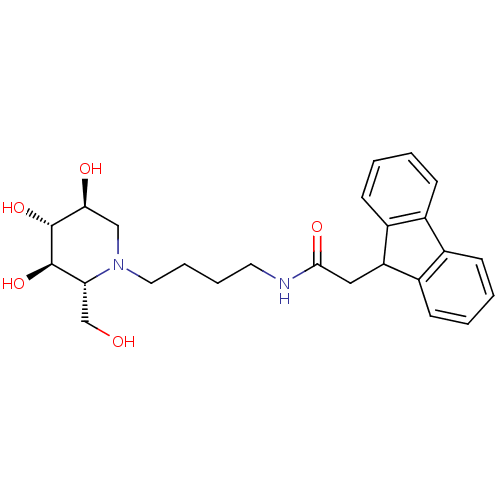
(CHEMBL2414880)Show SMILES OC[C@@H]1[C@@H](O)[C@H](O)[C@@H](O)CN1CCCCNC(=O)CC1c2ccccc2-c2ccccc12 |r| Show InChI InChI=1S/C25H32N2O5/c28-15-21-24(31)25(32)22(29)14-27(21)12-6-5-11-26-23(30)13-20-18-9-3-1-7-16(18)17-8-2-4-10-19(17)20/h1-4,7-10,20-22,24-25,28-29,31-32H,5-6,11-15H2,(H,26,30)/t21-,22+,24-,25-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genomics Research Center
Curated by ChEMBL
| Assay Description
Competitive inhibition of human beta-glucocerebrosidase by Lineweaver-Burk double reciprocal plot method |
Bioorg Med Chem 21: 5021-8 (2013)
Article DOI: 10.1016/j.bmc.2013.06.054
BindingDB Entry DOI: 10.7270/Q2XG9SJX |
More data for this
Ligand-Target Pair | |
Lysosomal acid glucosylceramidase
(Homo sapiens (Human)) | BDBM50438752
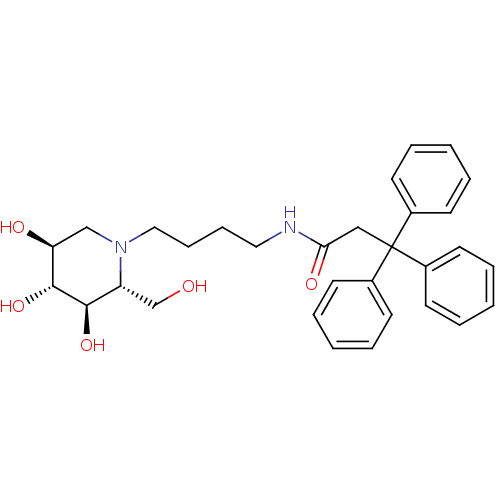
(CHEMBL2414882)Show SMILES OC[C@@H]1[C@@H](O)[C@H](O)[C@@H](O)CN1CCCCNC(=O)CC(c1ccccc1)(c1ccccc1)c1ccccc1 |r| Show InChI InChI=1S/C31H38N2O5/c34-22-26-29(37)30(38)27(35)21-33(26)19-11-10-18-32-28(36)20-31(23-12-4-1-5-13-23,24-14-6-2-7-15-24)25-16-8-3-9-17-25/h1-9,12-17,26-27,29-30,34-35,37-38H,10-11,18-22H2,(H,32,36)/t26-,27+,29-,30-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genomics Research Center
Curated by ChEMBL
| Assay Description
Competitive inhibition of human beta-glucocerebrosidase by Lineweaver-Burk double reciprocal plot method |
Bioorg Med Chem 21: 5021-8 (2013)
Article DOI: 10.1016/j.bmc.2013.06.054
BindingDB Entry DOI: 10.7270/Q2XG9SJX |
More data for this
Ligand-Target Pair | |
Lysosomal acid glucosylceramidase
(Homo sapiens (Human)) | BDBM50438753
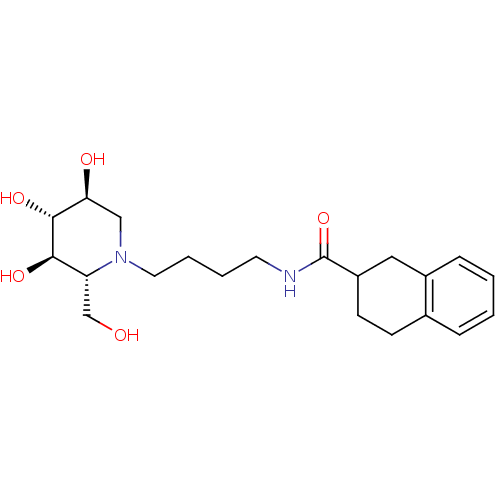
