 Found 25325 hits with Last Name = 'hu' and Initial = 't'
Found 25325 hits with Last Name = 'hu' and Initial = 't' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Dihydrofolate reductase
(Mus musculus (Mouse)) | BDBM50016326
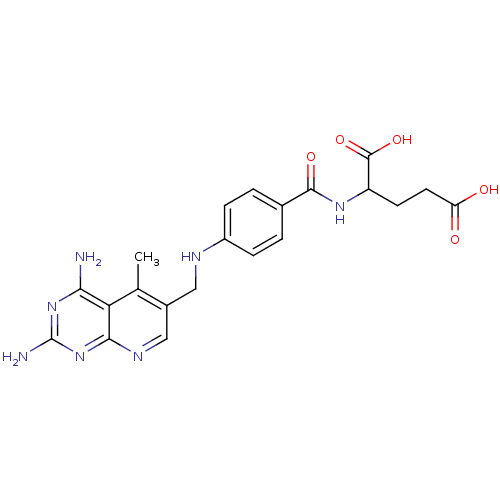
(2-{4-[(2,4-Diamino-5-methyl-pyrido[2,3-d]pyrimidin...)Show SMILES Cc1c(CNc2ccc(cc2)C(=O)NC(CCC(O)=O)C(O)=O)cnc2nc(N)nc(N)c12 Show InChI InChI=1S/C21H23N7O5/c1-10-12(9-25-18-16(10)17(22)27-21(23)28-18)8-24-13-4-2-11(3-5-13)19(31)26-14(20(32)33)6-7-15(29)30/h2-5,9,14,24H,6-8H2,1H3,(H,26,31)(H,29,30)(H,32,33)(H4,22,23,25,27,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Cornell University
Curated by ChEMBL
| Assay Description
Inhibitory activity against dihydrofolate reductase (DHFR) of L-1210 cells |
J Med Chem 31: 1209-15 (1988)
BindingDB Entry DOI: 10.7270/Q2930S6N |
More data for this
Ligand-Target Pair | |
Dihydrofolate reductase
(Mus musculus (Mouse)) | BDBM66082
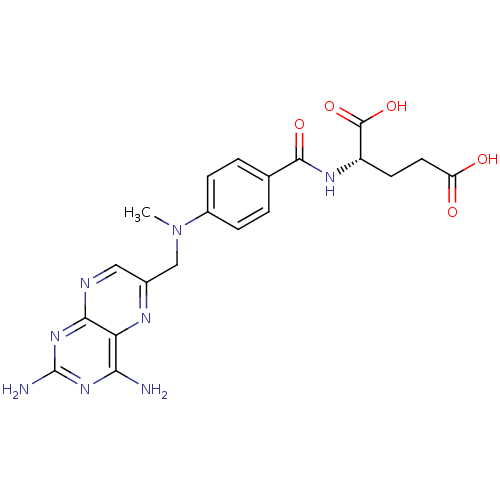
((2S)-2-[[4-[(2,4-diaminopteridin-6-yl)methyl-methy...)Show SMILES CN(Cc1cnc2nc(N)nc(N)c2n1)c1ccc(cc1)C(=O)N[C@@H](CCC(O)=O)C(O)=O Show InChI InChI=1S/C20H22N8O5/c1-28(9-11-8-23-17-15(24-11)16(21)26-20(22)27-17)12-4-2-10(3-5-12)18(31)25-13(19(32)33)6-7-14(29)30/h2-5,8,13H,6-7,9H2,1H3,(H,25,31)(H,29,30)(H,32,33)(H4,21,22,23,26,27)/t13-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| PDB
PubMed
| 0 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Cornell University
Curated by ChEMBL
| Assay Description
Inhibitory activity against dihydrofolate reductase (DHFR) of L-1210 cells |
J Med Chem 31: 1209-15 (1988)
BindingDB Entry DOI: 10.7270/Q2930S6N |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Dihydrofolate reductase
(Mus musculus (Mouse)) | BDBM50023681

(2-{4-[(2,4-Diamino-5,7-dimethyl-pyrido[2,3-d]pyrim...)Show SMILES Cc1nc2nc(N)nc(N)c2c(C)c1CNc1ccc(cc1)C(=O)NC(CCC(O)=O)C(O)=O Show InChI InChI=1S/C22H25N7O5/c1-10-14(11(2)26-19-17(10)18(23)28-22(24)29-19)9-25-13-5-3-12(4-6-13)20(32)27-15(21(33)34)7-8-16(30)31/h3-6,15,25H,7-9H2,1-2H3,(H,27,32)(H,30,31)(H,33,34)(H4,23,24,26,28,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.00200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Cornell University
Curated by ChEMBL
| Assay Description
Inhibitory activity against dihydrofolate reductase (DHFR) of L-1210 cells |
J Med Chem 31: 1209-15 (1988)
BindingDB Entry DOI: 10.7270/Q2930S6N |
More data for this
Ligand-Target Pair | |
Dihydrofolate reductase
(Mus musculus (Mouse)) | BDBM50023680

(2-{4-[(2,4-Diamino-7-phenyl-pyrido[2,3-d]pyrimidin...)Show SMILES Nc1nc(N)c2cc(CNc3ccc(cc3)C(=O)NC(CCC(O)=O)C(O)=O)c(nc2n1)-c1ccccc1 Show InChI InChI=1S/C26H25N7O5/c27-22-18-12-16(21(14-4-2-1-3-5-14)31-23(18)33-26(28)32-22)13-29-17-8-6-15(7-9-17)24(36)30-19(25(37)38)10-11-20(34)35/h1-9,12,19,29H,10-11,13H2,(H,30,36)(H,34,35)(H,37,38)(H4,27,28,31,32,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| >0.00400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Cornell University
Curated by ChEMBL
| Assay Description
Inhibitory activity against dihydrofolate reductase (DHFR) of L-1210 cells |
J Med Chem 31: 1209-15 (1988)
BindingDB Entry DOI: 10.7270/Q2930S6N |
More data for this
Ligand-Target Pair | |
Dihydrofolate reductase
(Mus musculus (Mouse)) | BDBM50023682

(2-{4-[(2,4-Diamino-5-methyl-7-phenyl-pyrido[2,3-d]...)Show SMILES Cc1c(CNc2ccc(cc2)C(=O)NC(CCC(O)=O)C(O)=O)c(nc2nc(N)nc(N)c12)-c1ccccc1 Show InChI InChI=1S/C27H27N7O5/c1-14-18(22(15-5-3-2-4-6-15)32-24-21(14)23(28)33-27(29)34-24)13-30-17-9-7-16(8-10-17)25(37)31-19(26(38)39)11-12-20(35)36/h2-10,19,30H,11-13H2,1H3,(H,31,37)(H,35,36)(H,38,39)(H4,28,29,32,33,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| >0.00400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Cornell University
Curated by ChEMBL
| Assay Description
Inhibitory activity against dihydrofolate reductase (DHFR) of L-1210 cells |
J Med Chem 31: 1209-15 (1988)
BindingDB Entry DOI: 10.7270/Q2930S6N |
More data for this
Ligand-Target Pair | |
Dihydrofolate reductase
(Mus musculus (Mouse)) | BDBM50023683
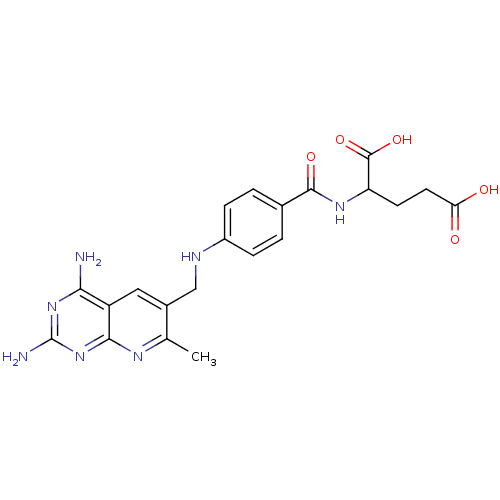
(2-{4-[(2,4-Diamino-7-methyl-pyrido[2,3-d]pyrimidin...)Show SMILES Cc1nc2nc(N)nc(N)c2cc1CNc1ccc(cc1)C(=O)NC(CCC(O)=O)C(O)=O Show InChI InChI=1S/C21H23N7O5/c1-10-12(8-14-17(22)27-21(23)28-18(14)25-10)9-24-13-4-2-11(3-5-13)19(31)26-15(20(32)33)6-7-16(29)30/h2-5,8,15,24H,6-7,9H2,1H3,(H,26,31)(H,29,30)(H,32,33)(H4,22,23,25,27,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| >0.00400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Cornell University
Curated by ChEMBL
| Assay Description
Inhibitory activity against dihydrofolate reductase (DHFR) of L-1210 cells |
J Med Chem 31: 1209-15 (1988)
BindingDB Entry DOI: 10.7270/Q2930S6N |
More data for this
Ligand-Target Pair | |
Dihydrofolate reductase
(Mus musculus (Mouse)) | BDBM50023684
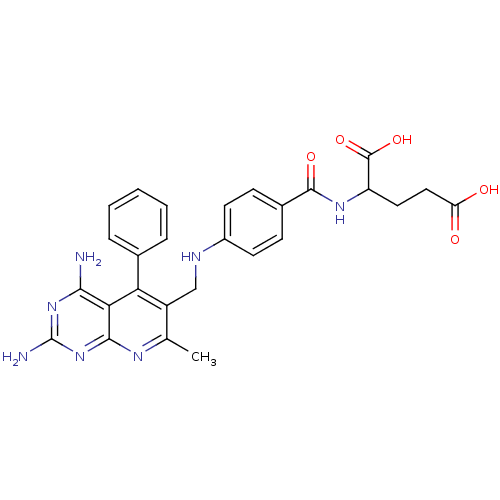
(2-{4-[(2,4-Diamino-7-methyl-5-phenyl-pyrido[2,3-d]...)Show SMILES Cc1nc2nc(N)nc(N)c2c(-c2ccccc2)c1CNc1ccc(cc1)C(=O)NC(CCC(O)=O)C(O)=O Show InChI InChI=1S/C27H27N7O5/c1-14-18(21(15-5-3-2-4-6-15)22-23(28)33-27(29)34-24(22)31-14)13-30-17-9-7-16(8-10-17)25(37)32-19(26(38)39)11-12-20(35)36/h2-10,19,30H,11-13H2,1H3,(H,32,37)(H,35,36)(H,38,39)(H4,28,29,31,33,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| >0.00400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Cornell University
Curated by ChEMBL
| Assay Description
Inhibitory activity against dihydrofolate reductase (DHFR) of L-1210 cells |
J Med Chem 31: 1209-15 (1988)
BindingDB Entry DOI: 10.7270/Q2930S6N |
More data for this
Ligand-Target Pair | |
Substance-P receptor
(Homo sapiens (Human)) | BDBM50470670
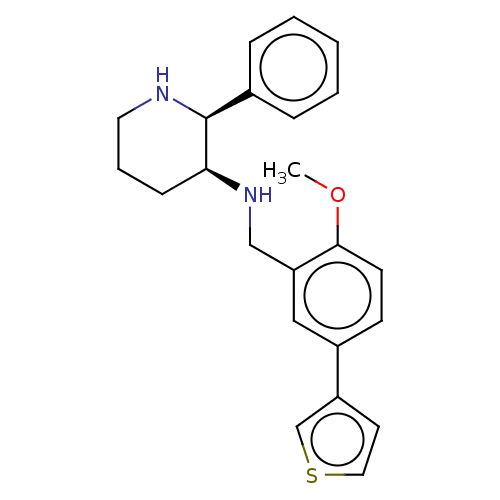
(CHEMBL149557)Show SMILES COc1ccc(cc1CN[C@H]1CCCN[C@H]1c1ccccc1)-c1ccsc1 Show InChI InChI=1S/C23H26N2OS/c1-26-22-10-9-18(19-11-13-27-16-19)14-20(22)15-25-21-8-5-12-24-23(21)17-6-3-2-4-7-17/h2-4,6-7,9-11,13-14,16,21,23-25H,5,8,12,15H2,1H3/t21-,23-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0316 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GSK UK
Curated by ChEMBL
| Assay Description
Binding affinity to Tachykinin receptor 1 stably expressed in chinese hamster ovary (CHO) cells |
J Med Chem 38: 4985-92 (1995)
Article DOI: 10.1021/jm00026a005
BindingDB Entry DOI: 10.7270/Q2S46VQ5 |
More data for this
Ligand-Target Pair | |
RAF proto-oncogene serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50557775
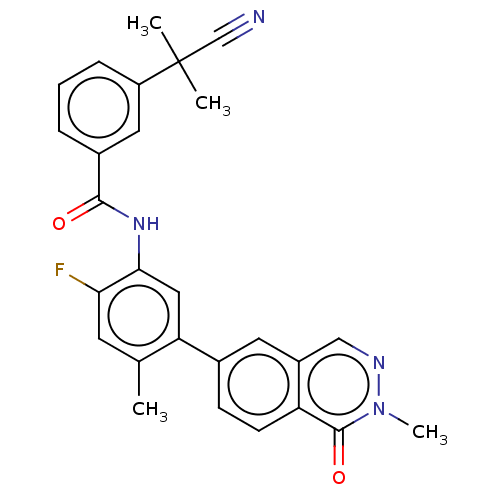
(CHEMBL4758903)Show SMILES Cc1cc(F)c(NC(=O)c2cccc(c2)C(C)(C)C#N)cc1-c1ccc2c(cnn(C)c2=O)c1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| <0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CRAF Y340D/Y341D mutant (unknown origin) assessed as using inactive phosphorylated MAP2K1 substrate preincubated for 30 mins measured a... |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.1c00063
BindingDB Entry DOI: 10.7270/Q2DZ0D0Z |
More data for this
Ligand-Target Pair | |
RAF proto-oncogene serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50580083
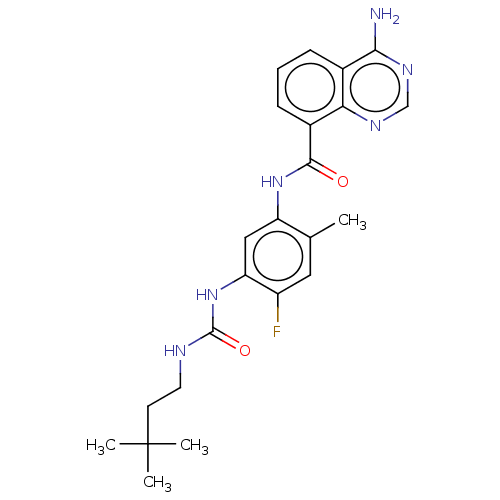
(CHEMBL5094268)Show SMILES Cc1cc(F)c(NC(=O)NCCC(C)(C)C)cc1NC(=O)c1cccc2c(N)ncnc12 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| <0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CRAF Y340D/Y341D mutant (unknown origin) using inactive MAP2K1 as substrate preincubated for 30 mins measured after 90 mins by DELFIA a... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c02085
BindingDB Entry DOI: 10.7270/Q2GT5S13 |
More data for this
Ligand-Target Pair | |
RAF proto-oncogene serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50580082
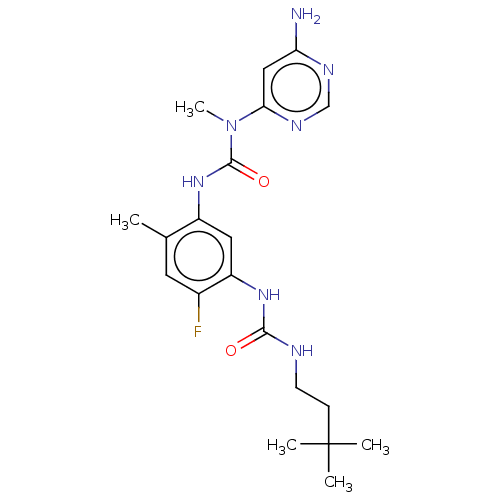
(CHEMBL5079215)Show SMILES CN(C(=O)Nc1cc(NC(=O)NCCC(C)(C)C)c(F)cc1C)c1cc(N)ncn1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| <0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CRAF Y340D/Y341D mutant (unknown origin) using inactive MAP2K1 as substrate preincubated for 30 mins measured after 90 mins by DELFIA a... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c02085
BindingDB Entry DOI: 10.7270/Q2GT5S13 |
More data for this
Ligand-Target Pair | |
RAF proto-oncogene serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50557772
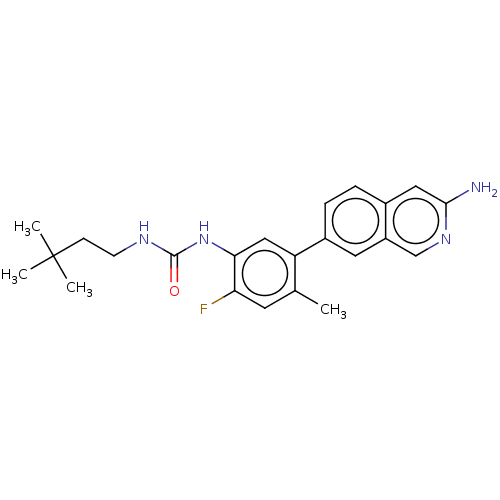
(CHEMBL4775998)Show SMILES Cc1cc(F)c(NC(=O)NCCC(C)(C)C)cc1-c1ccc2cc(N)ncc2c1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| <0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CRAF Y340D/Y341D mutant (unknown origin) assessed as using inactive phosphorylated MAP2K1 substrate preincubated for 30 mins measured a... |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.1c00063
BindingDB Entry DOI: 10.7270/Q2DZ0D0Z |
More data for this
Ligand-Target Pair | |
RAF proto-oncogene serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50557773
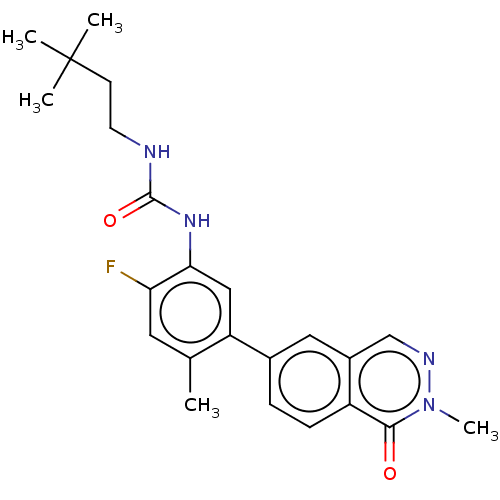
(CHEMBL4778772)Show SMILES Cc1cc(F)c(NC(=O)NCCC(C)(C)C)cc1-c1ccc2c(cnn(C)c2=O)c1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| <0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CRAF Y340D/Y341D mutant (unknown origin) assessed as using inactive phosphorylated MAP2K1 substrate preincubated for 30 mins measured a... |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.1c00063
BindingDB Entry DOI: 10.7270/Q2DZ0D0Z |
More data for this
Ligand-Target Pair | |
RAF proto-oncogene serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50580084
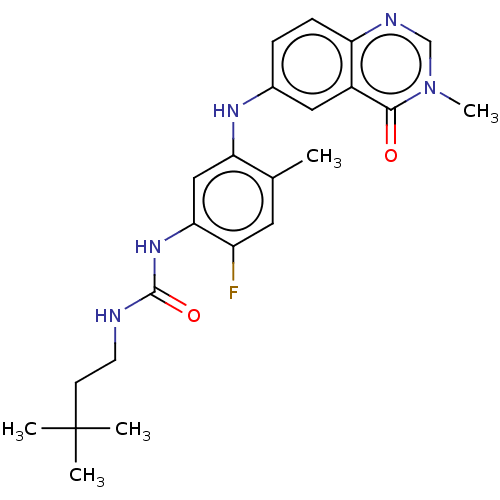
(CHEMBL5075174)Show SMILES Cc1cc(F)c(NC(=O)NCCC(C)(C)C)cc1Nc1ccc2ncn(C)c(=O)c2c1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| <0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CRAF Y340D/Y341D mutant (unknown origin) using inactive MAP2K1 as substrate preincubated for 30 mins measured after 90 mins by DELFIA a... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c02085