(CHEMBL2414881)Show SMILES OC[C@@H]1[C@@H](O)[C@H](O)[C@@H](O)CN1CCCCNC(=O)C1CCc2ccccc2C1 |r| Show InChI InChI=1S/C21H32N2O5/c24-13-17-19(26)20(27)18(25)12-23(17)10-4-3-9-22-21(28)16-8-7-14-5-1-2-6-15(14)11-16/h1-2,5-6,16-20,24-27H,3-4,7-13H2,(H,22,28)/t16?,17-,18+,19-,20-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genomics Research Center
Curated by ChEMBL
| Assay Description
Competitive inhibition of human beta-glucocerebrosidase by Lineweaver-Burk double reciprocal plot method |
Bioorg Med Chem 21: 5021-8 (2013)
Article DOI: 10.1016/j.bmc.2013.06.054
BindingDB Entry DOI: 10.7270/Q2XG9SJX |
More data for this
Ligand-Target Pair | |
Lysosomal acid glucosylceramidase
(Homo sapiens (Human)) | BDBM50438748
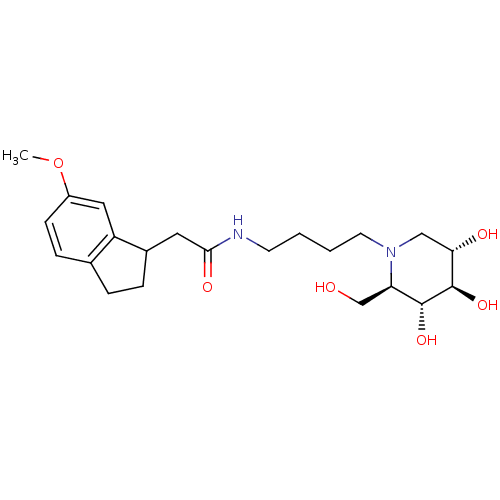
(CHEMBL2414879)Show SMILES COc1ccc2CCC(CC(=O)NCCCCN3C[C@H](O)[C@@H](O)[C@H](O)[C@H]3CO)c2c1 |r| Show InChI InChI=1S/C22H34N2O6/c1-30-16-7-6-14-4-5-15(17(14)11-16)10-20(27)23-8-2-3-9-24-12-19(26)22(29)21(28)18(24)13-25/h6-7,11,15,18-19,21-22,25-26,28-29H,2-5,8-10,12-13H2,1H3,(H,23,27)/t15?,18-,19+,21-,22-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genomics Research Center
Curated by ChEMBL
| Assay Description
Non-competitive inhibition of human beta-glucocerebrosidase by Lineweaver-Burk double reciprocal plot method |
Bioorg Med Chem 21: 5021-8 (2013)
Article DOI: 10.1016/j.bmc.2013.06.054
BindingDB Entry DOI: 10.7270/Q2XG9SJX |
More data for this
Ligand-Target Pair | |
Lysosomal acid glucosylceramidase
(Homo sapiens (Human)) | BDBM18358

((2R,3R,4R,5S)-2-(hydroxymethyl)-1-nonylpiperidine-...)Show SMILES CCCCCCCCCN1C[C@H](O)[C@@H](O)[C@H](O)[C@H]1CO Show InChI InChI=1S/C15H31NO4/c1-2-3-4-5-6-7-8-9-16-10-13(18)15(20)14(19)12(16)11-17/h12-15,17-20H,2-11H2,1H3/t12-,13+,14-,15-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 1.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genomics Research Center
Curated by ChEMBL
| Assay Description
Competitive inhibition of human beta-glucocerebrosidase by Lineweaver-Burk double reciprocal plot method |
Bioorg Med Chem 21: 5021-8 (2013)
Article DOI: 10.1016/j.bmc.2013.06.054
BindingDB Entry DOI: 10.7270/Q2XG9SJX |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Lysosomal acid glucosylceramidase
(Homo sapiens (Human)) | BDBM50438751
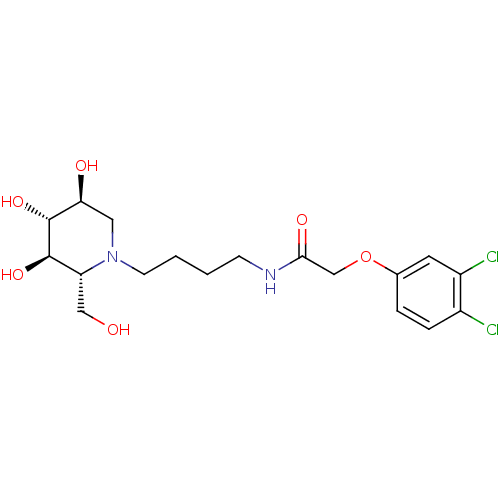
(CHEMBL2414883)Show SMILES OC[C@@H]1[C@@H](O)[C@H](O)[C@@H](O)CN1CCCCNC(=O)COc1ccc(Cl)c(Cl)c1 |r| Show InChI InChI=1S/C18H26Cl2N2O6/c19-12-4-3-11(7-13(12)20)28-10-16(25)21-5-1-2-6-22-8-15(24)18(27)17(26)14(22)9-23/h3-4,7,14-15,17-18,23-24,26-27H,1-2,5-6,8-10H2,(H,21,25)/t14-,15+,17-,18-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 3.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genomics Research Center
Curated by ChEMBL
| Assay Description
Competitive inhibition of human beta-glucocerebrosidase by Lineweaver-Burk double reciprocal plot method |
Bioorg Med Chem 21: 5021-8 (2013)
Article DOI: 10.1016/j.bmc.2013.06.054
BindingDB Entry DOI: 10.7270/Q2XG9SJX |
More data for this
Ligand-Target Pair | |
Lysosomal acid glucosylceramidase
(Homo sapiens (Human)) | BDBM50438749
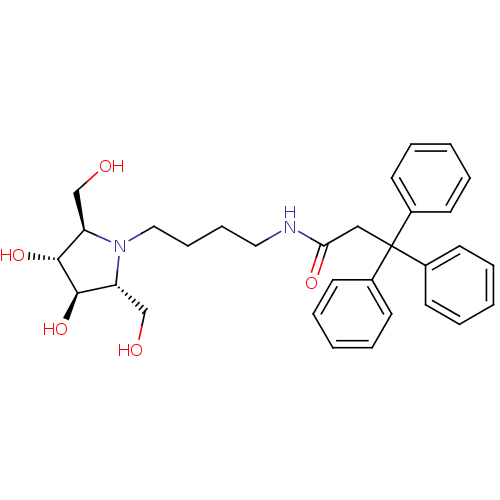
(CHEMBL2414888)Show SMILES OC[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)N1CCCCNC(=O)CC(c1ccccc1)(c1ccccc1)c1ccccc1 |r| Show InChI InChI=1S/C31H38N2O5/c34-21-26-29(37)30(38)27(22-35)33(26)19-11-10-18-32-28(36)20-31(23-12-4-1-5-13-23,24-14-6-2-7-15-24)25-16-8-3-9-17-25/h1-9,12-17,26-27,29-30,34-35,37-38H,10-11,18-22H2,(H,32,36)/t26-,27-,29-,30-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genomics Research Center