BindingDB Entry DOI: 10.7270/Q2GT5S13 |
More data for this
Ligand-Target Pair | |
RAF proto-oncogene serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50580080
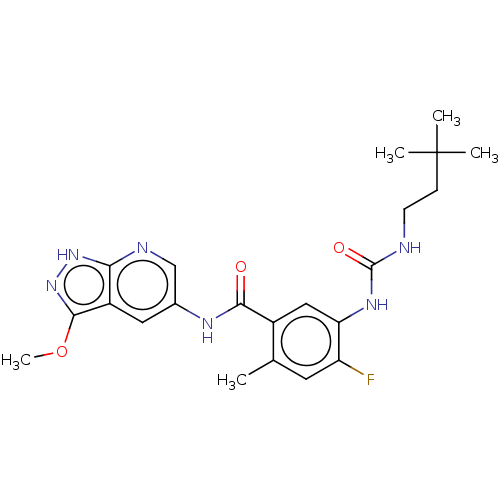
(CHEMBL5090624)Show SMILES COc1n[nH]c2ncc(NC(=O)c3cc(NC(=O)NCCC(C)(C)C)c(F)cc3C)cc12 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| <0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CRAF Y340D/Y341D mutant (unknown origin) using inactive MAP2K1 as substrate preincubated for 30 mins measured after 90 mins by DELFIA a... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c02085
BindingDB Entry DOI: 10.7270/Q2GT5S13 |
More data for this
Ligand-Target Pair | |
Substance-P receptor
(Homo sapiens (Human)) | BDBM50470675
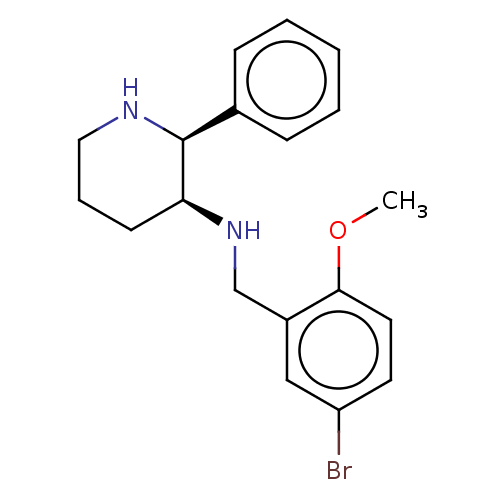
(CHEMBL344536)Show InChI InChI=1S/C19H23BrN2O/c1-23-18-10-9-16(20)12-15(18)13-22-17-8-5-11-21-19(17)14-6-3-2-4-7-14/h2-4,6-7,9-10,12,17,19,21-22H,5,8,11,13H2,1H3/t17-,19-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0501 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GSK UK
Curated by ChEMBL
| Assay Description
Binding affinity to Tachykinin receptor 1 stably expressed in chinese hamster ovary (CHO) cells |
J Med Chem 38: 4985-92 (1995)
Article DOI: 10.1021/jm00026a005
BindingDB Entry DOI: 10.7270/Q2S46VQ5 |
More data for this
Ligand-Target Pair | |
Substance-P receptor
(Homo sapiens (Human)) | BDBM50410193
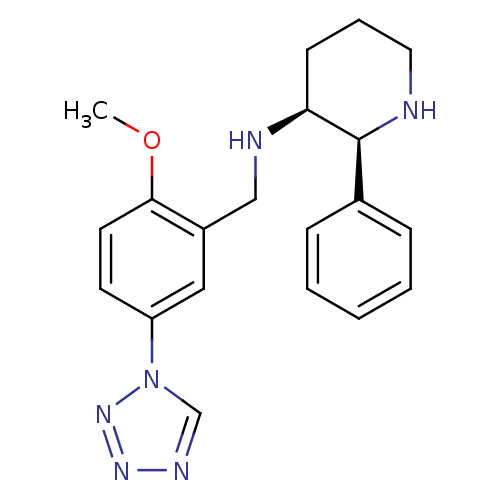
(CHEMBL356062 | GR-203040)Show SMILES COc1ccc(cc1CN[C@H]1CCCN[C@H]1c1ccccc1)-n1cnnn1 Show InChI InChI=1S/C20H24N6O/c1-27-19-10-9-17(26-14-23-24-25-26)12-16(19)13-22-18-8-5-11-21-20(18)15-6-3-2-4-7-15/h2-4,6-7,9-10,12,14,18,20-22H,5,8,11,13H2,1H3/t18-,20-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0501 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GSK UK
Curated by ChEMBL
| Assay Description
Binding affinity to Tachykinin receptor 1 stably expressed in chinese hamster ovary (CHO) cells |
J Med Chem 38: 4985-92 (1995)
Article DOI: 10.1021/jm00026a005
BindingDB Entry DOI: 10.7270/Q2S46VQ5 |
More data for this
Ligand-Target Pair | |
Substance-P receptor
(Homo sapiens (Human)) | BDBM50470678
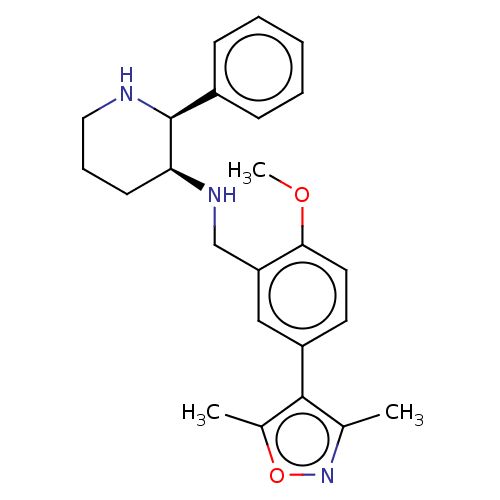
(CHEMBL146885)Show SMILES COc1ccc(cc1CN[C@H]1CCCN[C@H]1c1ccccc1)-c1c(C)noc1C Show InChI InChI=1S/C24H29N3O2/c1-16-23(17(2)29-27-16)19-11-12-22(28-3)20(14-19)15-26-21-10-7-13-25-24(21)18-8-5-4-6-9-18/h4-6,8-9,11-12,14,21,24-26H,7,10,13,15H2,1-3H3/t21-,24-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0501 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GSK UK
Curated by ChEMBL
| Assay Description
Binding affinity to Tachykinin receptor 1 stably expressed in chinese hamster ovary (CHO) cells |
J Med Chem 38: 4985-92 (1995)
Article DOI: 10.1021/jm00026a005
BindingDB Entry DOI: 10.7270/Q2S46VQ5 |
More data for this
Ligand-Target Pair | |
RAF proto-oncogene serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50557774
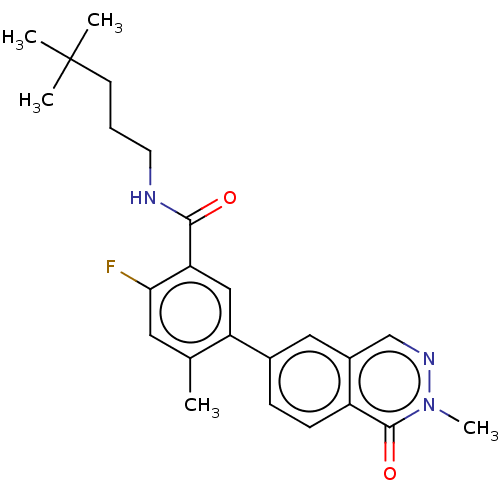
(CHEMBL4776565)Show SMILES Cc1cc(F)c(cc1-c1ccc2c(cnn(C)c2=O)c1)C(=O)NCCCC(C)(C)C | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CRAF Y340D/Y341D mutant (unknown origin) assessed as using inactive phosphorylated MAP2K1 substrate preincubated for 30 mins measured a... |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.1c00063
BindingDB Entry DOI: 10.7270/Q2DZ0D0Z |
More data for this
Ligand-Target Pair | |
RAF proto-oncogene serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50580081
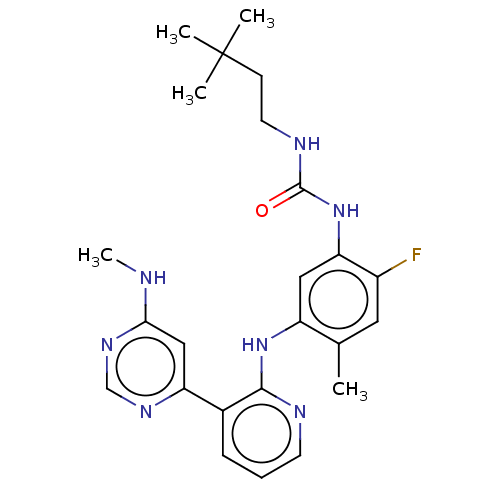
(CHEMBL5094514)Show SMILES CNc1cc(ncn1)-c1cccnc1Nc1cc(NC(=O)NCCC(C)(C)C)c(F)cc1C | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CRAF Y340D/Y341D mutant (unknown origin) using inactive MAP2K1 as substrate preincubated for 30 mins measured after 90 mins by DELFIA a... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c02085
BindingDB Entry DOI: 10.7270/Q2GT5S13 |
More data for this
Ligand-Target Pair | |
Plasma kallikrein
(Homo sapiens (Human)) | BDBM50256265
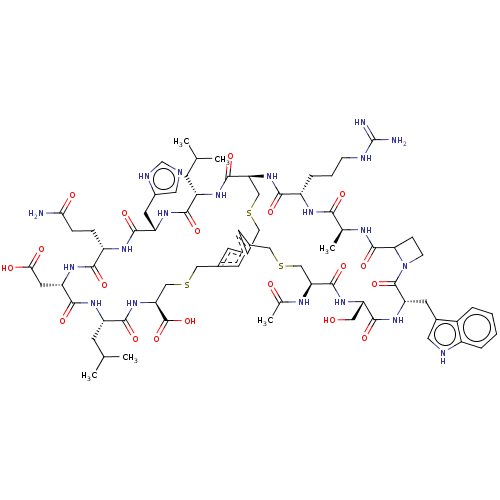
(CHEMBL4089486)Show SMILES CC(C)C[C@@H]1NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](Cc2cnc[nH]2)NC(=O)[C@H](CC(C)C)NC(=O)[C@@H]2CSCc3cc(CSC[C@H](NC1=O)C(O)=O)cc(CSC[C@H](NC(C)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](Cc1c[nH]c4ccccc14)C(=O)N1CCC1C(=O)N[C@@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N2)c3 |r| Show InChI InChI=1S/C74H104N20O19S3/c1-36(2)18-49-64(103)88-51(24-44-27-78-35-81-44)66(105)85-48(13-14-59(75)97)63(102)89-52(25-60(98)99)67(106)86-50(19-37(3)4)65(104)93-57(73(112)113)34-116-31-42-21-40-20-41(22-42)30-115-33-56(70(109)87-49)92-62(101)47(12-9-16-79-74(76)77)84-61(100)38(5)82-71(110)58-15-17-94(58)72(111)53(23-43-26-80-46-11-8-7-10-45(43)46)90-68(107)54(28-95)91-69(108)55(32-114-29-40)83-39(6)96/h7-8,10-11,20-22,26-27,35-38,47-58,80,95H,9,12-19,23-25,28-34H2,1-6H3,(H2,75,97)(H,78,81)(H,82,110)(H,83,96)(H,84,100)(H,85,105)(H,86,106)(H,87,109)(H,88,103)(H,89,102)(H,90,107)(H,91,108)(H,92,101)(H,93,104)(H,98,99)(H,112,113)(H4,76,77,79)/t38-,47-,48-,49-,50-,51-,52-,53-,54-,55-,56-,57-,58?/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bicycle Therapeutics Limited, Building 900, Babraham Research Campus , Cambridge CB22 3AT , U.K.
Curated by ChEMBL
| Assay Description
Inhibition of platelet derived growth factor receptor beta phosphorylation in MG63 cells in the presence of human plasma |
J Med Chem 61: 2823-2836 (2018)
Article DOI: 10.1021/acs.jmedchem.7b01625
BindingDB Entry DOI: 10.7270/Q2862JW6 |
More data for this
Ligand-Target Pair | |
RAF proto-oncogene serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50096279
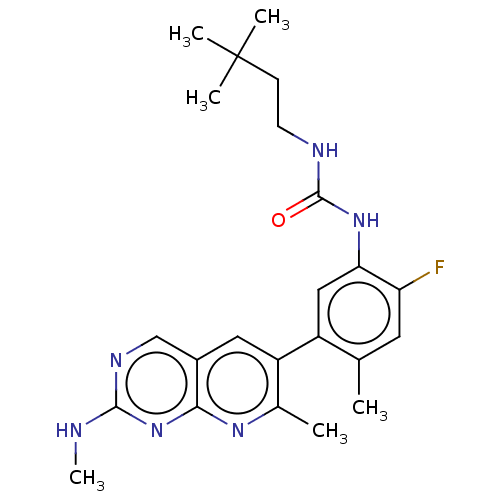
(CHEMBL3577124)Show SMILES CNc1ncc2cc(c(C)nc2n1)-c1cc(NC(=O)NCCC(C)(C)C)c(F)cc1C Show InChI InChI=1S/C18H13N3O5/c22-14(23)5-4-10-8-21-13-7-12-9(6-15(24)25)2-1-3-11(12)16(13)20-18(26)17(21)19-10/h1-5,8H,6-7H2,(H,20,26)(H,22,23)(H,24,25)/b5-4+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.0610 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CRAF Y340D/Y341D mutant (unknown origin) assessed as using inactive phosphorylated MAP2K1 substrate preincubated for 30 mins measured a... |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.1c00063
BindingDB Entry DOI: 10.7270/Q2DZ0D0Z |
More data for this
Ligand-Target Pair | |
RAF proto-oncogene serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50557770