Curated by ChEMBL
| Assay Description
Competitive inhibition of human beta-glucocerebrosidase by Lineweaver-Burk double reciprocal plot method |
Bioorg Med Chem 21: 5021-8 (2013)
Article DOI: 10.1016/j.bmc.2013.06.054
BindingDB Entry DOI: 10.7270/Q2XG9SJX |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein kinase FLT3
(Homo sapiens (Human)) | BDBM50593957
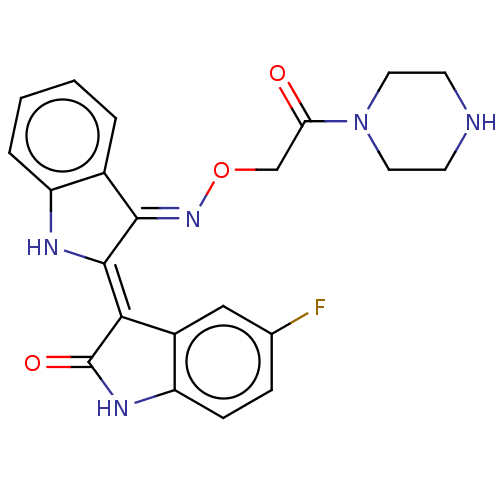
(CHEMBL5201185)Show SMILES Cl.Fc1ccc2NC(=O)\C(=C3/Nc4ccccc4/C/3=N\OCC(=O)N3CCNCC3)c2c1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.180 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114356
BindingDB Entry DOI: 10.7270/Q2D50RX8 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein kinase FLT3
(Homo sapiens (Human)) | BDBM50593961
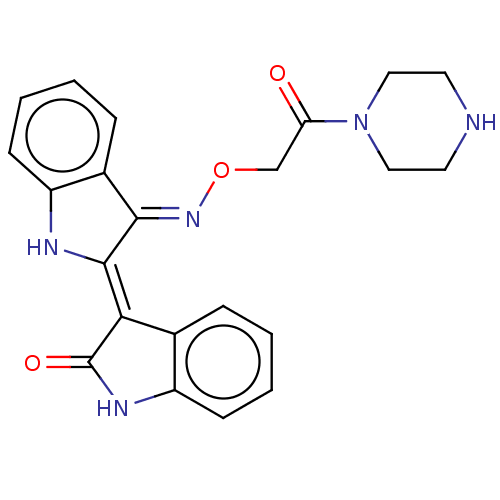
(CHEMBL5195819)Show SMILES Cl.O=C(CO\N=C1\C(\Nc2ccccc\12)=C1\C(=O)Nc2ccccc12)N1CCNCC1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.180 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114356
BindingDB Entry DOI: 10.7270/Q2D50RX8 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein kinase FLT3
(Homo sapiens (Human)) | BDBM50593962
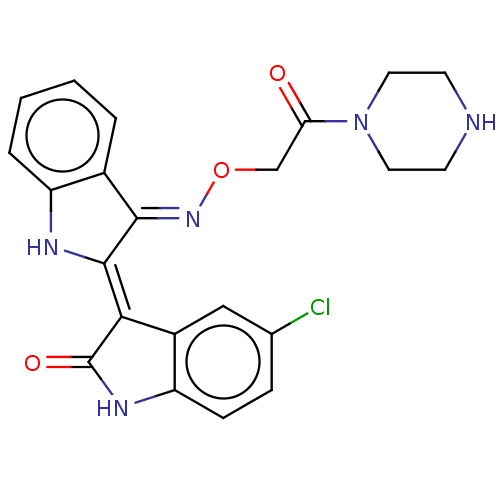
(CHEMBL5188834)Show SMILES Cl.Clc1ccc2NC(=O)\C(=C3/Nc4ccccc4/C/3=N\OCC(=O)N3CCNCC3)c2c1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114356
BindingDB Entry DOI: 10.7270/Q2D50RX8 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein kinase FLT3
(Homo sapiens (Human)) | BDBM50593981
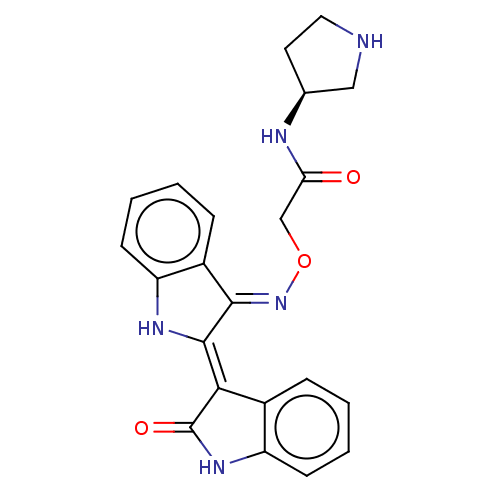
(CHEMBL5170345)Show SMILES Cl.O=C(CO\N=C1\C(\Nc2ccccc\12)=C1\C(=O)Nc2ccccc12)N[C@H]1CCNC1 |r| | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114356
BindingDB Entry DOI: 10.7270/Q2D50RX8 |
More data for this
Ligand-Target Pair | |
Hepatocyte growth factor receptor
(Homo sapiens (Human)) | BDBM50384024
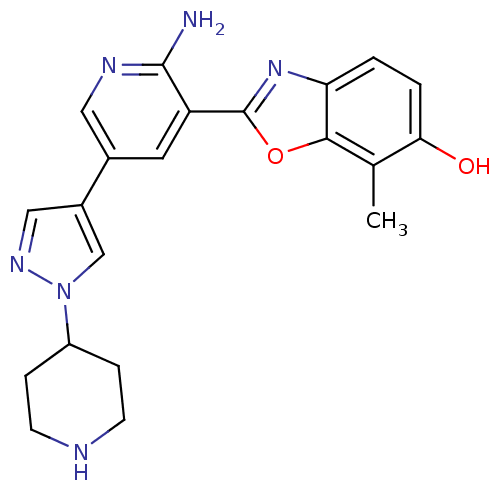
(CHEMBL2032280 | CHEMBL2079349)Show SMILES Cc1c(O)ccc2nc(oc12)-c1cc(cnc1N)-c1cnn(c1)C1CCNCC1 Show InChI InChI=1S/C21H22N6O2/c1-12-18(28)3-2-17-19(12)29-21(26-17)16-8-13(9-24-20(16)22)14-10-25-27(11-14)15-4-6-23-7-5-15/h2-3,8-11,15,23,28H,4-7H2,1H3,(H2,22,24) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Research Institute of Chemical Technology
Curated by ChEMBL
| Assay Description
Inhibition of recombinant c-Met by time resolved-fluorescence resonance energy transfer analysis |
Bioorg Med Chem Lett 22: 4044-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.04.083
BindingDB Entry DOI: 10.7270/Q2FF3TC4 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein kinase FLT3
(Homo sapiens (Human)) | BDBM50593979
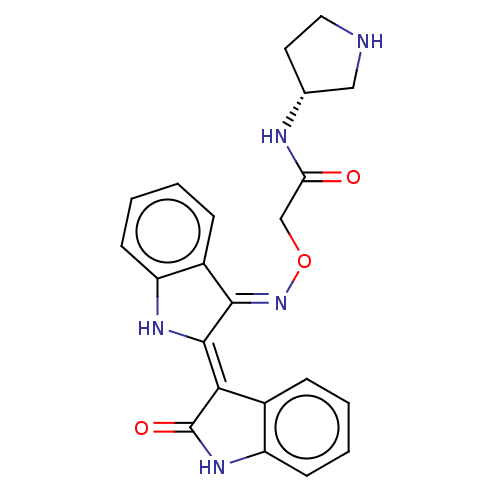
(CHEMBL5188978)Show SMILES Cl.O=C(CO\N=C1\C(\Nc2ccccc\12)=C1\C(=O)Nc2ccccc12)N[C@@H]1CCNC1 |r| | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114356
BindingDB Entry DOI: 10.7270/Q2D50RX8 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein kinase FLT3
(Homo sapiens (Human)) | BDBM50593963
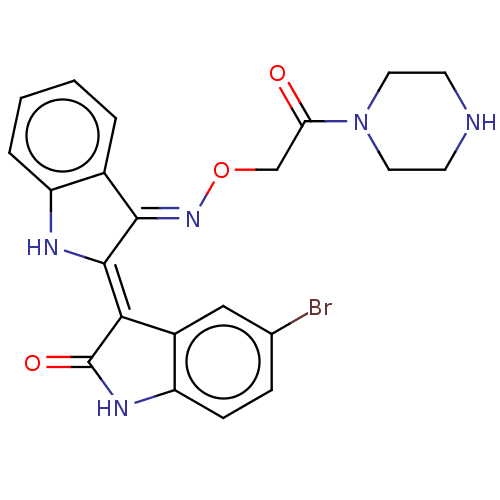
(CHEMBL5209537)Show SMILES Cl.Brc1ccc2NC(=O)\C(=C3/Nc4ccccc4/C/3=N\OCC(=O)N3CCNCC3)c2c1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.220 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114356
BindingDB Entry DOI: 10.7270/Q2D50RX8 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein kinase FLT3
(Homo sapiens (Human)) | BDBM50593961

(CHEMBL5195819)Show SMILES Cl.O=C(CO\N=C1\C(\Nc2ccccc\12)=C1\C(=O)Nc2ccccc12)N1CCNCC1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.260 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114356