(CHEMBL4780060)Show SMILES Cc1cc(F)c(NC(=O)NCCC(C)(C)C)cc1Nc1ccc2ncn(C)c(=O)c2c1F | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| 0.0610 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CRAF Y340D/Y341D mutant (unknown origin) assessed as using inactive phosphorylated MAP2K1 substrate preincubated for 30 mins measured a... |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.1c00063
BindingDB Entry DOI: 10.7270/Q2DZ0D0Z |
More data for this
Ligand-Target Pair | |
RAF proto-oncogene serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50096279

(CHEMBL3577124)Show SMILES CNc1ncc2cc(c(C)nc2n1)-c1cc(NC(=O)NCCC(C)(C)C)c(F)cc1C Show InChI InChI=1S/C18H13N3O5/c22-14(23)5-4-10-8-21-13-7-12-9(6-15(24)25)2-1-3-11(12)16(13)20-18(26)17(21)19-10/h1-5,8H,6-7H2,(H,20,26)(H,22,23)(H,24,25)/b5-4+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.0610 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CRAF Y340D/Y341D mutant (unknown origin) using inactive MAP2K1 as substrate preincubated for 30 mins measured after 90 mins by DELFIA a... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c02085
BindingDB Entry DOI: 10.7270/Q2GT5S13 |
More data for this
Ligand-Target Pair | |
RAF proto-oncogene serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50557770

(CHEMBL4780060)Show SMILES Cc1cc(F)c(NC(=O)NCCC(C)(C)C)cc1Nc1ccc2ncn(C)c(=O)c2c1F | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| 0.0610 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CRAF Y340D/Y341D mutant (unknown origin) using inactive MAP2K1 as substrate preincubated for 30 mins measured after 90 mins by DELFIA a... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c02085
BindingDB Entry DOI: 10.7270/Q2GT5S13 |
More data for this
Ligand-Target Pair | |
RAF proto-oncogene serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50557776
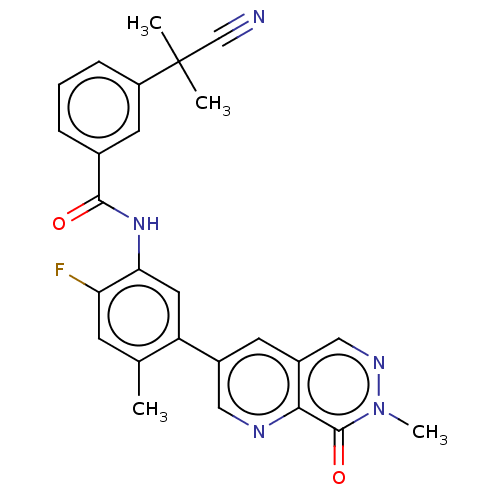
(CHEMBL4778419)Show SMILES Cc1cc(F)c(NC(=O)c2cccc(c2)C(C)(C)C#N)cc1-c1cnc2c(cnn(C)c2=O)c1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| 0.0620 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CRAF Y340D/Y341D mutant (unknown origin) assessed as using inactive phosphorylated MAP2K1 substrate preincubated for 30 mins measured a... |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.1c00063
BindingDB Entry DOI: 10.7270/Q2DZ0D0Z |
More data for this
Ligand-Target Pair | |
Substance-P receptor
(Homo sapiens (Human)) | BDBM50470669