BindingDB Entry DOI: 10.7270/Q2D50RX8 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein kinase FLT3
(Homo sapiens (Human)) | BDBM50593962

(CHEMBL5188834)Show SMILES Cl.Clc1ccc2NC(=O)\C(=C3/Nc4ccccc4/C/3=N\OCC(=O)N3CCNCC3)c2c1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.270 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114356
BindingDB Entry DOI: 10.7270/Q2D50RX8 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein kinase FLT3
(Homo sapiens (Human)) | BDBM144315
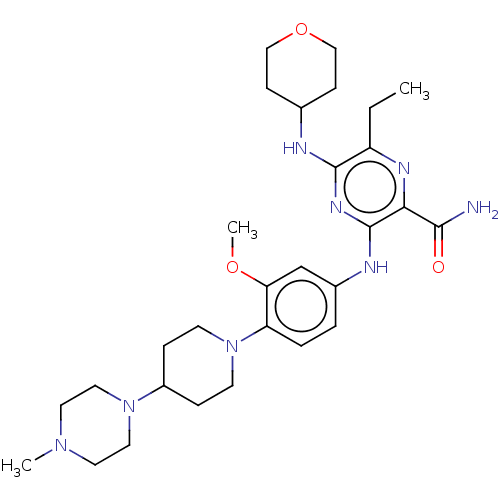
(Gilteritinib | US11512074, Example T-9 | US8969336...)Show SMILES CCc1nc(C(N)=O)c(Nc2ccc(N3CCC(CC3)N3CCN(C)CC3)c(OC)c2)nc1NC1CCOCC1 Show InChI InChI=1S/C29H44N8O3/c1-4-23-28(31-20-9-17-40-18-10-20)34-29(26(33-23)27(30)38)32-21-5-6-24(25(19-21)39-3)37-11-7-22(8-12-37)36-15-13-35(2)14-16-36/h5-6,19-20,22H,4,7-18H2,1-3H3,(H2,30,38)(H2,31,32,34) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.290 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of FLT3 expressed in human SEMK2 cells assessed as reduction in FLT3 phosphorylation incubated for 1 hr by immunoblotting analysis |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112205
BindingDB Entry DOI: 10.7270/Q2VM4H1R |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein kinase FLT3
(Homo sapiens (Human)) | BDBM50593979

(CHEMBL5188978)Show SMILES Cl.O=C(CO\N=C1\C(\Nc2ccccc\12)=C1\C(=O)Nc2ccccc12)N[C@@H]1CCNC1 |r| | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.290 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114356
BindingDB Entry DOI: 10.7270/Q2D50RX8 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein kinase FLT3
(Homo sapiens (Human)) | BDBM50593957

(CHEMBL5201185)Show SMILES Cl.Fc1ccc2NC(=O)\C(=C3/Nc4ccccc4/C/3=N\OCC(=O)N3CCNCC3)c2c1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.290 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114356
BindingDB Entry DOI: 10.7270/Q2D50RX8 |
More data for this
Ligand-Target Pair | |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM50380974

(CHEMBL2016903)Show SMILES Fc1ccc2[nH]c(=O)c(cc2c1)-c1nc2CCN(Cc2[nH]1)C(=O)c1ccncc1 Show InChI InChI=1S/C21H16FN5O2/c22-14-1-2-16-13(9-14)10-15(20(28)26-16)19-24-17-5-8-27(11-18(17)25-19)21(29)12-3-6-23-7-4-12/h1-4,6-7,9-10H,5,8,11H2,(H,24,25)(H,26,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Research Institute of Chemical Technology
Curated by ChEMBL
| Assay Description
Inhibition of recombinant VEGFR-2 kinase domain by homogeneous time-resolved fluorescence assay |
Bioorg Med Chem Lett 22: 2837-42 (2012)
Article DOI: 10.1016/j.bmcl.2012.02.073
BindingDB Entry DOI: 10.7270/Q2RN38W3 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein kinase FLT3
(Homo sapiens (Human)) | BDBM50564363
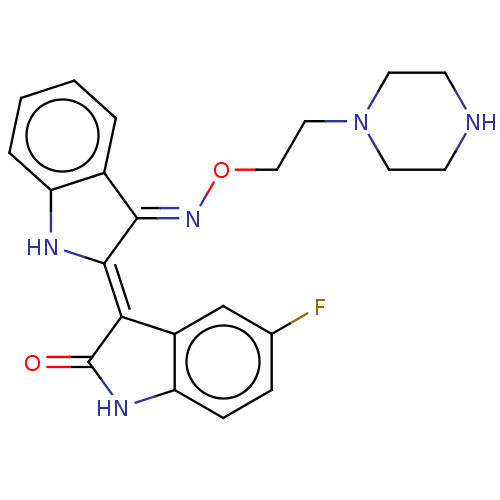
(CHEMBL4791998)Show SMILES Cl.Fc1ccc2NC(=O)\C(=C3/Nc4ccccc4/C/3=N\OCCN3CCNCC3)c2c1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.320 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human FLT3 D835Y mutant in presence of ATP |
Citation and Details
Article DOI: 10.1016/j.ejmech.2020.112205
BindingDB Entry DOI: 10.7270/Q2VM4H1R |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein kinase FLT3
(Homo sapiens (Human)) | BDBM559465
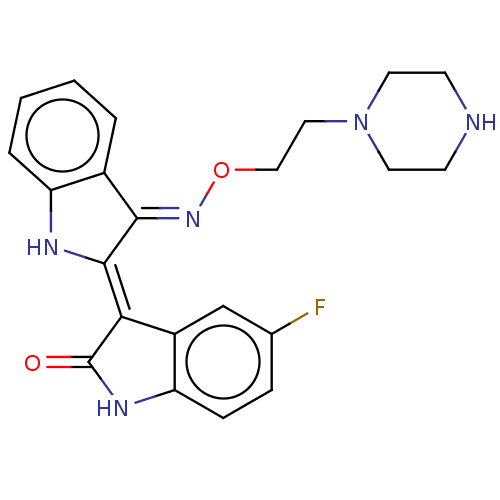
(US11370779, Compound 33)Show SMILES Fc1ccc2NC(=O)\C(=C3/Nc4ccccc4/C/3=N\OCCN3CCNCC3)c2c1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.320 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114356