(CHEMBL359188)Show SMILES COc1ccc(cc1CN[C@H]1CCCN[C@H]1c1ccccc1)-c1ccncc1 Show InChI InChI=1S/C24H27N3O/c1-28-23-10-9-20(18-11-14-25-15-12-18)16-21(23)17-27-22-8-5-13-26-24(22)19-6-3-2-4-7-19/h2-4,6-7,9-12,14-16,22,24,26-27H,5,8,13,17H2,1H3/t22-,24-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0631 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GSK UK
Curated by ChEMBL
| Assay Description
Binding affinity to Tachykinin receptor 1 stably expressed in chinese hamster ovary (CHO) cells |
J Med Chem 38: 4985-92 (1995)
Article DOI: 10.1021/jm00026a005
BindingDB Entry DOI: 10.7270/Q2S46VQ5 |
More data for this
Ligand-Target Pair | |
Substance-P receptor
(Homo sapiens (Human)) | BDBM50470683
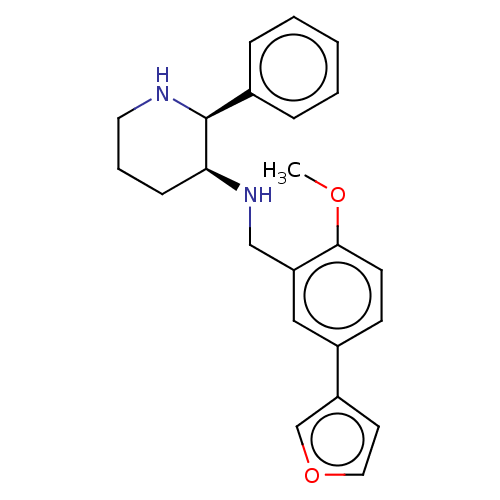
(CHEMBL356786)Show SMILES COc1ccc(cc1CN[C@H]1CCCN[C@H]1c1ccccc1)-c1ccoc1 Show InChI InChI=1S/C23H26N2O2/c1-26-22-10-9-18(19-11-13-27-16-19)14-20(22)15-25-21-8-5-12-24-23(21)17-6-3-2-4-7-17/h2-4,6-7,9-11,13-14,16,21,23-25H,5,8,12,15H2,1H3/t21-,23-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0631 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GSK UK
Curated by ChEMBL
| Assay Description
Binding affinity to Tachykinin receptor 1 stably expressed in chinese hamster ovary (CHO) cells |
J Med Chem 38: 4985-92 (1995)
Article DOI: 10.1021/jm00026a005
BindingDB Entry DOI: 10.7270/Q2S46VQ5 |
More data for this
Ligand-Target Pair | |
Plasma kallikrein
(Homo sapiens (Human)) | BDBM50256283
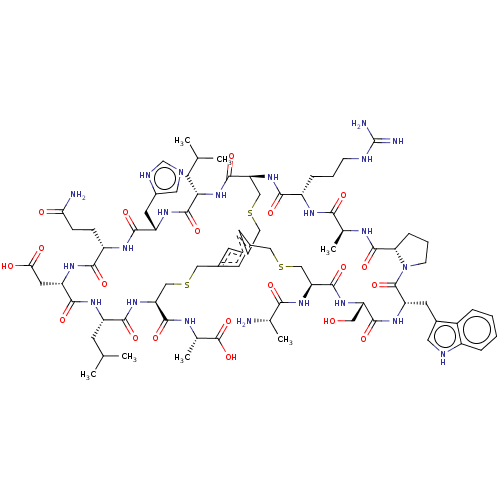
(CHEMBL4079711)Show SMILES CC(C)C[C@@H]1NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](Cc2cnc[nH]2)NC(=O)[C@H](CC(C)C)NC(=O)[C@@H]2CSCc3cc(CSC[C@H](NC1=O)C(=O)N[C@@H](C)C(O)=O)cc(CSC[C@H](NC(=O)[C@H](C)N)C(=O)N[C@@H](CO)C(=O)N[C@@H](Cc1c[nH]c4ccccc14)C(=O)N1CCC[C@H]1C(=O)N[C@@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N2)c3 |r| Show InChI InChI=1S/C79H114N22O20S3/c1-38(2)20-52-68(110)94-54(26-47-29-84-37-87-47)70(112)91-51(16-17-62(81)103)67(109)95-55(27-63(104)105)71(113)92-53(21-39(3)4)69(111)100-58(73(115)89-42(7)78(120)121)34-122-31-43-22-44-24-45(23-43)33-124-36-60(74(116)93-52)99-66(108)50(14-10-18-85-79(82)83)90-65(107)41(6)88-76(118)61-15-11-19-101(61)77(119)56(25-46-28-86-49-13-9-8-12-48(46)49)96-72(114)57(30-102)97-75(117)59(35-123-32-44)98-64(106)40(5)80/h8-9,12-13,22-24,28-29,37-42,50-61,86,102H,10-11,14-21,25-27,30-36,80H2,1-7H3,(H2,81,103)(H,84,87)(H,88,118)(H,89,115)(H,90,107)(H,91,112)(H,92,113)(H,93,116)(H,94,110)(H,95,109)(H,96,114)(H,97,117)(H,98,106)(H,99,108)(H,100,111)(H,104,105)(H,120,121)(H4,82,83,85)/t40-,41-,42-,50-,51-,52-,53-,54-,55-,56-,57-,58-,59-,60-,61-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bicycle Therapeutics Limited, Building 900, Babraham Research Campus , Cambridge CB22 3AT , U.K.
Curated by ChEMBL
| Assay Description
Inhibition of human plasma kallikrein using fluorogenic H-Pro-Phe-Arg-AMC peptide as substrate preincubated for 15 mins followed by substrate additio... |
J Med Chem 61: 2823-2836 (2018)
Article DOI: 10.1021/acs.jmedchem.7b01625
BindingDB Entry DOI: 10.7270/Q2862JW6 |
More data for this
Ligand-Target Pair | |
Substance-P receptor
(Homo sapiens (Human)) | BDBM50470672
(CHEMBL434884)Show InChI InChI=1S/C20H26N2OS/c1-23-19-11-10-17(24-2)13-16(19)14-22-18-9-6-12-21-20(18)15-7-4-3-5-8-15/h3-5,7-8,10-11,13,18,20-22H,6,9,12,14H2,1-2H3/t18-,20-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0794 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GSK UK
Curated by ChEMBL
| Assay Description
Binding affinity to Tachykinin receptor 1 stably expressed in chinese hamster ovary (CHO) cells |
J Med Chem 38: 4985-92 (1995)
Article DOI: 10.1021/jm00026a005
BindingDB Entry DOI: 10.7270/Q2S46VQ5 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50580084

(CHEMBL5075174)Show SMILES Cc1cc(F)c(NC(=O)NCCC(C)(C)C)cc1Nc1ccc2ncn(C)c(=O)c2c1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| <0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of BRAF (unknown origin) (416 to 766) inactive MAP2K1 as substrate preincubated for 30 mins measured after 90 mins by DELFIA assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c02085
BindingDB Entry DOI: 10.7270/Q2GT5S13 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50580081

(CHEMBL5094514)Show SMILES CNc1cc(ncn1)-c1cccnc1Nc1cc(NC(=O)NCCC(C)(C)C)c(F)cc1C | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| <0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of BRAF (unknown origin) (416 to 766) inactive MAP2K1 as substrate preincubated for 30 mins measured after 90 mins by DELFIA assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c02085
BindingDB Entry DOI: 10.7270/Q2GT5S13 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50580082

(CHEMBL5079215)Show SMILES CN(C(=O)Nc1cc(NC(=O)NCCC(C)(C)C)c(F)cc1C)c1cc(N)ncn1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| <0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of BRAF (unknown origin) (416 to 766) inactive MAP2K1 as substrate preincubated for 30 mins measured after 90 mins by DELFIA assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c02085
BindingDB Entry DOI: 10.7270/Q2GT5S13 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50580083

(CHEMBL5094268)Show SMILES Cc1cc(F)c(NC(=O)NCCC(C)(C)C)cc1NC(=O)c1cccc2c(N)ncnc12 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| <0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of BRAF (unknown origin) (416 to 766) inactive MAP2K1 as substrate preincubated for 30 mins measured after 90 mins by DELFIA assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c02085
BindingDB Entry DOI: 10.7270/Q2GT5S13 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50557775

(CHEMBL4758903)Show SMILES Cc1cc(F)c(NC(=O)c2cccc(c2)C(C)(C)C#N)cc1-c1ccc2c(cnn(C)c2=O)c1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| <0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of BRAF (unknown origin) (416 to 766) assessed as using inactive phosphorylated MAP2K1 substrate preincubated for 30 mins measured after 9... |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.1c00063
BindingDB Entry DOI: 10.7270/Q2DZ0D0Z |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50557770

(CHEMBL4780060)Show SMILES Cc1cc(F)c(NC(=O)NCCC(C)(C)C)cc1Nc1ccc2ncn(C)c(=O)c2c1F | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| <0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of BRAF (unknown origin) (416 to 766) inactive MAP2K1 as substrate preincubated for 30 mins measured after 90 mins by DELFIA assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c02085
BindingDB Entry DOI: 10.7270/Q2GT5S13 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50557770

(CHEMBL4780060)Show SMILES Cc1cc(F)c(NC(=O)NCCC(C)(C)C)cc1Nc1ccc2ncn(C)c(=O)c2c1F | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| <0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of BRAF (unknown origin) (416 to 766) assessed as using inactive phosphorylated MAP2K1 substrate preincubated for 30 mins measured after 9... |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.1c00063
BindingDB Entry DOI: 10.7270/Q2DZ0D0Z |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50557772