BindingDB Entry DOI: 10.7270/Q2D50RX8 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein kinase FLT3
(Homo sapiens (Human)) | BDBM50593973
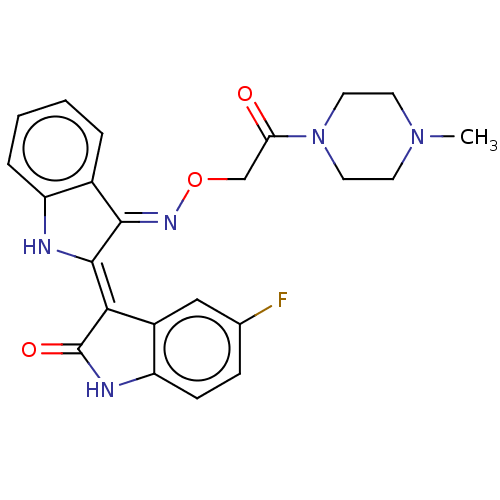
(CHEMBL5202926)Show SMILES Cl.CN1CCN(CC1)C(=O)CO\N=C1\C(\Nc2ccccc\12)=C1\C(=O)Nc2ccc(F)cc12 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.350 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114356
BindingDB Entry DOI: 10.7270/Q2D50RX8 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Yes
(Homo sapiens (Human)) | BDBM50610910

(CHEMBL5280844)Show SMILES OC[C@@H]1CCC(CO1)Nc1ncnc2[nH]cc(C(=O)c3ccc(Oc4ccccc4)cc3Cl)c12 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 0.390 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein kinase FLT3
(Homo sapiens (Human)) | BDBM50593977
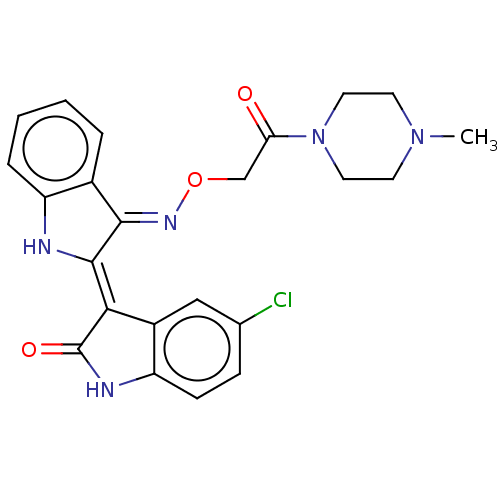
(CHEMBL5195070)Show SMILES Cl.CN1CCN(CC1)C(=O)CO\N=C1\C(\Nc2ccccc\12)=C1\C(=O)Nc2ccc(Cl)cc12 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.390 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114356
BindingDB Entry DOI: 10.7270/Q2D50RX8 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50610910

(CHEMBL5280844)Show SMILES OC[C@@H]1CCC(CO1)Nc1ncnc2[nH]cc(C(=O)c3ccc(Oc4ccccc4)cc3Cl)c12 |r| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 0.390 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein kinase FLT3
(Homo sapiens (Human)) | BDBM50593964
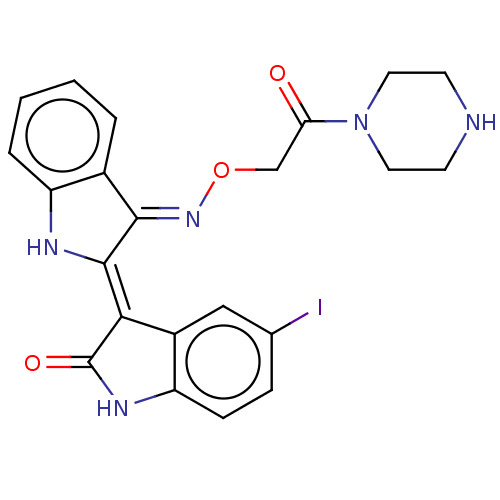
(CHEMBL5170760)Show SMILES Cl.Ic1ccc2NC(=O)\C(=C3/Nc4ccccc4/C/3=N\OCC(=O)N3CCNCC3)c2c1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.450 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114356
BindingDB Entry DOI: 10.7270/Q2D50RX8 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data