(CHEMBL4775998)Show SMILES Cc1cc(F)c(NC(=O)NCCC(C)(C)C)cc1-c1ccc2cc(N)ncc2c1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| <0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of BRAF (unknown origin) (416 to 766) assessed as using inactive phosphorylated MAP2K1 substrate preincubated for 30 mins measured after 9... |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.1c00063
BindingDB Entry DOI: 10.7270/Q2DZ0D0Z |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50557773

(CHEMBL4778772)Show SMILES Cc1cc(F)c(NC(=O)NCCC(C)(C)C)cc1-c1ccc2c(cnn(C)c2=O)c1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| <0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of BRAF (unknown origin) (416 to 766) assessed as using inactive phosphorylated MAP2K1 substrate preincubated for 30 mins measured after 9... |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.1c00063
BindingDB Entry DOI: 10.7270/Q2DZ0D0Z |
More data for this
Ligand-Target Pair | |
RAF proto-oncogene serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50557771
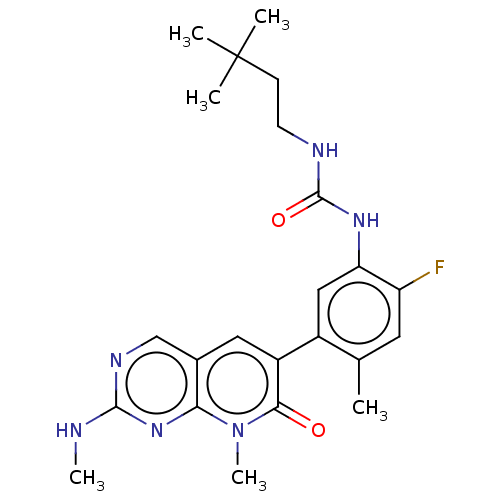
(CHEMBL4740241)Show SMILES CNc1ncc2cc(-c3cc(NC(=O)NCCC(C)(C)C)c(F)cc3C)c(=O)n(C)c2n1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0810 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CRAF Y340D/Y341D mutant (unknown origin) assessed as using inactive phosphorylated MAP2K1 substrate preincubated for 30 mins measured a... |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.1c00063
BindingDB Entry DOI: 10.7270/Q2DZ0D0Z |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50529509
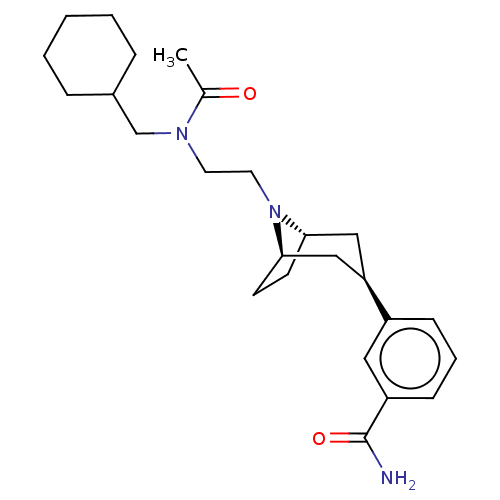
(CHEMBL4467513)Show SMILES [H][C@]12CC[C@]([H])(C[C@@H](C1)c1cccc(c1)C(N)=O)N2CCN(CC1CCCCC1)C(C)=O |r,TLB:9:7:18:3.2| Show InChI InChI=1S/C25H37N3O2/c1-18(29)27(17-19-6-3-2-4-7-19)12-13-28-23-10-11-24(28)16-22(15-23)20-8-5-9-21(14-20)25(26)30/h5,8-9,14,19,22-24H,2-4,6-7,10-13,15-17H2,1H3,(H2,26,30)/t22-,23+,24- | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Theravance Biopharma US, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-DPN from recombinant human mu opioid receptor expressed in CHO-K1 cell membranes by radioligand binding assay |
ACS Med Chem Lett 10: 1641-1647 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00406
BindingDB Entry DOI: 10.7270/Q21C219X |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50045758
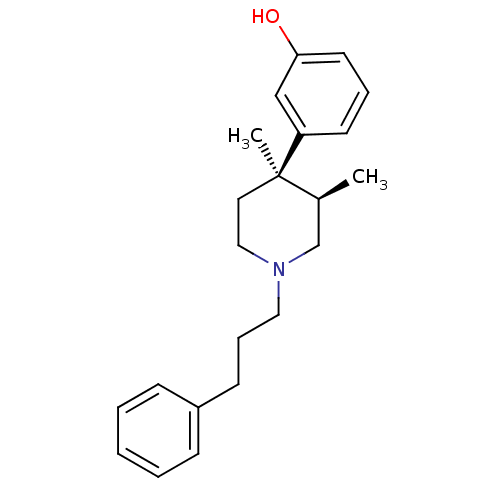
(3-[3,4-Dimethyl-1-(3-phenyl-propyl)-piperidin-4-yl...)Show SMILES C[C@H]1CN(CCCc2ccccc2)CC[C@@]1(C)c1cccc(O)c1 Show InChI InChI=1S/C22H29NO/c1-18-17-23(14-7-10-19-8-4-3-5-9-19)15-13-22(18,2)20-11-6-12-21(24)16-20/h3-6,8-9,11-12,16,18,24H,7,10,13-15,17H2,1-2H3/t18-,22+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Theravance Biopharma US, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-DPN from recombinant human mu opioid receptor expressed in CHO-K1 cell membranes by radioligand binding assay |
ACS Med Chem Lett 10: 1641-1647 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00406
BindingDB Entry DOI: 10.7270/Q21C219X |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Cavia porcellus (domestic guinea pig)) | BDBM50529515

(CHEMBL3919761)Show SMILES CS(=O)(=O)CC(=O)N(CCN1[C@H]2CC[C@@H]1C[C@@H](C2)c1cccc(c1)C(N)=O)CC1CCCCC1 |r| Show InChI InChI=1S/C26H39N3O4S/c1-34(32,33)18-25(30)28(17-19-6-3-2-4-7-19)12-13-29-23-10-11-24(29)16-22(15-23)20-8-5-9-21(14-20)26(27)31/h5,8-9,14,19,22-24H,2-4,6-7,10-13,15-18H2,1H3,(H2,27,31)/t22-,23+,24- | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Theravance Biopharma US, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-DPN from guinea pig kappa opioid receptor expressed in CHO-K1 cell membranes by radioligand binding assay |
ACS Med Chem Lett 10: 1641-1647 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00406
BindingDB Entry DOI: 10.7270/Q21C219X |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50529522

(CHEMBL4471516)Show SMILES [H][C@]12CC[C@]([H])(C[C@@H](C1)c1cccc(c1)C(N)=O)N2CCN(CC1CCCCC1)C(=O)c1ccco1 |r,TLB:9:7:18:3.2| Show InChI InChI=1S/C28H37N3O3/c29-27(32)22-9-4-8-21(16-22)23-17-24-11-12-25(18-23)31(24)14-13-30(19-20-6-2-1-3-7-20)28(33)26-10-5-15-34-26/h4-5,8-10,15-16,20,23-25H,1-3,6-7,11-14,17-19H2,(H2,29,32)/t23-,24+,25- | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Theravance Biopharma US, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-DPN from recombinant human mu opioid receptor expressed in CHO-K1 cell membranes by radioligand binding assay |
ACS Med Chem Lett 10: 1641-1647 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00406
BindingDB Entry DOI: 10.7270/Q21C219X |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50529505
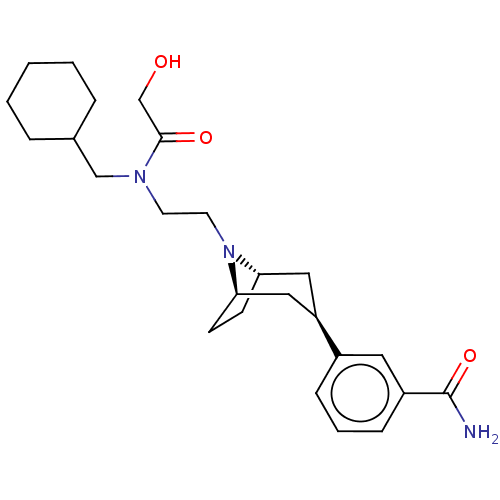
(CHEMBL3954068)Show SMILES NC(=O)c1cccc(c1)[C@@H]1C[C@@H]2CC[C@H](C1)N2CCN(CC1CCCCC1)C(=O)CO |r| Show InChI InChI=1S/C25H37N3O3/c26-25(31)20-8-4-7-19(13-20)21-14-22-9-10-23(15-21)28(22)12-11-27(24(30)17-29)16-18-5-2-1-3-6-18/h4,7-8,13,18,21-23,29H,1-3,5-6,9-12,14-17H2,(H2,26,31)/t21-,22+,23- | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Theravance Biopharma US, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-DPN from recombinant human mu opioid receptor expressed in CHO-K1 cell membranes by radioligand binding assay |
ACS Med Chem Lett 10: 1641-1647 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00406
BindingDB Entry DOI: 10.7270/Q21C219X |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Cavia porcellus (domestic guinea pig)) | BDBM50529522

(CHEMBL4471516)Show SMILES [H][C@]12CC[C@]([H])(C[C@@H](C1)c1cccc(c1)C(N)=O)N2CCN(CC1CCCCC1)C(=O)c1ccco1 |r,TLB:9:7:18:3.2| Show InChI InChI=1S/C28H37N3O3/c29-27(32)22-9-4-8-21(16-22)23-17-24-11-12-25(18-23)31(24)14-13-30(19-20-6-2-1-3-7-20)28(33)26-10-5-15-34-26/h4-5,8-10,15-16,20,23-25H,1-3,6-7,11-14,17-19H2,(H2,29,32)/t23-,24+,25- | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Theravance Biopharma US, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-DPN from guinea pig kappa opioid receptor expressed in CHO-K1 cell membranes by radioligand binding assay |
ACS Med Chem Lett 10: 1641-1647 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00406
BindingDB Entry DOI: 10.7270/Q21C219X |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50529514
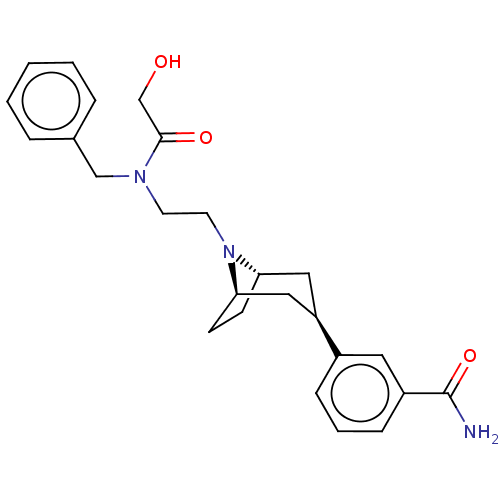
(CHEMBL3906031)Show SMILES NC(=O)c1cccc(c1)[C@@H]1C[C@@H]2CC[C@H](C1)N2CCN(Cc1ccccc1)C(=O)CO |r| Show InChI InChI=1S/C25H31N3O3/c26-25(31)20-8-4-7-19(13-20)21-14-22-9-10-23(15-21)28(22)12-11-27(24(30)17-29)16-18-5-2-1-3-6-18/h1-8,13,21-23,29H,9-12,14-17H2,(H2,26,31)/t21-,22+,23- | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Theravance Biopharma US, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-DPN from recombinant human mu opioid receptor expressed in CHO-K1 cell membranes by radioligand binding assay |
ACS Med Chem Lett 10: 1641-1647 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00406
BindingDB Entry DOI: 10.7270/Q21C219X |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50529518
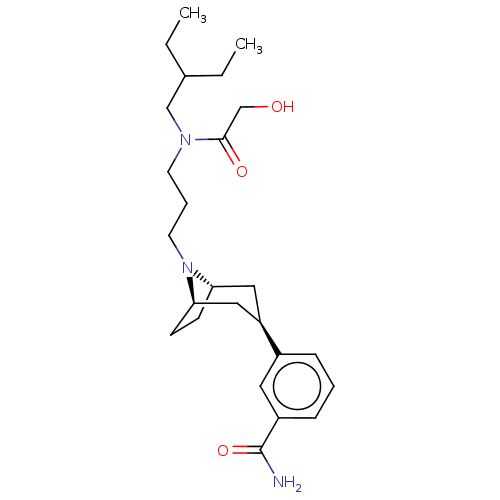
(CHEMBL4463675)Show SMILES [H][C@]12CC[C@]([H])(C[C@@H](C1)c1cccc(c1)C(N)=O)N2CCCN(CC(CC)CC)C(=O)CO |r,TLB:9:7:18:3.2| Show InChI InChI=1S/C25H39N3O3/c1-3-18(4-2)16-27(24(30)17-29)11-6-12-28-22-9-10-23(28)15-21(14-22)19-7-5-8-20(13-19)25(26)31/h5,7-8,13,18,21-23,29H,3-4,6,9-12,14-17H2,1-2H3,(H2,26,31)/t21-,22+,23- | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Theravance Biopharma US, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-DPN from recombinant human mu opioid receptor expressed in CHO-K1 cell membranes by radioligand binding assay |
ACS Med Chem Lett 10: 1641-1647 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00406
BindingDB Entry DOI: 10.7270/Q21C219X |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50529515

(CHEMBL3919761)Show SMILES CS(=O)(=O)CC(=O)N(CCN1[C@H]2CC[C@@H]1C[C@@H](C2)c1cccc(c1)C(N)=O)CC1CCCCC1 |r| Show InChI InChI=1S/C26H39N3O4S/c1-34(32,33)18-25(30)28(17-19-6-3-2-4-7-19)12-13-29-23-10-11-24(29)16-22(15-23)20-8-5-9-21(14-20)26(27)31/h5,8-9,14,19,22-24H,2-4,6-7,10-13,15-18H2,1H3,(H2,27,31)/t22-,23+,24- | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Theravance Biopharma US, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-DPN from recombinant human mu opioid receptor expressed in CHO-K1 cell membranes by radioligand binding assay |
ACS Med Chem Lett 10: 1641-1647 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00406
BindingDB Entry DOI: 10.7270/Q21C219X |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 6
(Homo sapiens (Human)) | BDBM50579331
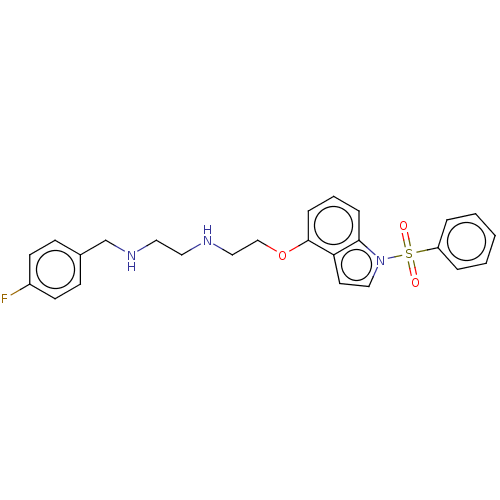
(CHEMBL4852099)Show SMILES Fc1ccc(CNCCNCCOc2cccc3n(ccc23)S(=O)(=O)c2ccccc2)cc1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-LSD from human 5HT6R expressed in CHO-K1 cell membranes incubated for 1 hr by scintillation counter method |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.113783
BindingDB Entry DOI: 10.7270/Q21V5JSC |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data